Scalable Synthesis of 4-Substituted-1H-Indoles-3-Glyoxamides for Commercial API Production
Scalable Synthesis of 4-Substituted-1H-Indoles-3-Glyoxamides for Commercial API Production
The pharmaceutical industry's relentless pursuit of potent anti-inflammatory agents has placed significant focus on secretory phospholipase A2 (sPLA2) inhibitors. Patent CN1305459A discloses a groundbreaking process for preparing 4-substituted-1H-indoles-3-glyoxamides, which serve as critical intermediates for these therapeutic compounds. Unlike traditional synthetic routes that often rely on harsh demethylation conditions or expensive precious metal catalysts, this invention introduces a streamlined methodology utilizing sulfinating agents. This technical advancement not only enhances the purity profile of the resulting pharmaceutical intermediates but also significantly mitigates environmental hazards associated with legacy manufacturing processes. For procurement and supply chain leaders, understanding this shift is vital for securing reliable sources of high-quality API precursors.
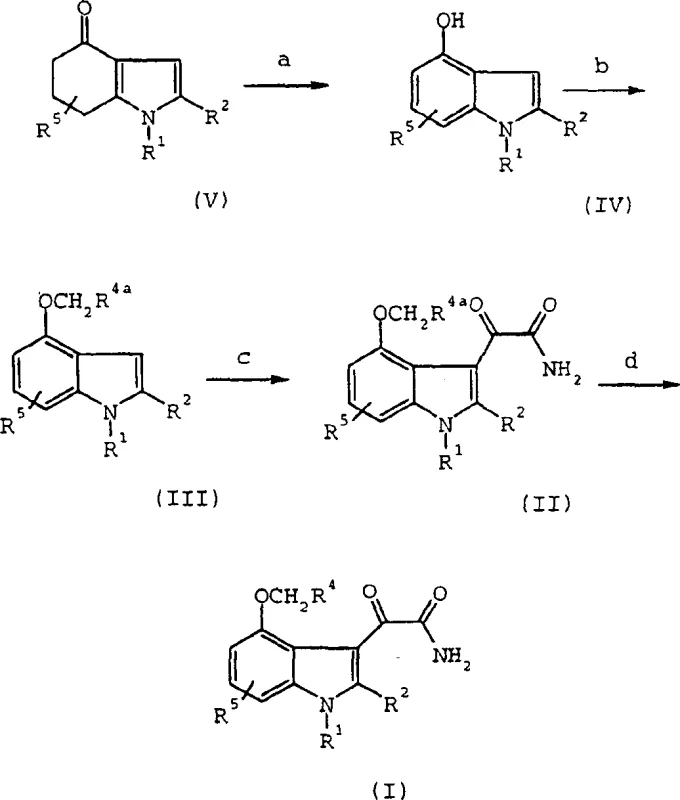
The core innovation lies in the efficient transformation of 4-oxo-4,5,6,7-tetrahydroindoles into 4-hydroxyindoles. Historically, this transformation was a bottleneck, requiring either ortho-demethylation of methoxy precursors using corrosive boron tribromide or high-temperature aerobic oxidation with palladium catalysts. The disclosed process circumvents these issues by employing sulfinating reagents, such as methyl p-tolylsulfinate, in the presence of strong bases like sodium hydride. This approach allows for the direct introduction of the hydroxyl functionality under relatively mild thermal conditions, typically ranging from 25°C to reflux, thereby preserving the integrity of sensitive substituents on the indole ring.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing 4-substituted-1H-indole-3-glyoxylic acid amide derivatives were fraught with operational and economic inefficiencies. A predominant strategy involved the preparation of 4-methoxyindoles followed by demethylation. This step frequently necessitated the use of boron tribromide (BBr3), a reagent that is not only expensive but also highly corrosive and difficult to handle on a large scale, generating significant amounts of boron-containing waste that requires complex disposal protocols. Furthermore, alternative oxidative routes relied on refluxing 4-oxo-indoles in high-boiling polar hydrocarbon solvents like carbitol in the presence of palladium on carbon. This heterogeneous catalysis presents severe challenges for commercial scale-up, including the risk of catalyst poisoning, the necessity for rigorous filtration to remove trace metals to meet ppm-level specifications, and the substantial capital cost associated with recovering and regenerating precious metal catalysts. These factors collectively inflate the cost of goods sold (COGS) and introduce supply chain vulnerabilities.
