Scalable Manganese-Promoted Synthesis of Dibenzophosphole Derivatives for Advanced Optoelectronic Applications
Scalable Manganese-Promoted Synthesis of Dibenzophosphole Derivatives for Advanced Optoelectronic Applications
The rapidly evolving landscape of organic electronics demands increasingly sophisticated molecular architectures, particularly within the realm of phosphorus-containing heterocycles. Patent CN103214521A introduces a transformative methodology for the preparation of dibenzophosphole derivatives, a class of compounds critical for constructing novel electroluminescent materials used in OLEDs and solar cells. This innovation leverages manganese acetate as a cost-effective promoter to facilitate the intramolecular cyclization of readily available biphenylphosphine oxide derivatives. By shifting away from hazardous organometallic reagents towards a transition-metal promoted oxidative coupling, this technology addresses significant bottlenecks in the manufacturing of high-purity OLED material intermediates. The process operates under remarkably mild thermal conditions, eliminating the need for cryogenic temperatures or rigorous inert atmospheres, thereby establishing a new benchmark for the reliable dibenzophosphole derivative supplier seeking to optimize production efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dibenzophosphole scaffolds has relied heavily on stepwise reactions involving highly reactive and unstable organolithium species, such as xenyl lithium or diethylamino lithium. These traditional pathways necessitate stringent anhydrous and oxygen-free environments, often requiring specialized glassware and cryogenic cooling to prevent catastrophic decomposition or side reactions. Furthermore, the precursors required for these lithiation steps, such as aryl dichloride phosphines or terphenylyl dichloride phosphines, are frequently difficult to source commercially and command premium pricing due to their complex synthesis. The operational hazards associated with handling pyrophoric reagents on a large scale pose substantial safety risks and increase the overall cost of goods sold, making the commercial scale-up of complex organophosphorus compounds economically challenging for many manufacturers.
The Novel Approach
In stark contrast, the disclosed invention utilizes widely sourced biphenylphosphine oxide derivatives as starting materials, reacting them with manganese acetate in common organic solvents to achieve efficient ring closure. This manganese-promoted cyclization proceeds smoothly at temperatures ranging from 20°C to 120°C, completely bypassing the need for moisture-sensitive reagents or inert gas protection. The versatility of this method is evidenced by its compatibility with a broad spectrum of substituents, including alkyl, alkoxy, halogen, and cyano groups, allowing for the facile tuning of electronic properties for specific optoelectronic applications. By simplifying the reaction setup to a standard heating protocol followed by straightforward chromatographic purification, this approach drastically reduces both the technical barrier to entry and the operational expenditure associated with producing these valuable intermediates.

Mechanistic Insights into Manganese-Promoted Cyclization
The core of this technological advancement lies in the ability of manganese acetate to act as a single-electron oxidant or Lewis acid promoter that activates the C-H bond adjacent to the phosphorus center. While the exact mechanistic cycle may involve radical intermediates generated via single-electron transfer (SET) from the phosphine oxide to the manganese species, the net result is an oxidative dehydrogenative coupling that forms the rigid dibenzophosphole core. This mechanism avoids the formation of unstable carbanionic intermediates typical of lithiation routes, thereby minimizing the generation of regio-isomeric impurities that are difficult to separate. The tolerance of the catalytic system to various functional groups suggests that the manganese species selectively targets the specific ortho-position required for cyclization without interfering with sensitive moieties like esters, nitriles, or halides present on the aromatic rings.
From an impurity control perspective, the mild reaction conditions play a pivotal role in ensuring high product purity. Traditional high-temperature pyrolysis or aggressive Lewis acid methods often lead to skeletal rearrangements or over-oxidation of the phosphorus center; however, the controlled oxidation potential of manganese acetate mitigates these risks. The resulting crude products typically exhibit clean profiles, as demonstrated by the high isolated yields reported in the patent examples, which often exceed 80%. For instance, the synthesis of 9-phenyl-phosphorus fluorenes-9-oxide proceeds with an impressive 85% yield, indicating that the reaction kinetics favor the desired cyclization pathway overwhelmingly over competing decomposition routes. This inherent selectivity reduces the burden on downstream purification processes, directly contributing to cost reduction in electronic chemical manufacturing.
