Advanced Manufacturing of N-Benzyl Dioxothiazolidinyl Benzamide Intermediates for Diabetes Therapy
The pharmaceutical landscape for treating type 2 diabetes and hyperlipidemia has long been dominated by biguanides and sulfonylureas, yet these classes often suffer from significant side effects such as lactic acidosis and delayed hypoglycemia. Addressing these critical safety concerns, Chinese Patent CN1133627C introduces a novel class of N-benzyldioxothiazolidinylbenzamide derivatives that exhibit potent hypoglycemic and lipid-lowering activities with an improved safety profile. This patent details not only the final active compounds but, crucially for the supply chain, the robust manufacturing intermediates required to access this chemical space efficiently. As a divisional application of earlier research, this document refines the synthetic pathways, focusing on specific substitution patterns that differentiate these molecules from prior art thiazolidine-2,4-dione derivatives, which typically feature para-substituted benzene rings without additional substituents.
For R&D directors and procurement specialists, understanding the nuances of this patent is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials. The invention specifically targets the improvement of insulin resistance, a core mechanism in metabolic syndrome, by utilizing a unique structural scaffold where the thiazolidinedione ring is linked to an aromatic system via a meta-substituted benzene ring. This structural deviation is not merely academic; it represents a strategic pivot in medicinal chemistry that requires specialized synthetic expertise to execute at scale. The patent provides comprehensive data on the preparation of these intermediates, ensuring that manufacturers can replicate the high yields and purity levels demonstrated in the examples, thereby reducing the risk associated with developing new antidiabetic formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazolidinedione-based antidiabetic agents has relied heavily on structures where the connection between the heterocyclic ring and the aromatic moiety occurs at the para-position of a benzene ring. These conventional methods often result in compounds where the central benzene ring lacks substituents, or where the aromatic component is restricted to simple oxazole rings or sulfamoyl groups. Such structural limitations can constrain the pharmacokinetic properties and binding affinity of the resulting drug candidates, potentially leading to suboptimal therapeutic indices or unforeseen metabolic liabilities. Furthermore, traditional synthetic routes for these classes of compounds may involve harsh conditions or expensive reagents that complicate the purification process, leading to lower overall yields and higher production costs. The reliance on these older scaffolds limits the chemical diversity available to medicinal chemists seeking to optimize potency and selectivity against nuclear receptors like PPAR-gamma.
The Novel Approach
The approach detailed in CN1133627C breaks away from these constraints by introducing a versatile intermediate structure characterized by a meta-substituted benzene ring connecting the thiazolidinedione moiety to the rest of the molecule. This novel architecture allows for greater flexibility in modifying the R1, R2, and R3 substituents, enabling fine-tuning of the compound's lipophilicity and electronic properties. The patent outlines a convergent synthesis strategy where key intermediates, such as the 5-(2,4-dioxothiazolidin-5-ylidene)methyl benzoic acid derivatives, are prepared through efficient condensation and hydrolysis steps. This methodology not only expands the accessible chemical space but also streamlines the manufacturing process by utilizing readily available starting materials like substituted benzaldehydes and thiourea. By shifting the substitution pattern and optimizing the linkage chemistry, this novel approach offers a pathway to generate libraries of compounds with superior efficacy in lowering blood glucose and lipids compared to their predecessors.
Mechanistic Insights into Knoevenagel Condensation and Amide Coupling
The core of the synthetic strategy relies on a classic yet highly effective Knoevenagel condensation to construct the exocyclic double bond linking the thiazolidinedione ring to the benzyl system. In this mechanism, a substituted benzaldehyde derivative reacts with thiazolidine-2,4-dione in the presence of a weak base catalyst, such as ammonium acetate, and a dehydrating solvent like benzene or toluene. The reaction proceeds through the formation of an enamine intermediate, followed by elimination of water to yield the stable 5-arylidene-thiazolidine-2,4-dione structure. This step is critical as it establishes the rigid planar geometry required for receptor binding. The patent specifies that this reaction can be driven to completion under reflux conditions, often utilizing a Dean-Stark apparatus to continuously remove water, thereby pushing the equilibrium towards the product side and achieving high conversion rates without the need for exotic transition metal catalysts.
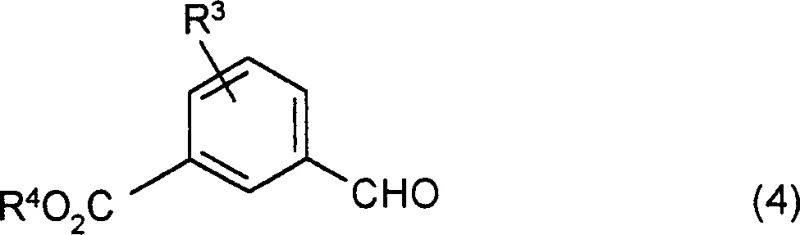
Following the formation of the core heterocyclic scaffold, the synthesis proceeds through a hydrolysis step to convert the ester functionality into a carboxylic acid, which serves as the handle for the final amide coupling. This transformation is typically achieved under acidic conditions using a mixture of acetic acid and concentrated hydrochloric acid under reflux. The resulting acid intermediate is then activated using coupling reagents such as diethyl cyanophosphate or EDC in polar aprotic solvents like DMF. This activation allows for the nucleophilic attack by various substituted benzylamines, forming the final amide bond that defines the N-benzyl dioxothiazolidinyl benzamide structure. The mechanistic precision of this coupling ensures that the stereochemical integrity of the molecule is maintained, and the mild reaction conditions prevent the degradation of the sensitive thiazolidinedione ring, which is prone to hydrolysis under strongly basic conditions.