The Novel Approach
The novel approach detailed in the patent fundamentally reengineers the oxidation step to enhance both safety and cost-efficiency. By utilizing sulfinating agents, the process eliminates the dependency on precious metal catalysts entirely. The reaction proceeds via the treatment of the tetrahydroindole ketone with a base, preferably sodium hydride, followed by the addition of the sulfinating reagent. This chemistry is robust and tolerant of various substituents, allowing for greater structural diversity in the final API. Moreover, the process facilitates 'one-pot' operations for upstream steps, such as the halogenation and decarboxylation of propionoacetic acids, minimizing the isolation of volatile and lachrymatory intermediates. This consolidation of unit operations reduces solvent consumption and processing time, directly translating to lower manufacturing costs and a reduced environmental footprint, making it an ideal candidate for green chemistry initiatives in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Sulfinating Agent Oxidation
The mechanistic pathway for the conversion of the 4-oxo-tetrahydroindole to the aromatic 4-hydroxyindole is a sophisticated example of oxidative aromatization. The process initiates with the deprotonation of the alpha-carbon adjacent to the carbonyl group by a strong base, generating a reactive enolate species. Upon introduction of the sulfinating agent, such as methyl p-tolylsulfinate, a nucleophilic attack occurs, installing the sulfinyl moiety. Subsequent elimination reactions, driven by the thermodynamic stability of the aromatic indole system, result in the expulsion of the sulfur-containing leaving group and the formation of the phenolic hydroxyl group at the 4-position. This mechanism avoids the radical pathways often associated with metal-catalyzed oxidations, thereby reducing the formation of non-specific byproducts and polymeric tars that can complicate downstream purification.
Impurity control is inherently superior in this route due to the specificity of the sulfinating reagent. In traditional metal-catalyzed oxidations, over-oxidation or ring degradation can occur, leading to complex impurity profiles that are difficult to purge. In contrast, the sulfinating oxidation is highly selective for the desired aromatization. The use of stoichiometric amounts of reagents, typically a molar equivalent or slight excess relative to the pyrrole starting material, ensures complete conversion while minimizing side reactions. Furthermore, the reaction conditions allow for the use of standard crystallization or chromatography methods to isolate Intermediate IV with high purity. The ability to monitor the reaction progress via TLC or HPLC ensures that the endpoint is precisely determined, preventing the degradation of the sensitive indole core and ensuring a consistent quality profile essential for regulatory compliance in drug substance production.
How to Synthesize 4-Substituted-1H-Indoles-3-Glyoxamides Efficiently
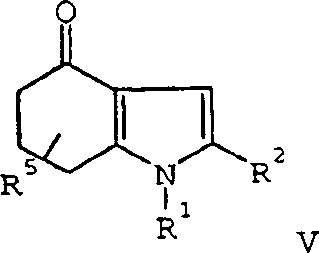
The synthesis of these valuable intermediates begins with the preparation of the key 4-oxo-4,5,6,7-tetrahydroindole scaffold. This involves the halogenation of a substituted propionoacetate using sulfuryl chloride at controlled temperatures (0°C to 25°C), followed by hydrolysis and decarboxylation to yield an acylated cyclohexanedione. This triketone is then condensed with a primary amine in a high-boiling solvent like toluene to effect cyclization and dehydration. The resulting ketone is then subjected to the novel oxidation protocol described above. Following the formation of the 4-hydroxyindole, the phenolic oxygen is alkylated using agents like methyl bromoacetate. The final glyoxylamide functionality is installed by reacting the 3-position with oxalyl chloride followed by ammonia treatment. Detailed standardized operating procedures for each of these critical unit operations are provided in the technical guide below.
- Halogenate substituted propionoacetate with sulfuryl chloride, followed by hydrolysis and decarboxylation to form the acylated cyclohexanedione.
- Cyclize the triketone with a primary amine in refluxing toluene to form the 4-oxo-4,5,6,7-tetrahydroindole intermediate.
- Oxidize the tetrahydroindole using a sulfinating agent (e.g., methyl p-tolylsulfinate) and base to generate the 4-hydroxyindole core.