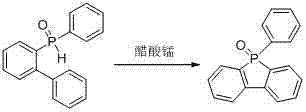
How to Synthesize 9-Phenyl-phosphorus fluorenes-9-oxide Efficiently
To implement this synthesis effectively, operators should begin by selecting the appropriate biphenylphosphine oxide substrate corresponding to the desired final substitution pattern. The patent outlines a generalized procedure where the substrate is combined with manganese acetate in a polar or semi-polar organic solvent, such as acetonitrile or ethanol, depending on solubility requirements. The reaction mixture is then heated to a moderate temperature, typically between 40°C and 80°C for optimal kinetics, and monitored via thin-layer chromatography (TLC) to determine the endpoint. Once the starting material is consumed, the reaction is quenched, and the product is isolated using standard column chromatography techniques, often utilizing an ethyl acetate and petroleum ether eluent system to achieve high-purity OLED material intermediates suitable for device fabrication.
- Charge a reactor with the specific biphenylphosphine oxide derivative substrate and manganese acetate promoter in a molar ratio ranging from 1: 0.5 to 1:2.5.
- Add a suitable organic solvent such as ethanol, acetonitrile, or DMF to the reaction mixture and heat to a temperature between 20°C and 120°C.
- Monitor reaction progress via TLC until completion, then isolate the target dibenzophosphole derivative through rapid column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this manganese-promoted methodology represents a strategic opportunity to de-risk the supply of critical optoelectronic intermediates. The elimination of exotic reagents and specialized reaction conditions translates directly into a more resilient supply chain, as the raw materials are commodity chemicals available from multiple global vendors. This diversification of the supply base reduces dependency on single-source suppliers for hazardous reagents, thereby enhancing supply chain reliability and ensuring continuity of production even during market fluctuations. Furthermore, the simplified operational protocol allows for manufacturing in standard multipurpose reactors rather than dedicated hazardous processing units, increasing facility utilization rates and flexibility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive, moisture-sensitive organolithium reagents with inexpensive manganese acetate salts. By removing the requirement for cryogenic cooling and inert atmosphere gloveboxes, manufacturers can achieve substantial cost savings in both energy consumption and capital equipment investment. Additionally, the high yields and simplified workup procedures reduce solvent usage and waste disposal costs, further optimizing the overall cost structure without compromising on the quality of the final dibenzophosphole derivatives.
- Enhanced Supply Chain Reliability: The use of stable, shelf-stable starting materials ensures that inventory can be maintained safely for extended periods without degradation, mitigating the risk of raw material spoilage. Since the reaction does not rely on reagents with short shelf-lives or strict storage requirements, logistics become simpler and less prone to disruption. This stability allows for better demand forecasting and inventory management, ensuring that production schedules can be met consistently to support the tight timelines of the display and lighting industries.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to industrial production is straightforward due to the absence of exothermic hazards associated with organometallic additions. The mild thermal profile allows for safe operation in large-scale reactors, facilitating the commercial scale-up of complex organophosphorus compounds with minimal engineering modifications. Moreover, the reduced generation of hazardous waste streams aligns with increasingly stringent environmental regulations, positioning manufacturers as responsible partners in the green chemistry initiative while avoiding potential compliance penalties.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese-promoted synthesis route. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: Does this synthesis method require strict anhydrous or oxygen-free conditions?
A: No, unlike conventional methods utilizing organolithium reagents, this manganese-promoted protocol operates effectively under ambient atmospheric conditions, significantly simplifying operational requirements and reducing equipment costs.
Q: What is the typical yield range for these dibenzophosphole derivatives?
A: The patent data indicates consistently high yields, typically ranging from 76% to 90% across various substituted substrates, demonstrating the robustness and efficiency of the catalytic system.
Q: Can this method accommodate diverse functional groups on the biphenyl backbone?
A: Yes, the method exhibits excellent substrate universality, successfully tolerating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as chloro, trifluoromethyl, and cyano substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzophosphole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance phosphorus heterocycles play in the next generation of organic electronic devices. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to mass manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of dibenzophosphole derivatives performs consistently in your final applications. Our expertise in process optimization allows us to adapt this manganese-promoted technology to meet your specific volume and quality requirements.
We invite you to collaborate with us to leverage these technological advancements for your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current manufacturing processes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized synthesis methods can enhance your competitive edge in the global market for advanced electronic materials.
