Advanced Catalytic Hydrogenation Strategy for Commercial Scale-up of Complex Pharmaceutical Intermediates
The global pharmaceutical landscape is constantly evolving, demanding more efficient and cost-effective synthetic routes for critical non-steroidal anti-inflammatory drugs (NSAIDs). A pivotal advancement in this domain is documented in Chinese Patent CN100480224C, which outlines a robust preparation method for Nabumetone, a widely prescribed medication for rheumatic and rheumatoid arthritis. This patent introduces a transformative catalytic hydrogenation strategy that replaces traditional, expensive precious metal catalysts with cost-efficient Raney Nickel variants. For R&D Directors and Procurement Managers seeking a reliable pharmaceutical intermediates supplier, this technology represents a significant leap forward in process economics and operational stability. By shifting the paradigm from Palladium on Carbon to Raney-Ni Wn (where n=2~7), Raney Cu, or Raney Co, the method addresses long-standing issues regarding catalyst recovery, equipment corrosion, and overall production costs. The technical breakthrough lies not just in the substitution of materials, but in the precise optimization of reaction conditions that maintain high selectivity while drastically simplifying the workflow. This report provides a deep-dive analysis of the mechanistic advantages and commercial implications of this patented route, offering actionable insights for stakeholders aiming to optimize their supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Nabumetone has relied heavily on condensation reactions followed by catalytic reduction using Palladium on Carbon (Pd/C). While chemically feasible, this conventional approach presents substantial bottlenecks for commercial scale-up of complex pharmaceutical intermediates. The primary drawback is the exorbitant cost associated with palladium, a precious metal that significantly inflates the raw material expenditure. Furthermore, the selectivity of Pd/C in this specific transformation is often suboptimal, leading to the formation of unwanted by-products that complicate downstream purification. From a supply chain perspective, the recovery and recycling of palladium catalysts are technically demanding and often inconsistent, leading to variable batch quality and potential metal contamination issues. The equipment corrosion caused by the acidic conditions often required in conjunction with these catalysts further exacerbates maintenance costs and downtime. Additionally, the sensitivity of Pd/C to poisoning and the need for strict inert atmospheres add layers of operational complexity that hinder efficient large-scale manufacturing. These cumulative factors render the traditional Pd/C route less attractive for manufacturers aiming for cost reduction in pharmaceutical intermediates manufacturing, especially in a market where margin compression is a constant pressure.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this process by employing Raney-Ni Wn, Raney Cu, or Raney Co as the primary hydrogenation catalysts. This strategic shift eliminates the dependency on precious metals, immediately offering a pathway for substantial cost savings without compromising on reaction efficiency. The new method utilizes 6-methoxy-2-naphthaldehyde as the starting feed, which undergoes condensation with acetoacetic ester to form an unsaturated intermediate, followed by the critical catalytic hydrogenation step. What sets this approach apart is its remarkable tolerance to mild reaction conditions, operating effectively at pressures between 0.1~4.0MPa and temperatures ranging from 20~100℃. This gentleness not only preserves the integrity of the sensitive carbonyl groups, preventing over-reduction, but also significantly reduces the corrosive stress on reactor vessels. The process is characterized by its simplicity and robustness, allowing for easier handling and safer operations compared to the finicky nature of palladium systems. By achieving a two-step overall yield of more than 80% with a product purity exceeding 99% (GC content), this method proves that cost-effective catalysis does not have to come at the expense of quality. It stands as a testament to how intelligent catalyst selection can drive both technical excellence and commercial viability in the fine chemical sector.
Mechanistic Insights into Raney-Ni Catalyzed Hydrogenation
At the heart of this synthetic innovation is the mechanistic behavior of Raney Nickel during the hydrogenation of the intermediate 3-alkoxycarbonyl-4-(6-methoxy-2-naphthyl)but-2-ene-2-ketone. Unlike noble metals that may promote non-selective hydrogenolysis, the specific crystal structure and surface activity of Raney-Ni Wn (n=2~7) facilitate a highly chemoselective reduction of the carbon-carbon double bond while leaving the ketone and ester functionalities intact. The catalyst, used at a weight ratio of 5-10% relative to the substrate, provides an optimal surface area for hydrogen adsorption and activation. The reaction mechanism involves the synchronous addition of hydrogen atoms across the olefinic bond, a process that is kinetically favored under the specified mild pressure of 0.1~1.0MPa. This selectivity is crucial because over-reduction of the carbonyl group would lead to alcohol by-products, severely impacting the purity profile required for API production. The use of solvents such as methanol, ethanol, or mixtures with toluene further modulates the solvation environment, ensuring that the substrate remains accessible to the catalyst surface without promoting side reactions. This precise control over the reaction trajectory is what enables the consistent achievement of high yields and purity, making it a superior choice for R&D teams focused on impurity control and process robustness.