- Alkylate the phenolic hydroxyl group, react with oxalyl chloride and ammonia to form the glyoxylamide, and optionally hydrolyze to the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio. By removing the need for palladium catalysts and boron tribromide, the process relies on commodity chemicals that are widely available from multiple global suppliers, thereby reducing single-source dependency risks. The elimination of precious metals also removes the logistical and financial burden of catalyst recovery and refining, which often requires specialized third-party vendors. Additionally, the 'one-pot' nature of the upstream synthesis minimizes the number of isolation steps, reducing solvent usage and waste disposal costs, which are significant drivers of overall manufacturing expenses in the fine chemical sector.
- Cost Reduction in Manufacturing: The economic impact of switching to this sulfinating agent-based process is profound. Traditional methods incur high costs associated with the purchase and recovery of palladium catalysts, as well as the disposal of hazardous boron waste. By replacing these with inexpensive sulfinating reagents and common bases like sodium hydride or potassium carbonate, the variable cost per kilogram of the intermediate is significantly lowered. Furthermore, the higher overall yields reported in the patent examples, such as the 95.2% molar yield in the cyclization step, mean that less raw material is required to produce the same amount of product, directly improving the gross margin for the final API. The avoidance of cryogenic conditions for the majority of the synthesis also reduces energy consumption related to cooling and heating cycles.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-specialized reagents. Sulfuryl chloride, methyl bromoacetate, and oxalyl chloride are bulk commodities with stable global supply chains, unlike specialized catalysts which can face availability bottlenecks. The process tolerance for various solvents, including toluene, THF, and acetone, provides flexibility in sourcing; if one solvent faces a shortage, alternatives can often be substituted without re-validating the entire process. This flexibility ensures continuous production capability even during market fluctuations. Moreover, the stability of the intermediates allows for longer storage times if necessary, providing a buffer against demand spikes without the risk of rapid degradation seen in more sensitive organometallic intermediates.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is straightforward due to the absence of heterogeneous catalysis and hazardous demethylation reagents. The reactions are homogeneous and exothermic profiles are manageable, facilitating safe scale-up in standard glass-lined or stainless steel reactors. From an environmental perspective, the process generates less hazardous waste, particularly avoiding heavy metal contamination in wastewater streams. This simplifies effluent treatment and ensures compliance with increasingly stringent environmental regulations regarding heavy metal discharge. The reduced E-factor (mass of waste per mass of product) aligns with corporate sustainability goals, making the supply chain more attractive to environmentally conscious stakeholders and regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is crucial for R&D teams evaluating technology transfer and for procurement teams negotiating supply agreements. The clarity on reaction conditions and impurity profiles helps in setting realistic expectations for project timelines and quality specifications.
Q: How does this process improve upon conventional 4-hydroxyindole synthesis?
A: Conventional methods often rely on expensive and hazardous reagents like boron tribromide for demethylation or require high-temperature oxidation with precious metal catalysts like palladium on carbon. This patented process utilizes readily available sulfinating agents under milder conditions, eliminating the need for expensive catalyst recovery and reducing hazardous waste generation.
Q: What are the typical yields for the key oxidation step?
A: According to the experimental data in the patent, the oxidation of the tetrahydroindole intermediate to the 4-hydroxyindole using sodium hydride and methyl p-tolylsulfinate proceeds efficiently. In specific examples, the overall yield from the ketone precursor through to the alkylated ester was reported around 53.7%, with the initial cyclization step achieving molar yields exceeding 95%.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for scalability. It employs 'one-pot' methodologies where possible to minimize isolation of volatile intermediates and uses common industrial solvents like toluene, THF, and acetone. The avoidance of cryogenic conditions for most steps and the use of robust reagents make it highly adaptable for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Substituted-1H-Indoles-3-Glyoxamides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving sPLA2 inhibitors. Our technical team has extensively analyzed the process disclosed in CN1305459A and possesses the expertise to execute this chemistry with precision. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of 4-substituted-1H-indoles-3-glyoxamides meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for these complex intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this advanced synthetic route can reduce your overall COGS. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to accelerate your drug development timeline while maintaining the highest standards of quality and cost-efficiency.