How to Synthesize N-Benzyl Dioxothiazolidinyl Benzamide Efficiently
The synthesis of these high-value intermediates requires precise control over reaction parameters to ensure consistent quality and yield. The process begins with the condensation of the appropriate aldehyde and thiazolidinedione, followed by purification via recrystallization to remove unreacted starting materials. Subsequent hydrolysis must be carefully monitored to avoid over-degradation, and the final coupling step demands anhydrous conditions to maximize efficiency. For detailed operational parameters, stoichiometry, and workup procedures, refer to the standardized synthesis guide below which encapsulates the best practices derived from the patent examples.
- Perform Knoevenagel condensation between a substituted benzaldehyde and thiazolidine-2,4-dione using ammonium acetate and acetic acid in refluxing benzene.
- Hydrolyze the resulting ester intermediate under acidic conditions (acetic acid/concentrated hydrochloric acid) to obtain the free carboxylic acid.
- Couple the carboxylic acid intermediate with a substituted benzylamine using a condensing agent like diethyl cyanophosphate in DMF.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the synthetic route described in this patent offers significant advantages in terms of raw material availability and process simplicity. The starting materials, including various substituted benzaldehydes, thiourea, and common alkyl acrylates, are commodity chemicals available from multiple global suppliers, reducing the risk of supply chain bottlenecks. The avoidance of precious metal catalysts or cryogenic conditions further simplifies the manufacturing infrastructure requirements, allowing for production in standard multipurpose reactors. This accessibility translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the process eliminates the need for expensive specialized equipment or hazardous reagents that require complex disposal protocols. The robust nature of the chemistry ensures that batch-to-batch variability is minimized, a key factor for maintaining long-term supply contracts.
- Cost Reduction in Manufacturing: The synthetic pathway leverages inexpensive reagents like ammonium acetate and acetic acid, avoiding the high costs associated with palladium-catalyzed cross-couplings often seen in similar medicinal chemistry programs. By utilizing thermal reflux conditions rather than high-pressure hydrogenation for the initial scaffold construction, the process significantly lowers energy consumption and capital expenditure. Furthermore, the high yields reported in the patent examples, often exceeding 80 percent for key steps, reduce the amount of waste generated per kilogram of product, contributing to substantial cost savings in raw material utilization and waste treatment.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable intermediates ensures that production can be scheduled flexibly without the degradation risks associated with highly reactive species. The ability to isolate and store the carboxylic acid intermediate allows manufacturers to decouple the upstream synthesis from the final coupling step, creating a buffer stock that enhances supply continuity. This modularity means that disruptions in the supply of specific benzylamines do not necessarily halt the entire production line, as the core thiazolidinedione scaffold can be produced and stockpiled independently, providing a strategic advantage in managing inventory levels.
- Scalability and Environmental Compliance: The processes described utilize solvents such as ethanol, ethyl acetate, and benzene (or safer alternatives like toluene), which are well-understood in industrial hygiene and can be efficiently recovered and recycled. The absence of heavy metal residues simplifies the purification workflow, reducing the burden on downstream processing and ensuring compliance with strict regulatory limits for elemental impurities in drug substances. The straightforward crystallization steps described for purification facilitate easy scale-up from laboratory to pilot plant and eventually to commercial tonnage, ensuring that the technology is ready for immediate industrial deployment without extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, scalability, and application of these intermediates in drug development. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: What distinguishes this intermediate from conventional thiazolidinedione derivatives?
A: Unlike conventional derivatives where the substitution position is typically para, this patent describes intermediates with specific meta-substitution patterns on the central benzene ring, offering improved insulin resistance profiles.
Q: Is the synthesis scalable for commercial production?
A: Yes, the process utilizes standard industrial reactions such as reflux condensation and acid hydrolysis, avoiding exotic catalysts, which facilitates commercial scale-up from pilot to multi-ton production.
Q: What are the purity specifications for these intermediates?
A: The patent examples demonstrate high purity through recrystallization steps (e.g., from ethanol or ethyl acetate), yielding crystalline products suitable for stringent pharmaceutical quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Benzyl Dioxothiazolidinyl Benzamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation antidiabetic therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and purity of every batch, guaranteeing that our N-benzyl dioxothiazolidinyl benzamide intermediates meet the highest industry standards for safety and efficacy.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our manufacturing capabilities can support your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized processes can reduce your overall COGS. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to being your trusted partner in the complex landscape of pharmaceutical intermediate supply.
