Advanced Catalytic Hydrogenation for High-Purity 1,8-Diamino-3,6-Naphthalene Disulfonic Acid Production
The global demand for high-purity dye intermediates and fine chemicals necessitates a shift towards greener, more efficient synthetic methodologies. Patent CN101993398B introduces a transformative approach for the preparation of 1,8-diamino-3,6-naphthalene disulfonic acid (DANS), a critical precursor for H-acid and reactive dyes. This technology replaces the traditional, pollution-heavy iron powder reduction with a sophisticated liquid-phase catalytic hydrogenation process. By utilizing noble metal catalysts such as Pt/C or Pd/C under controlled pressure and temperature, the method achieves yields greater than 90% and purity exceeding 98%. For R&D directors and procurement specialists, this patent represents a significant leap forward in sustainable manufacturing, offering a pathway to reduce environmental liability while enhancing product consistency. The ability to operate under relatively mild conditions (40-150°C) while maintaining high throughput makes this technology particularly attractive for large-scale industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,8-diamino-3,6-naphthalene disulfonic acid has relied heavily on the reduction of the corresponding dinitro compound using iron powder. This legacy technology presents severe operational and environmental challenges that modern supply chains can no longer tolerate. The process generates substantial quantities of iron sludge, a hazardous solid waste that requires complex and costly disposal procedures, thereby increasing the overall carbon footprint of the manufacturing operation. Furthermore, the acidic conditions required for iron reduction are highly corrosive to standard reactor vessels, leading to frequent equipment maintenance, shortened asset lifecycles, and potential contamination of the product stream with metal ions. The operational complexity is further compounded by the need for extensive post-reaction processing to separate the organic product from the inorganic sludge, which often results in product loss and variable batch-to-batch quality. These inefficiencies create bottlenecks in production capacity and drive up the cost of goods sold, making the traditional route increasingly uncompetitive in a market that demands both economic and ecological efficiency.
The Novel Approach
In stark contrast, the novel catalytic hydrogenation method described in the patent offers a streamlined, environmentally benign alternative that addresses the core deficiencies of the iron reduction route. By employing heterogeneous catalysts like Pt/C or Pd/C, the reaction proceeds cleanly in an aqueous medium without generating solid waste byproducts. The process is designed such that upon completion, the catalyst can be simply filtered off, leaving behind a clear aqueous solution of the diamino product that is suitable for direct use in subsequent hydrolysis steps to produce H-acid. This elimination of isolation and purification steps significantly reduces processing time and solvent consumption. Moreover, the absence of corrosive reagents preserves reactor integrity, ensuring long-term equipment reliability. The selectivity of the noble metal catalysts ensures that the reduction targets the nitro groups specifically, minimizing the formation of side products and delivering a product with liquid chromatography content consistently above 98%. This technological shift not only aligns with green chemistry principles but also optimizes the entire value chain for cost and speed.
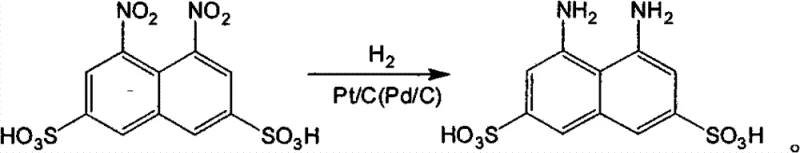
Mechanistic Insights into Pt/C and Pd/C Catalyzed Nitro Reduction
The core of this innovation lies in the precise manipulation of catalytic hydrogenation kinetics on a complex naphthalene scaffold. The substrate, 1,8-dinitro-3,6-naphthalene disulfonic acid, presents a unique challenge due to the presence of two electron-withdrawing nitro groups on the aromatic ring, which generally increases the difficulty of reduction compared to mononitro analogs. The patent elucidates that successful conversion requires a delicate balance of thermodynamic and kinetic parameters. The noble metal surfaces (Pt or Pd) facilitate the dissociative adsorption of hydrogen molecules, creating active atomic hydrogen species that attack the nitro groups. However, the patent notes that overly vigorous conditions can lead to over-reduction or degradation of the naphthalene ring system, generating impurities that compromise the quality of the final dye intermediate. Therefore, the selection of catalyst loading (0.1%-10% by weight) and the specific metal loading on the carbon support (0.5%-5% for Pt, 0.5%-10% for Pd) are critical variables. These parameters dictate the density of active sites available for the reaction, ensuring that the reduction proceeds rapidly enough to be commercially viable but selectively enough to maintain the structural integrity of the sulfonated naphthalene core.
