Advanced Synthesis of Halogenated Hydroxypropyltrimethylammonium Intermediates for L-Carnitine Production
Advanced Synthesis of Halogenated Hydroxypropyltrimethylammonium Intermediates for L-Carnitine Production
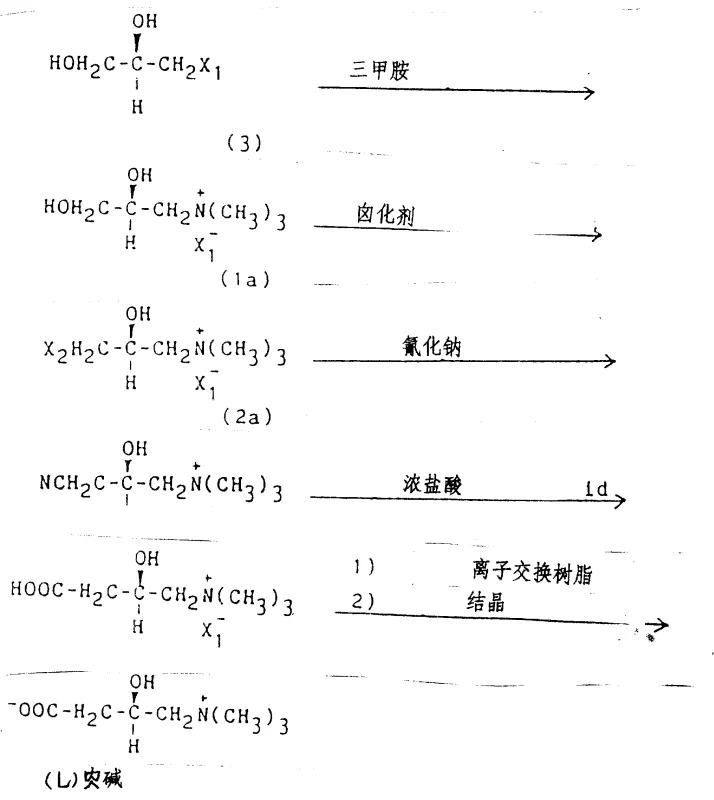
The pharmaceutical and nutritional supplement industries rely heavily on the efficient production of L-carnitine, a vital compound for fatty acid metabolism, yet traditional synthetic routes have long been plagued by high costs and complex stereochemical challenges. Patent CN87105804A introduces a transformative methodology for preparing halogenated-3-halogenated-2-hydroxypropyltrimethylammonium, a critical precursor that bypasses the limitations of conventional epihalohydrin-based chemistry. This innovation allows for the economical and high-yield production of the (S)-configuration compound, which is essential for synthesizing biologically active (L)-carnitine without the need for difficult resolution steps. By leveraging readily available starting materials and robust halogenation techniques, this process represents a significant leap forward in fine chemical manufacturing, offering a reliable pathway for scalable production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carnitine intermediates has depended heavily on the use of epihalohydrins, which present substantial logistical and economic hurdles for large-scale manufacturing operations. The procurement of optically pure (S)-epihalohydrin is notoriously difficult and expensive, often requiring complex resolution of racemic mixtures that drastically reduces overall process efficiency and yield. Furthermore, reacting epihalohydrins with trimethylamine halides can lead to side reactions and impurities that complicate downstream purification, increasing the burden on quality control laboratories and extending production lead times. These traditional pathways often suffer from poor atom economy and generate significant waste streams, making them less attractive in an era where environmental compliance and cost reduction are paramount concerns for supply chain directors.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes halogenated-2,3-dihydroxypropyltrimethylammonium or its chiral (S)-isomer as the starting point, effectively circumventing the need for unstable epihalohydrin precursors. This strategy employs a selective halogenation of the primary hydroxyl group, a reaction that proceeds with high specificity and minimal risk of racemization at the chiral center. By starting with (R)-3-halogeno-1,2-propanediol, which is more accessible and easier to handle, manufacturers can achieve superior optical purity in the final intermediate. This shift not only simplifies the reaction workflow but also enhances the robustness of the supply chain by relying on commodity chemicals rather than specialized, high-cost reagents, thereby driving down the total cost of ownership for the final API.
Mechanistic Insights into Selective Primary Hydroxyl Halogenation
The core of this technological breakthrough lies in the precise chemoselectivity of the halogenation step, where the primary hydroxyl group is targeted exclusively while leaving the secondary hydroxyl group and the quaternary ammonium moiety intact. The reaction mechanism typically involves the activation of the primary alcohol by a halogenating agent such as thionyl chloride or phosphorus halides, forming a reactive intermediate that undergoes nucleophilic substitution by the halide ion. This process is carefully controlled to prevent elimination reactions or attack on the secondary alcohol, which would compromise the stereochemical integrity of the molecule. The use of dipolar organic solvents like dimethylformamide further stabilizes the transition state, facilitating the rapid and clean conversion of the diol precursor into the desired di-halogenated product with yields often exceeding ninety percent.
