Advanced One-Step Synthesis of Vindesine Sulfate for Commercial Scale-Up
Advanced One-Step Synthesis of Vindesine Sulfate for Commercial Scale-Up
The pharmaceutical industry continuously seeks more efficient pathways for producing complex antineoplastic agents, and the preparation method disclosed in patent CN112592357B represents a significant technological leap in the synthesis of Vindesine Sulfate. This innovative approach fundamentally alters the traditional manufacturing landscape by utilizing Vinblastine Sulfate as a direct starting material, reacting it within a sealed system containing an ammonia methanol solution and a methanol solution of methoxy magnesium. Unlike conventional multi-step routes that often suffer from low yields and complex purification challenges, this novel technique achieves the conversion to Vindesine in a streamlined manner that drastically reduces reaction byproducts. The strategic implementation of a sealed nitrogen-protected environment ensures optimal reaction kinetics while maintaining safety standards, ultimately yielding a crude product that is far easier to purify than those generated by legacy methods. This breakthrough not only enhances the chemical efficiency of the process but also establishes a robust foundation for reliable vindesine sulfate supplier operations globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Vindesine has been plagued by intricate reaction sequences that introduce significant operational friction and cost inefficiencies into the supply chain. Traditional methodologies typically rely on the initial synthesis of desacetyl vindesine hydrazide as a critical intermediate, a step that demands stringent reaction conditions and often results in a plethora of difficult-to-remove side products. The requirement to synthesize and isolate this hydrazide intermediate adds considerable time and material costs, creating bottlenecks that hinder the ability to achieve cost reduction in API manufacturing. Furthermore, the harsh conditions often necessary for these transformations can lead to the degradation of the sensitive vinca alkaloid backbone, compromising the overall yield and necessitating extensive downstream processing to meet pharmacopeial standards. These cumulative inefficiencies make the conventional route less attractive for manufacturers aiming to secure a stable and economical source of high-purity anticancer intermediates.
The Novel Approach
In stark contrast to the convoluted legacy processes, the patented method introduces a direct and elegant solution by leveraging the nucleophilic properties of ammonia in the presence of methoxy magnesium to transform Vinblastine Sulfate directly into Vindesine. This one-step conversion eliminates the need for the problematic hydrazide intermediate entirely, thereby simplifying the workflow and reducing the accumulation of impurities that typically complicate purification. The use of a sealed system allows for precise control over the reaction parameters, specifically maintaining temperatures between 73-78°C for a duration of 12 hours, which optimizes the conversion rate without degrading the molecular structure. By bypassing multiple synthetic steps, this approach not only accelerates the production timeline but also significantly lowers the consumption of solvents and reagents, providing a clear pathway for the commercial scale-up of complex alkaloids. The result is a process that is not only chemically superior but also economically advantageous for large-scale enterprise production.
Mechanistic Insights into Ammonolysis and Salt Formation
The core of this synthetic advancement lies in the precise mechanistic interaction between the Vinblastine Sulfate substrate and the ammoniacal reagents under basic catalysis. The methoxy magnesium serves a dual purpose: it acts as a base to deprotonate the ammonia, generating a more potent nucleophile, and it likely coordinates with the ester functionality at the C23 position of the vinca alkaloid framework. This coordination facilitates the nucleophilic attack by the ammonia molecule, leading to the displacement of the acetyl group and the formation of the primary amide characteristic of Vindesine. The reaction is meticulously monitored using thin-layer chromatography with a specific developing agent system of dichloromethane, methanol, and ethyl acetate, ensuring that the reaction proceeds to completion before workup begins. Understanding this mechanism is crucial for R&D teams aiming to replicate the high purity levels, as the balance between the ammonia concentration and the basicity of the methoxy magnesium is critical to preventing over-reaction or hydrolysis of other sensitive functional groups within the molecule.
Following the synthesis of the crude base, the purification strategy employs a sophisticated column chromatography technique designed to separate Vindesine from closely related vinca alkaloid impurities. The use of 100-200 mesh silica gel with a gradient elution system, shifting from a dichloromethane-methanol ratio of 80:1 to 40:1, allows for the fine-tuned resolution of polar differences between the target molecule and its byproducts. Once the pure Vindesine base is isolated, the final step involves a controlled salt formation reaction with sulfuric acid in absolute ethanol. The pH is carefully adjusted to the range of 3.8-4.0, a critical window that ensures the protonation of the alkaline nitrogen atoms without causing acid-catalyzed decomposition. The subsequent addition of a non-solvent like ethyl acetate or methyl tert-butyl ether induces crystallization, effectively trapping the Vindesine Sulfate in a highly pure solid state while leaving residual impurities in the solution. 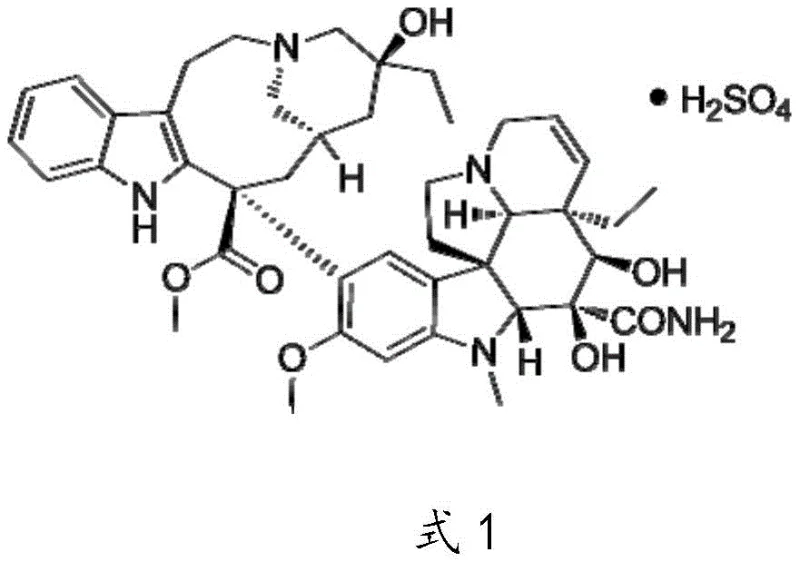
How to Synthesize Vindesine Sulfate Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction conditions to ensure reproducibility and safety on a production scale. The process begins with the preparation of the reaction vessel, which must be thoroughly purged with nitrogen to create an inert atmosphere, preventing oxidation of the sensitive alkaloid starting material. Operators must sequentially add the Vinblastine Sulfate, the ammonia methanol solution, and the methoxy magnesium solution into a pressure-resistant bottle, ensuring that the mass-to-volume ratios are maintained within the specified ranges to optimize reaction kinetics. Detailed standardized operating procedures regarding temperature ramping, pressure monitoring, and the specific workup extraction protocols using dichloromethane and semi-saturated ammonium chloride are essential for consistent results. For a comprehensive breakdown of the exact experimental parameters and safety precautions required for execution, please refer to the technical guide below.