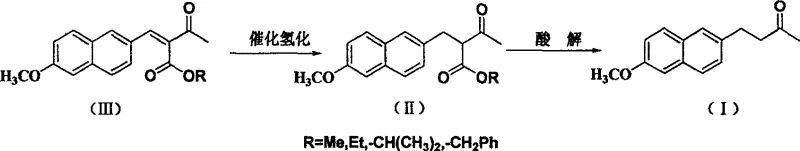
Furthermore, the impurity control mechanism inherent in this Raney-Ni catalyzed route is a significant advantage for regulatory compliance. The mild conditions (20~60℃ preferred) minimize thermal degradation and polymerization of the intermediate, which are common sources of difficult-to-remove impurities in harsher catalytic systems. The subsequent acidolysis step, conducted with hydrochloric acid, efficiently cleaves the ester group to yield the final Nabumetone structure without generating complex waste streams. The crystallization from dehydrated alcohol serves as a final polishing step, leveraging the high intrinsic purity of the crude product to achieve specifications of mp79.5~81℃ and GC content ≥99%. For Quality Assurance and R&D Directors, this means a more predictable impurity profile and a reduced burden on analytical testing. The elimination of heavy metal residues, which is a persistent challenge with Pd/C, simplifies the toxicological assessment and ensures that the final product meets stringent international pharmacopoeia standards. This mechanistic clarity provides the confidence needed for technology transfer and commercial validation.
How to Synthesize Nabumetone Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters to ensure reproducibility and safety at scale. The process begins with the condensation of 6-methoxy-2-naphthaldehyde and methyl acetoacetate in a solvent like cyclohexane, catalyzed by piperidine p-toluenesulfonate under reflux. Once the unsaturated intermediate is isolated, the critical hydrogenation phase commences in an autoclave. The choice of Raney-Ni catalyst grade (e.g., W-2, W-4) and the precise control of hydrogen pressure are paramount to success. The reaction mixture, typically dissolved in methanol or a methanol-toluene blend, is subjected to hydrogen gas at controlled pressures (0.1-1.0 MPa) and temperatures (25-60℃) for a duration of 2-20 hours, depending on the specific catalyst activity. Monitoring via TLC ensures complete conversion before proceeding to the acidolysis stage. The detailed standardized synthesis steps, including specific work-up procedures and safety protocols for handling pyrophoric Raney catalysts, are outlined in the technical guide below.
- Condense 6-methoxy-2-naphthaldehyde with methyl acetoacetate in cyclohexane using piperidine p-toluenesulfonate to form the unsaturated intermediate.
- Perform catalytic hydrogenation on the intermediate using Raney-Ni Wn (5-10% weight ratio) in methanol at 0.1-1.0 MPa and 20-60°C.
- Conduct acidolysis with hydrochloric acid followed by dehydration and ethanol recrystallization to isolate the final Nabumetone product with >99% GC content.
Commercial Advantages for Procurement and Supply Chain Teams
For Procurement Managers and Supply Chain Heads, the transition to this Raney-Ni based methodology offers compelling strategic advantages that extend far beyond simple chemistry. The primary value proposition lies in the drastic simplification of the supply chain for catalysts. By removing the need for palladium, a metal subject to extreme price volatility and geopolitical supply constraints, manufacturers can secure a more stable and predictable cost structure. This shift directly contributes to cost reduction in pharmaceutical intermediates manufacturing by eliminating the expensive and complex recovery loops associated with precious metals. The operational simplicity of the new process also translates into reduced downtime and lower maintenance costs, as the equipment is subjected to less corrosive stress. Furthermore, the high selectivity of the reaction minimizes waste generation, aligning with increasingly strict environmental regulations and reducing the costs associated with waste disposal. These factors combine to create a manufacturing process that is not only economically superior but also more resilient to external market shocks, ensuring a continuous and reliable supply of high-quality intermediates for downstream API production.
