Advanced Ester-Based Synthesis of Whiskey Lactone for Commercial Scale-Up and Cost Efficiency
The global demand for high-quality flavor and fragrance ingredients continues to drive innovation in organic synthesis, particularly for valued compounds like whiskey lactone, known for its distinct coconut and creamy notes. A pivotal advancement in this domain is detailed in patent CN110437181B, which discloses a highly efficient synthesis process utilizing n-valeraldehyde and crotonate esters as primary feedstocks. This technological breakthrough addresses long-standing inefficiencies in traditional manufacturing routes by eliminating the need for acidic cocatalysts and excessive raw material ratios. For industry stakeholders, this represents a significant shift towards more sustainable and economically viable production methods. The process leverages a Knoevenagel addition reaction followed by a catalytic hydrogenation step, achieving yields exceeding 85 percent under mild conditions. By optimizing the molar ratio of reactants to nearly 1:1 and employing low-pressure hydrogenation, this method offers a compelling value proposition for manufacturers seeking to enhance purity while reducing operational expenditures. As a reliable synthetic flavors & fragrances supplier, understanding these mechanistic nuances is critical for evaluating the scalability and commercial potential of next-generation spice intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of whiskey lactone has relied on the condensation of crotonic acid with n-valeraldehyde, a pathway fraught with significant chemical and economic drawbacks. As illustrated in the traditional reaction scheme below, this legacy process necessitates the use of an initiator and an acidic cocatalyst to drive the reaction forward. However, n-valeraldehyde is chemically unstable in acidic media, leading to rapid polymerization and side reactions that severely depress the conversion rate of this valuable aldehyde. To compensate for these losses, prior art methods, such as those disclosed in patent CN200610047696.9, were forced to employ a massive excess of n-valeraldehyde, often at molar ratios as high as 4:1 relative to the acid. This gross inefficiency not only inflates raw material costs but also creates a complex downstream purification burden, as recovering and recycling the unreacted aldehyde from the reaction mixture is technically difficult and energy-intensive. Furthermore, the acidic environment necessitates rigorous washing and neutralization steps, generating substantial volumes of saline wastewater that complicate environmental compliance and increase waste treatment costs.
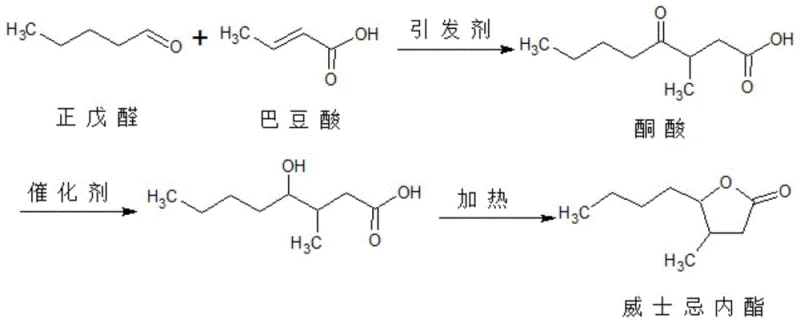
The Novel Approach
In stark contrast, the novel methodology presented in CN110437181B fundamentally reengineers the synthetic pathway by substituting crotonic acid with a crotonate ester, such as ethyl crotonate or methyl crotonate. This strategic substitution allows the Knoevenagel addition to proceed smoothly in an alcoholic solvent without the requirement for any acidic initiator or cocatalyst. By removing the acidic medium, the instability of n-valeraldehyde is effectively mitigated, enabling the reaction to proceed with a near-stoichiometric molar ratio of 0.8-1.2:0.8-1.2. This dramatic reduction in raw material excess translates directly into lower input costs and simplified recovery operations. The subsequent transformation involves a catalytic hydrogenation step that simultaneously reduces the ketone and cyclizes the molecule to form the lactone ring. This streamlined approach not only improves the overall atom economy but also simplifies the workup procedure, as the reaction mixture primarily contains the desired keto ester intermediate and recyclable alcohol solvent, avoiding the complex salt byproducts associated with acid-base neutralization in the conventional route.
