Scalable Total Synthesis of Bicuculline Oxide for Oncology Research and API Production
The pharmaceutical industry is constantly seeking robust supply chains for complex alkaloids with potent antitumor activities, and the recent disclosure of patent CN109456335B marks a significant advancement in the accessibility of Bicuculline Oxide. This specific intellectual property details a novel total synthesis method that bypasses the traditional limitations of plant extraction, offering a reliable pathway for producing this valuable pharmaceutical intermediate. The technology leverages a multi-step organic synthesis route starting from readily available 3,4-dimethoxyphenylacetic acid, transforming it through a series of precise chemical modifications including bromination, amidation, and cyclization. For R&D directors and procurement specialists, this patent represents a critical opportunity to secure high-purity materials without relying on fluctuating natural sources. The method achieves an overall yield exceeding 45%, which is a substantial improvement over previous methods, ensuring that research and development pipelines remain uninterrupted by supply shortages. By adopting this synthetic approach, manufacturers can guarantee the consistent quality and stereochemical integrity required for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of Bicuculline Oxide and related aporphine alkaloids has been heavily dependent on extraction from natural plant sources such as Lindera megaphylla or Dicentra spectabilis. This reliance introduces severe supply chain vulnerabilities, as the content of these bioactive compounds in nature is exceptionally low, often making large-scale isolation economically unfeasible. Furthermore, natural extraction processes are plagued by variability due to seasonal changes, geographical differences, and agricultural conditions, leading to inconsistent batch-to-batch purity. The purification of natural extracts often requires extensive chromatography to separate the target alkaloid from a complex matrix of structurally similar impurities, which drastically increases processing time and solvent consumption. For procurement managers, this translates to unpredictable lead times and inflated costs that can hinder the progress of preclinical and clinical studies. Additionally, the environmental footprint of processing tons of plant material to obtain grams of active ingredient is unsustainable in the context of modern green chemistry initiatives.
The Novel Approach
In stark contrast, the synthetic route disclosed in the patent data offers a deterministic and scalable solution that decouples production from agricultural constraints. By utilizing 3,4-dimethoxyphenylacetic acid as a stable starting material, the process ensures a consistent input quality that is not subject to harvest variations. The route is designed with operational simplicity in mind, utilizing common reagents like phosphorus oxychloride and standard reducing agents that are readily available in industrial chemical markets. This shift from extraction to synthesis allows for precise control over reaction parameters such as temperature and molar ratios, directly influencing the impurity profile and final yield. The ability to synthesize the compound from scratch means that production capacity can be ramped up from 100 kgs to 100 MT scale simply by increasing reactor size, rather than sourcing more raw plant material. This scalability is crucial for meeting the growing demand for oncology research materials and ensures a continuous supply for commercial API manufacturing.
Mechanistic Insights into Pd-Catalyzed Cyclization and Mn(III) Oxidation
The core chemical innovation of this synthesis lies in the strategic construction of the tetracyclic skeleton through a palladium-catalyzed intramolecular coupling reaction. In step 6 of the process, Compound 6 undergoes a cyclization in the presence of palladium acetate and tricyclohexylphosphine, which facilitates the formation of the critical carbon-carbon bond required to close the ring system. This transition metal-catalyzed step is highly efficient, operating at elevated temperatures between 120-135°C to drive the reaction to completion with yields above 82%. The use of a phosphine ligand stabilizes the palladium center, preventing premature catalyst deactivation and ensuring high turnover numbers. Following this, the synthesis employs a rigorous reduction step using lithium aluminum hydride to convert the intermediate into the amine precursor, setting the stage for the final oxidation. The mechanistic precision here ensures that the stereochemistry is maintained throughout the sequence, which is vital for the biological activity of the final alkaloid.
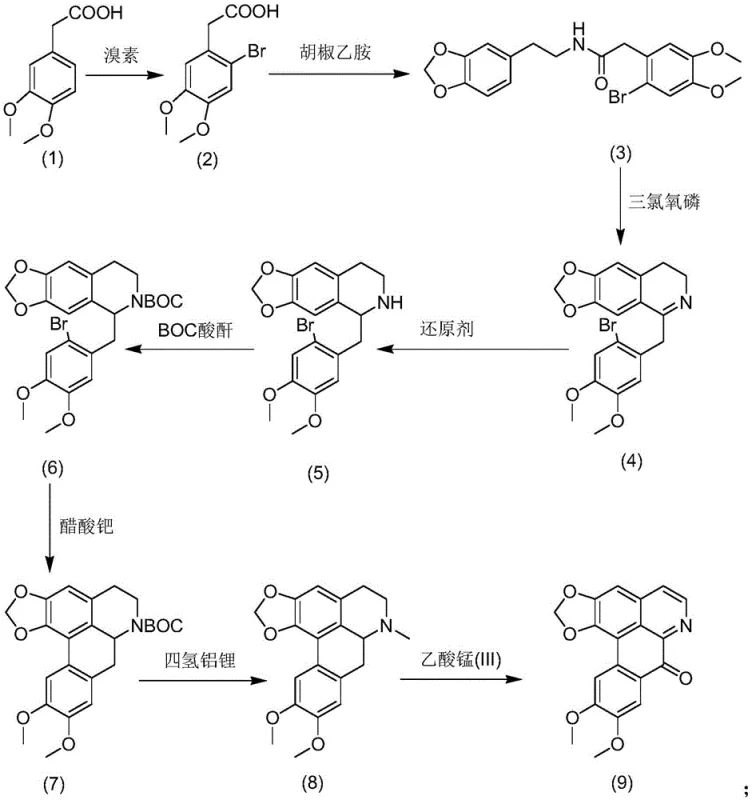
Following the construction of the core structure, the final transformation involves an oxidation step mediated by manganese(III) acetate to generate the characteristic ketone functionality of Bicuculline Oxide. This oxidation is performed in glacial acetic acid under reflux conditions, where the manganese species acts as a single-electron oxidant to introduce the oxygen atom at the specific position on the D-ring. The control of this oxidation step is critical, as over-oxidation can lead to degradation products that are difficult to separate. The patent specifies a molar ratio of manganese acetate to substrate of approximately 2 to 4 times by mass, ensuring complete conversion while minimizing side reactions. Impurity control is further enhanced by intermediate recrystallization steps using solvents like methanol and ethanol, which remove byproducts before they can propagate through subsequent steps. This layered approach to purification ensures that the final crude product requires minimal downstream processing to meet stringent pharmaceutical specifications.