Impurity control is intrinsically linked to the stability of the reaction environment, particularly temperature and pressure. The patent data indicates that temperatures exceeding the optimal range can trigger side reactions, while insufficient thermal energy fails to activate the hydrogenation fully, leaving residual nitro compounds. The mechanism relies on the solubility of hydrogen in the aqueous phase, which is governed by the applied pressure (0.8-3.5 MPa). Higher pressures increase the concentration of dissolved hydrogen at the catalyst surface, driving the reaction equilibrium towards the desired diamino product. Furthermore, the aqueous medium plays a dual role as both solvent and heat sink, preventing localized hot spots that could degrade the sensitive amino groups. The result is a reaction profile that maximizes the yield of the target molecule while suppressing the formation of azo or hydrazo byproducts, which are common pitfalls in nitro reductions. This mechanistic understanding allows for robust process control, ensuring that the reduction mother liquor meets the stringent purity specifications required for downstream pharmaceutical or dye applications without the need for recrystallization.
How to Synthesize 1,8-Diamino-3,6-Naphthalene Disulfonic Acid Efficiently
Implementing this synthesis route requires adherence to specific operational protocols to ensure safety and reproducibility. The process begins with the dissolution of the dinitro starting material in water, followed by the addition of the pre-activated catalyst. The reaction vessel must be rigorously purged to remove oxygen before introducing hydrogen, mitigating explosion risks inherent in high-pressure hydrogenation. Temperature ramping should be controlled to avoid exothermic runaways, and pressure must be maintained constant throughout the reaction duration to ensure consistent hydrogen availability. While the general principles are straightforward, the exact parameters depend on the specific catalyst batch and reactor geometry. For detailed standard operating procedures and specific parameter optimization, please refer to the technical guide below.
- Dissolve 1,8-dinitro-3,6-naphthalene disulfonic acid in water (5-30 times weight) and load into a high-pressure reactor.
- Add 0.1%-10% Pt/C or Pd/C catalyst relative to substrate weight, then purge the system with nitrogen and hydrogen.
- Heat to 40-150°C under 0.8-3.5 MPa hydrogen pressure for 0.5-4.5 hours, then filter to recover catalyst and obtain product solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic hydrogenation technology offers profound strategic benefits beyond simple chemical conversion. The most immediate impact is the drastic simplification of the waste management infrastructure. By eliminating the generation of iron sludge, facilities can avoid the substantial costs associated with hazardous waste treatment, storage, and transportation. This reduction in environmental liability translates directly into improved margins and reduced regulatory risk. Additionally, the ability to recycle the noble metal catalyst multiple times significantly lowers the effective cost of the catalyst per kilogram of product, offsetting the initial higher capital cost of precious metals compared to iron powder. The process also enhances supply chain resilience by reducing the dependency on bulk consumables like iron powder, which can be subject to market volatility and quality inconsistencies. Instead, the supply chain relies on durable, regenerable catalysts that provide consistent performance over extended periods.
- Cost Reduction in Manufacturing: The elimination of post-reaction separation steps and waste treatment processes leads to substantial operational expenditure savings. Since the product solution can be used directly in the next synthesis step, the need for drying, grinding, and re-dissolving is removed, saving significant energy and labor costs. Furthermore, the non-corrosive nature of the reaction medium extends the lifespan of reactors and piping, reducing capital expenditure on equipment replacement and maintenance downtime. These factors combine to lower the overall cost of production without compromising on quality.
- Enhanced Supply Chain Reliability: The robustness of the catalytic process ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification materials. The high yield and purity mean that less raw material is required to produce the same amount of finished good, optimizing inventory turnover. Moreover, the simplified workflow reduces the lead time from raw material intake to finished intermediate, allowing for more responsive fulfillment of customer orders. This agility is crucial in meeting the just-in-time delivery expectations of modern downstream manufacturers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively across a wide range of reactor sizes and water-to-substrate ratios. The absence of toxic sludge makes it easier to obtain and maintain environmental permits, facilitating expansion into regions with strict ecological regulations. The green nature of the process also aligns with the sustainability goals of major multinational corporations, making the supply chain more attractive to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology. They are derived from the specific experimental data and advantageous effects reported in the patent literature. Understanding these nuances is essential for evaluating the feasibility of adopting this route for commercial production.
Q: What are the advantages of catalytic hydrogenation over iron powder reduction for DANS?
A: Catalytic hydrogenation eliminates the generation of massive iron sludge waste, prevents equipment corrosion, and allows the product mother liquor to be used directly in subsequent steps without post-treatment.
Q: Can the noble metal catalysts be recycled in this process?
A: Yes, both Pt/C and Pd/C catalysts can be recovered by filtration and reused for more than five cycles while maintaining high activity and yield.
Q: What purity levels can be achieved with this hydrogenation method?
A: The process consistently achieves a liquid chromatography content of over 98% with a reaction yield exceeding 90%, ensuring high quality for downstream dye synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,8-Diamino-3,6-Naphthalene Disulfonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global fine chemicals market. Our technical team has extensively analyzed the catalytic hydrogenation pathways described in patent CN101993398B and possesses the expertise to scale this technology from laboratory benchtop to full industrial production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to commercial scale is seamless and risk-mitigated. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,8-diamino-3,6-naphthalene disulfonic acid meets the highest international standards.
We invite potential partners to engage with our technical procurement team to discuss how this green technology can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching from iron reduction to catalytic hydrogenation for your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and sustainability in your manufacturing operations.