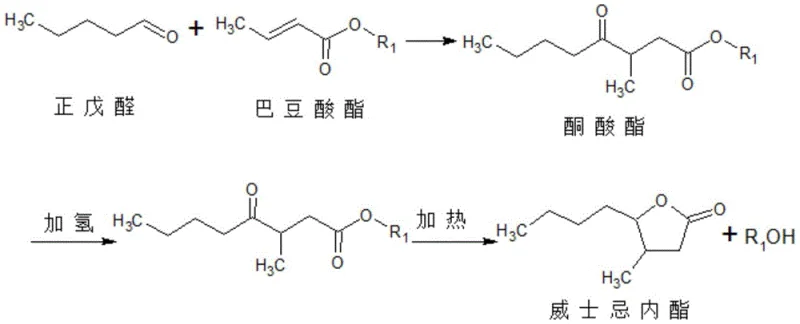
Mechanistic Insights into Ester-Based Knoevenagel Condensation and Hydrogenation
The core chemical innovation lies in the ability to perform the initial carbon-carbon bond formation via a Knoevenagel-type addition using an ester acceptor rather than a free acid. In the absence of an acidic cocatalyst, the reaction mechanism avoids the protonation of the aldehyde carbonyl which typically triggers decomposition pathways for n-valeraldehyde. Instead, the reaction proceeds through a concerted addition mechanism facilitated by the thermal energy provided at temperatures between 60-150°C. The use of an alcohol solvent serves a dual purpose: it acts as a homogeneous medium for the reactants and participates in the transesterification equilibrium if necessary, ensuring high solubility of the polar intermediates. This neutral environment preserves the integrity of the aldehyde functionality, allowing for high selectivity towards the desired gamma-keto ester intermediate. The absence of strong acids also prevents the premature cyclization or dehydration of the intermediate, ensuring that the molecular architecture remains intact for the subsequent hydrogenation step. This control over the reaction environment is paramount for maintaining a clean impurity profile, which is a critical quality attribute for food-grade flavor additives.
Following the formation of the keto ester, the process employs a heterogeneous catalytic hydrogenation using palladium carbon (Pd/C) or ruthenium carbon (Ru/C). This step is remarkably efficient, operating at normal temperature and low hydrogen pressures ranging from 0.1 to 1 MPa. The catalyst facilitates the reduction of the ketone group to a secondary alcohol, which spontaneously undergoes intramolecular esterification (lactonization) with the pendant ester group to close the five-membered ring. The mild conditions of this hydrogenation step are particularly advantageous for industrial safety and energy management, as they eliminate the need for high-pressure autoclaves and elevated temperatures that characterize many other hydrogenation processes. The choice of Pd/C or Ru/C provides robust activity and selectivity, minimizing the formation of over-reduced byproducts or ring-opened species. Furthermore, the heterogeneous nature of the catalyst allows for easy removal via filtration, ensuring that the final product is free from heavy metal contamination, a key requirement for regulatory compliance in the flavor and fragrance industry.
How to Synthesize Whiskey Lactone Efficiently
The practical implementation of this synthesis route involves a straightforward two-stage operation that is highly amenable to standard chemical processing equipment. The first stage requires the controlled addition of an alcohol solution of n-valeraldehyde into a heated mixture of the crotonate ester and solvent, maintaining reflux conditions to drive the addition reaction to completion. Once the keto ester intermediate is formed, the crude mixture is transferred directly to a hydrogenation vessel where the catalyst is introduced. This seamless transition between steps minimizes material handling and exposure to the environment, further enhancing the safety and efficiency of the overall manufacturing campaign.
- Perform a Knoevenagel addition reaction between n-valeraldehyde and a crotonate ester (such as ethyl crotonate) in an alcohol solvent at 60-150°C to form a keto ester intermediate.
- Transfer the crude keto ester solution to a hydrogenation reactor and add a palladium carbon or ruthenium carbon catalyst.