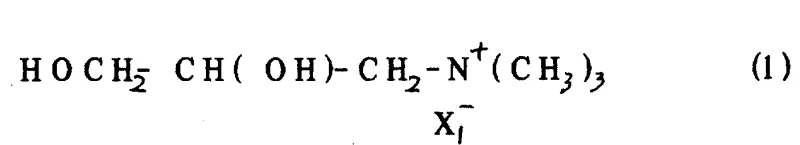
Impurity control is inherently built into this mechanistic pathway due to the distinct reactivity differences between primary and secondary alcohols under the specified conditions. The quaternary ammonium group, being positively charged, exerts an electron-withdrawing effect that can influence the acidity of neighboring protons, but the reaction conditions are optimized to avoid Hofmann elimination or other degradation pathways. Analytical monitoring via high-performance liquid chromatography confirms that residual starting material is minimal and that side products are easily removed through standard crystallization techniques. This high level of chemical purity is critical for downstream applications in pharmaceutical synthesis, where trace impurities can affect the safety profile and efficacy of the final therapeutic agent, thus validating the robustness of this catalytic-free transformation.
How to Synthesize Halogenated Hydroxypropyltrimethylammonium Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for converting readily available diols into high-value halogenated intermediates suitable for immediate use in carnitine production lines. The process begins with the quaternization of a chiral halohydrin followed by a straightforward halogenation step that can be performed in common industrial solvents. Operators can expect a streamlined workflow that minimizes unit operations and reduces the need for complex chromatographic separations, making it ideal for both pilot-scale validation and full commercial deployment. For a detailed breakdown of the specific reaction parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- React (R)-3-halogeno-1,2-propanediol with trimethylamine to form the quaternary ammonium diol intermediate.
- Treat the intermediate with a selective halogenating agent like thionyl chloride or hydrogen bromide.
- Isolate the final halogenated product through crystallization or solvent extraction methods.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers profound strategic benefits that extend far beyond simple laboratory curiosity, fundamentally altering the cost structure of carnitine intermediate manufacturing. By eliminating the dependency on scarce chiral epoxides, companies can secure a more stable and predictable supply of raw materials, insulating their production schedules from the volatility of the specialty chemical market. The simplified reaction sequence reduces the number of processing steps, which directly correlates to lower energy consumption, reduced labor hours, and decreased capital expenditure on reactor time, all of which contribute to a significantly leaner operational model. Furthermore, the high yields and purity achieved reduce the volume of waste generated, aligning with increasingly stringent environmental regulations and lowering the costs associated with waste disposal and treatment.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and the use of commodity-grade halogenating agents like thionyl chloride drive down the variable costs per kilogram of product substantially. This process avoids the low-yield resolution steps typical of older methods, meaning that more of the input mass is converted into saleable product, thereby improving the overall material efficiency and margin potential for the manufacturer. Additionally, the ability to use common solvents and standard equipment reduces the need for specialized infrastructure, allowing for flexible production across multiple facilities without significant retooling investments.
- Enhanced Supply Chain Reliability: Sourcing (R)-3-halogeno-1,2-propanediol is far more reliable than sourcing optically pure epihalohydrins, as the former can be produced via established microbial or chemical methods with high consistency. This stability in raw material availability ensures that production timelines are met consistently, reducing the risk of stockouts that can disrupt downstream API synthesis and delay time-to-market for finished pharmaceutical products. The robustness of the chemistry also means that the process is less sensitive to minor variations in feedstock quality, providing a buffer against supply chain fluctuations.
- Scalability and Environmental Compliance: The reaction conditions described, such as moderate temperatures and atmospheric pressure operations, are inherently safer and easier to scale from kilogram to multi-ton quantities without encountering the heat transfer or mixing limitations often seen in exothermic epoxide ring-openings. The process generates fewer hazardous byproducts compared to traditional routes, simplifying the effluent treatment process and reducing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major global pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, drawing directly from the experimental data and process descriptions provided in the intellectual property documentation. These insights are designed to clarify the operational feasibility and strategic value of adopting this technology for the production of L-carnitine precursors. Understanding these nuances is essential for technical teams evaluating the integration of this process into existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis method over traditional epihalohydrin routes?
A: The primary advantage is the avoidance of expensive and difficult-to-source chiral epihalohydrins. By starting with readily available (R)-3-halogeno-1,2-propanediol, the process significantly lowers raw material costs and simplifies the supply chain while maintaining high optical purity for L-carnitine synthesis.
Q: How is stereochemical integrity maintained during the halogenation step?
A: Stereochemical integrity is preserved because the halogenation reaction selectively targets the primary hydroxyl group at the 3-position. The chiral center at the 2-position, bearing the secondary hydroxyl group, remains untouched during the substitution, preventing racemization and ensuring the production of the biologically active (S)-configuration required for L-carnitine.
Q: What halogenating agents are suitable for this industrial process?
A: The patent identifies thionyl chloride and hydrogen bromide as the most suitable agents. Thionyl chloride is preferred for chlorination reactions, often used in dipolar solvents like dimethylformamide, while hydrogen bromide in acetic acid is effective for bromination, both offering high yields and ease of purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Hydroxypropyltrimethylammonium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development of life-saving therapies and nutritional supplements, which is why we have invested deeply in mastering complex synthetic pathways like the one described in CN87105804A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality and timely delivery. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of halogenated hydroxypropyltrimethylammonium meets the exacting standards required for pharmaceutical grade applications.
We invite you to collaborate with us to leverage this advanced technology for your next project, offering a partnership that combines technical expertise with commercial agility. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volumes and to obtain specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our premium intermediate solutions.