- Cost Reduction in Manufacturing: The substitution of expensive Palladium on Carbon with Raney Nickel represents a fundamental shift in the cost dynamics of Nabumetone production. Since Raney Nickel is a base metal catalyst, its acquisition cost is a fraction of that of precious metals, leading to immediate raw material savings. Moreover, the process eliminates the need for sophisticated metal recovery units, which are capital-intensive to install and operate. The high selectivity of the reaction reduces the formation of by-products, which in turn lowers the consumption of solvents and reagents during the purification stages. This cumulative effect results in a significantly leaner cost structure, allowing manufacturers to offer more competitive pricing without sacrificing margins. The economic efficiency is further enhanced by the ability to use standard stainless steel equipment without requiring exotic alloys to resist corrosion, thereby reducing capital expenditure for new production lines.
- Enhanced Supply Chain Reliability: Supply chain resilience is critical in the pharmaceutical industry, and this method strengthens it by diversifying the source of critical catalysts. Unlike palladium, which is sourced from a limited number of geographic regions and is prone to supply disruptions, Raney Nickel is widely available from multiple global suppliers. This availability ensures that production schedules are not held hostage by raw material shortages. Additionally, the mild reaction conditions reduce the risk of batch failures due to thermal runaway or catalyst deactivation, leading to more consistent output and reliable delivery times. The robustness of the process allows for flexible manufacturing strategies, enabling producers to scale up or down in response to market demand without significant re-engineering. For Supply Chain Heads, this translates to a lower risk profile and the ability to guarantee lead times for high-purity pharmaceutical intermediates to their global clients.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to the plant is often fraught with challenges, but the simplicity of this Raney-Ni route facilitates a smoother transition to commercial scale-up of complex pharmaceutical intermediates. The reaction parameters are forgiving and easy to control in large reactors, minimizing the engineering bottlenecks often seen with sensitive precious metal catalysis. From an environmental perspective, the process generates less hazardous waste, particularly heavy metal-contaminated residues, which simplifies compliance with environmental protection regulations. The reduced corrosive nature of the reaction mixture also extends the lifespan of production equipment, reducing the frequency of replacements and the associated environmental footprint of manufacturing infrastructure. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, a key factor for modern procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for their own production lines. The clarity provided here aims to bridge the gap between theoretical patent claims and practical industrial application, ensuring that all stakeholders have a unified understanding of the process capabilities and limitations.
Q: Why is Raney-Ni preferred over Palladium on Carbon for Nabumetone production?
A: Raney-Ni offers significantly lower catalyst costs compared to precious metal Pd/C, reduces equipment corrosion, and provides excellent chemoselectivity without requiring complex recovery systems, making it ideal for large-scale manufacturing.
Q: What are the critical reaction parameters for the hydrogenation step?
A: The process operates optimally at a hydrogen pressure of 0.1 to 1.0 MPa and temperatures between 20°C and 60°C. Maintaining these mild conditions prevents over-reduction of the carbonyl group and ensures high selectivity.
Q: How does this method impact the impurity profile of the final API?
A: The use of Raney-Ni under controlled mild conditions minimizes side reactions and by-product formation, resulting in a crude product with high purity that simplifies downstream purification and ensures consistent quality for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nabumetone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN100480224C to maintain competitiveness in the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards. We understand that for R&D Directors, the consistency of the impurity profile is just as important as the yield, and our state-of-the-art facilities are designed to deliver exactly that. By leveraging our expertise in catalytic hydrogenation and process optimization, we help our partners unlock the full commercial potential of their pharmaceutical intermediates, ensuring a supply chain that is both robust and cost-effective.
We invite you to collaborate with us to explore how this optimized Nabumetone synthesis can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out to request specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this Raney-Ni catalyzed process. Whether you are looking to secure a long-term supply of high-purity intermediates or need assistance with process development, NINGBO INNO PHARMCHEM is equipped to support your strategic goals with precision and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