- Conduct hydrogenation at normal temperature and low pressure (0.1-1MPa) until hydrogen absorption ceases, then filter and recover solvent to isolate whiskey lactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ester-based synthesis route offers profound strategic advantages that extend beyond simple yield improvements. The shift from a 4:1 raw material ratio to a near 1:1 stoichiometry represents a massive reduction in the consumption of n-valeraldehyde, a volatile and cost-sensitive commodity chemical. By eliminating the need for excessive feedstock, manufacturers can significantly lower their direct material costs and reduce the capital tied up in inventory. Additionally, the removal of acidic cocatalysts simplifies the supply chain by reducing the number of hazardous chemicals that need to be sourced, stored, and handled on-site. The ability to operate the hydrogenation step at low pressure and normal temperature further reduces the barrier to entry for contract manufacturing organizations, as it does not require specialized high-pressure infrastructure, thereby expanding the pool of potential qualified suppliers and enhancing supply chain resilience.
- Cost Reduction in Manufacturing: The elimination of acidic cocatalysts and initiators removes the necessity for expensive neutralization agents and the associated waste disposal fees. Furthermore, the high conversion rate achieved without excessive aldehyde means that raw material utilization is maximized, directly lowering the cost of goods sold. The use of low-boiling point alcohols as solvents allows for efficient recovery and recycling through simple distillation, creating a closed-loop solvent system that minimizes purchase requirements and waste generation. These cumulative factors contribute to a substantially leaner cost structure compared to traditional acid-catalyzed processes.
- Enhanced Supply Chain Reliability: By relying on stable ester feedstocks rather than corrosive acids, the process reduces equipment maintenance needs and downtime associated with corrosion management. The mild reaction conditions ensure consistent batch-to-batch quality, reducing the risk of production failures or off-spec material that could disrupt downstream formulation schedules. The simplified purification process, which avoids complex aqueous workups, shortens the overall cycle time per batch, allowing for faster turnaround and more responsive fulfillment of customer orders. This operational stability is crucial for maintaining continuous supply to major flavor houses and food manufacturers.
- Scalability and Environmental Compliance: The process generates significantly less saline wastewater due to the absence of acid-base neutralization steps, aligning with increasingly stringent environmental regulations regarding industrial effluent. The low-pressure hydrogenation step presents a much lower safety risk profile, facilitating easier permitting and scale-up to multi-ton production capacities. The high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, ensuring that the final product meets rigorous purity specifications without the need for resource-intensive chromatographic purification. This combination of safety, environmental friendliness, and scalability makes the technology ideal for long-term commercial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the ester-based route superior to the traditional acid-based method for whiskey lactone?
A: The traditional method uses crotonic acid and requires an acidic cocatalyst, which destabilizes n-valeraldehyde leading to low conversion and difficult recovery. The novel ester-based route operates without acidic initiators, preventing raw material degradation and allowing for a near-stoichiometric molar ratio.
Q: What are the specific reaction conditions for the hydrogenation step?
A: The hydrogenation and cyclization occur simultaneously under mild conditions: normal temperature and low hydrogen pressure ranging from 0.1 to 1 MPa, using Pd/C or Ru/C catalysts, which significantly reduces energy consumption compared to high-pressure processes.
Q: How does this process impact waste treatment and environmental compliance?
A: By utilizing low-boiling point alcohols as solvents that can be easily recycled and eliminating the need for acidic washing steps required in the prior art, the process drastically reduces wastewater toxicity and treatment difficulty.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Whiskey Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthesis technologies to meet the evolving demands of the global flavor and fragrance market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ester-based whiskey lactone route can be seamlessly translated into robust industrial processes. We are committed to delivering high-purity whiskey lactone that adheres to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex catalytic hydrogenations and sensitive condensation reactions allows us to offer a product with a superior impurity profile, essential for high-end applications in food and beverage formulations.
We invite industry partners to collaborate with us to leverage these process efficiencies for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us to obtain specific COA data and route feasibility assessments, demonstrating our transparency and commitment to being a trusted partner in your success. Let us help you optimize your sourcing strategy with a reliable supply of premium quality whiskey lactone produced through sustainable and economically superior methods.