How to Synthesize Bicuculline Oxide Efficiently
The synthesis of Bicuculline Oxide described in this technical insight follows a logical eight-step sequence that balances yield with operational safety. The process begins with the bromination of the starting acid, followed by amidation and cyclization to build the molecular framework. Each step has been optimized for industrial feasibility, utilizing standard workup procedures such as aqueous washes and solvent evaporation. The detailed standardized synthesis steps see the guide below for specific reaction conditions and stoichiometry.
- Bromination of 3,4-dimethoxyphenylacetic acid followed by amidation with piperonylethylamine to form the amide intermediate.
- Cyclization using phosphorus oxychloride and subsequent reduction to form the tetrahydroisoquinoline core structure.
- Pd-catalyzed intramolecular coupling followed by LAH reduction and final Mn(III) mediated oxidation to yield Bicuculline Oxide.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the transition to this synthetic route offers profound advantages in terms of cost stability and logistical reliability. By eliminating the dependency on rare plant sources, manufacturers can secure long-term contracts with chemical raw material suppliers, locking in prices and ensuring uninterrupted production schedules. The use of commodity chemicals like acetic acid, thionyl chloride, and common organic solvents means that the supply chain is resilient to geopolitical disruptions that might affect botanical imports. Furthermore, the simplified purification protocol reduces the consumption of expensive chromatography media and solvents, leading to substantial cost savings in the overall manufacturing budget. The high yield at each individual step compounds to a robust overall process efficiency, minimizing waste generation and maximizing the output per batch. This efficiency directly translates to a lower cost of goods sold (COGS), allowing pharmaceutical companies to allocate more resources to clinical development rather than raw material acquisition.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive and inefficient natural extraction processes, which typically suffer from low recovery rates and high solvent usage. By utilizing catalytic amounts of palladium and stoichiometric oxidants, the process minimizes the consumption of high-cost reagents while maintaining high throughput. The ability to recycle solvents like dichloromethane and chloroform further drives down operational expenses, making the production of this complex alkaloid economically viable on a commercial scale. Additionally, the removal of transition metal catalysts is streamlined through standard aqueous workups, avoiding the need for specialized scavenging resins that add to the production cost.
- Enhanced Supply Chain Reliability: Sourcing 3,4-dimethoxyphenylacetic acid and other key reagents is significantly more reliable than harvesting specific plant species that may be subject to seasonal or regulatory restrictions. This synthetic approach ensures that production can continue year-round without interruption, providing a consistent flow of materials to R&D and manufacturing facilities. The robustness of the chemical steps means that batch failures are rare, reducing the risk of stockouts that could delay critical drug development timelines. Suppliers can offer firmer delivery commitments, knowing that their production capacity is limited only by reactor availability rather than biological growth cycles.
- Scalability and Environmental Compliance: The process is designed for easy scale-up, with reaction conditions that can be safely translated from laboratory glassware to industrial stainless steel reactors. The waste streams generated are primarily organic solvents and inorganic salts, which can be managed through standard wastewater treatment and solvent recovery systems, ensuring compliance with environmental regulations. By avoiding the massive biomass waste associated with plant extraction, this method aligns with green chemistry principles and reduces the overall environmental footprint of the manufacturing process. This sustainability profile is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Bicuculline Oxide synthesized via this patented method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance for industry professionals.
Q: Why is total synthesis preferred over extraction for Bicuculline Oxide?
A: Natural extraction yields are extremely low due to the scarce content of DCO alkaloids in plants like Lindera megaphylla. Total synthesis provides a consistent, scalable supply chain independent of agricultural variables, ensuring reliable availability for clinical research.
Q: What are the critical purity controls in this synthetic route?
A: The process utilizes specific recrystallization steps using methanol or ethanol after key intermediates (Compound 2, 6, 7) to remove impurities. Final purification via silica gel column chromatography ensures high-purity specifications suitable for biological testing.
Q: How does the Pd-catalyzed step impact manufacturing costs?
A: While palladium is a precious metal, the catalytic amount used (2-3% mass) is minimal compared to the value of the final API. The high yield (over 82% in this step) and the ability to recover catalyst residues through standard workups significantly offset the initial metal cost.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bicuculline Oxide Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of securing high-quality intermediates for oncology research and API development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and speed. Our facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of Bicuculline Oxide meets the highest international standards. We are committed to supporting your drug discovery pipeline with reliable materials that enable consistent and reproducible research results.
We invite you to discuss your specific requirements with our technical procurement team to explore how this synthetic route can optimize your supply chain. Request a Customized Cost-Saving Analysis today to understand the economic benefits of switching to this scalable method. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project's unique demands, ensuring a seamless transition to commercial manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