- React Vinblastine Sulfate with ammonia methanol solution and methoxy magnesium in a sealed nitrogen-protected system at 73-78°C for 12 hours.
- Purify the resulting crude Vindesine using silica gel column chromatography with a dichloromethane and methanol gradient elution system.
- Perform salt formation by reacting pure Vindesine with sulfuric acid in absolute ethanol, followed by precipitation with an organic solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers profound benefits that extend well beyond simple chemical yield improvements, addressing key pain points in the global supply chain for oncology ingredients. By eliminating the need for expensive and unstable hydrazide intermediates, the process inherently reduces the raw material cost burden, allowing for more competitive pricing structures in the final API market. The reliance on commodity chemicals such as ammonia, methanol, and sulfuric acid, rather than specialized proprietary reagents, mitigates the risk of supply disruptions and ensures long-term availability of necessary inputs for continuous manufacturing. Furthermore, the simplification of the workflow reduces the labor hours and equipment occupancy time required per batch, translating into substantial cost savings in terms of operational overhead and facility utilization rates. These factors combine to create a resilient supply model that can better withstand market fluctuations and demand surges for critical cancer therapeutics.
- Cost Reduction in Manufacturing: The elimination of the multi-step hydrazide synthesis route removes several unit operations, including intermediate isolation and drying, which are energy-intensive and costly. By consolidating the transformation into a single reactor step, the process drastically reduces solvent consumption and waste generation, leading to lower disposal costs and a smaller environmental footprint. The use of low-cost reagents like methanolic ammonia instead of complex coupling agents further drives down the variable cost per kilogram, enabling manufacturers to offer high-purity vindesine sulfate at a more accessible price point without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing Vinblastine Sulfate as a direct feedstock leverages an established and stable supply chain for this precursor, reducing dependency on niche intermediate suppliers who may face production volatility. The robustness of the reaction conditions, which tolerate a wide range of starting material purities (greater than 90%), means that manufacturers are not forced to source ultra-high-grade raw materials at a premium, thereby broadening the pool of eligible suppliers. This flexibility ensures that production schedules can be maintained even if there are minor variations in the quality of incoming bulk materials, securing the continuity of supply for downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the use of hazardous transition metal catalysts that require complex removal steps and generate heavy metal waste streams. The straightforward workup involving extraction and crystallization is easily adaptable from laboratory glassware to industrial-sized pressure reactors, facilitating a smooth technology transfer from pilot plant to full commercial production. Additionally, the reduced number of synthetic steps minimizes the total volume of organic waste generated, simplifying compliance with increasingly stringent environmental regulations and reducing the administrative burden associated with waste management and reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and practical application scenarios. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement officers assessing the quality assurances provided by suppliers utilizing this technology. The answers reflect the specific advantages in purity control and operational simplicity that distinguish this method from older techniques, providing clarity on how the process meets rigorous pharmaceutical standards.
Q: What are the primary advantages of this new Vindesine Sulfate synthesis method?
A: The method utilizes a direct one-step ammonolysis of Vinblastine Sulfate, eliminating the need for complex hydrazide intermediates. This results in significantly fewer byproducts, simpler operational procedures, and higher overall product purity suitable for industrial application.
Q: How is high purity achieved in the final Vindesine Sulfate product?
A: High purity is ensured through a rigorous two-stage purification process: first, column chromatography removes reaction impurities from the crude base, and second, the specific salt formation conditions using absolute ethanol and controlled pH precipitate the final sulfate salt while leaving soluble impurities in the mother liquor.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process is designed for scalability. It uses common, low-cost reagents like ammonia and methanol, operates in standard pressure-resistant vessels, and avoids exotic catalysts, making it economically viable for commercial production of complex alkaloid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vindesine Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of complex oncology intermediates like Vindesine Sulfate. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical markets without sacrificing quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch meets or exceeds the requirements set forth in major pharmacopoeias. Our commitment to excellence means that we do not just supply chemicals; we provide a guarantee of consistency and reliability that allows our clients to focus on their drug development and commercialization goals with confidence.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized synthesis route can improve your bottom line. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our solutions are perfectly aligned with your quality and supply chain objectives.
