Advanced Synthesis of Naphthoperylene Fluorescent Compounds for Commercial OLED Applications
Introduction to High-Performance Fluorescent Conjugated Compounds
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that combine high efficiency with structural stability and cost-effectiveness. Patent CN101792376B introduces a groundbreaking class of fluorescent conjugated compounds based on a naphthoperylene skeleton, offering a compelling solution for next-generation display technologies. This proprietary technology leverages a unique seven-aromatic-ring conjugated system that significantly enhances electron delocalization, resulting in superior optical properties such as long excitation and emission wavelengths. Specifically, these compounds demonstrate an excitation wavelength of 450nm and an emission wavelength of 478nm in chloroform or cyclohexane, accompanied by a remarkable quantum yield of up to 0.78. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this patent represents a critical advancement in achieving bright green fluorescence with simplified synthetic accessibility.
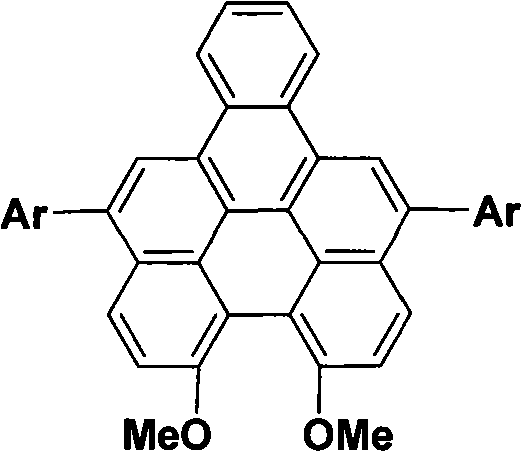
Beyond their optical performance, the commercial viability of these materials is underpinned by a robust and scalable synthesis method detailed in the patent. The process utilizes readily available starting materials like 7-methoxy-1-tetralone and employs standard organic transformations such as aluminum amalgam reduction and oxidative cyclization. This approach not only ensures high product yields but also facilitates easy derivatization, allowing manufacturers to tailor the electronic properties of the final material by modifying the aromatic substituents. As the global demand for high-purity electronic chemicals continues to surge, understanding the nuances of this synthesis pathway is essential for supply chain heads aiming to secure a continuous and cost-efficient source of advanced optoelectronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for polycyclic aromatic hydrocarbons (PAHs) and perylene derivatives often suffer from significant drawbacks that hinder their widespread commercial adoption. Conventional methods frequently rely on harsh reaction conditions, expensive transition metal catalysts, or multi-step sequences that result in cumulative yield losses. For instance, many existing protocols for constructing large fused ring systems require high-temperature pyrolysis or utilize toxic reagents that complicate waste management and increase environmental compliance costs. Furthermore, the lack of modularity in older synthetic strategies makes it difficult to introduce specific functional groups needed to tune the bandgap or solubility of the final OLED material. These inefficiencies lead to prolonged lead times for high-purity electronic chemicals and inflate the overall cost of goods sold, creating a bottleneck for manufacturers striving to meet the rigorous quality standards of the display industry.
The Novel Approach
In stark contrast, the methodology disclosed in CN101792376B offers a streamlined and highly efficient pathway to complex naphthoperylene structures. By utilizing an aluminum amalgam reduction followed by a strategic diazonium coupling, the process constructs the core helicene skeleton with impressive precision and yield. The subsequent oxidative cyclodehydrogenation steps, mediated by DDQ (2,3-dichloro-5,6-dicyano-1,4-benzoquinone) and Lewis acids, effectively aromatize the system under relatively mild conditions. This novel approach eliminates the need for extreme temperatures or exotic catalysts in the final ring-closing steps, thereby reducing energy consumption and operational risks. For procurement managers focused on cost reduction in electronic chemical manufacturing, this translates to a more predictable production schedule and lower raw material overheads, ensuring a competitive edge in the marketplace.
Mechanistic Insights into Oxidative Cyclodehydrogenation and Suzuki Coupling
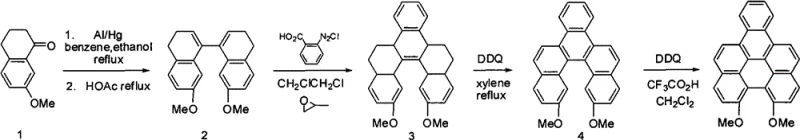
The core of this technology lies in the sophisticated manipulation of aromatic systems to achieve maximum conjugation. The synthesis begins with the reductive coupling of 7-methoxy-1-tetralone using aluminum amalgam, which generates a vicinal diol intermediate that spontaneously cyclizes to form the 7,7'-dimethoxy-3,4,3',4'-tetrahydro-1,1'-binaphthyl scaffold. This binaphthyl precursor is then subjected to a diazonium coupling reaction with phthalic acid diazonium chloride, a critical step that builds the steric complexity required for the helicene structure. The resulting tetrahydro-benzo[5]helicene is subsequently oxidized by DDQ in refluxing xylene to remove hydrogen atoms and establish partial aromaticity. The final transformation involves a Lewis acid-catalyzed oxidative cyclization, where the remaining saturated rings are fully aromatized to yield the planar, highly conjugated naphthoperylene core. This sequence ensures the formation of a rigid, planar structure essential for efficient charge transport and fluorescence.
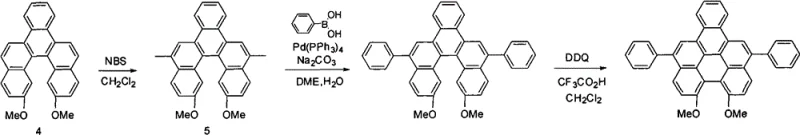
For derivatives requiring specific electronic tuning, the patent describes a modular functionalization strategy involving palladium-catalyzed cross-coupling. After the initial formation of the dimethoxy-benzo[5]helicene, selective bromination at the 5,12-positions using N-bromosuccinimide (NBS) introduces reactive handles for further modification. These bromo-intermediates serve as excellent substrates for Suzuki-Miyaura coupling with various aromatic boronic acids, allowing for the precise installation of electron-donating or electron-withdrawing groups. This push-pull electronic effect, created by the methoxy groups on the core and the variable aryl substituents, lowers the energy gap between the ground and excited states, facilitating easier electron excitation and enhancing fluorescence emission. Such mechanistic control over the molecular architecture is vital for R&D teams aiming to optimize the color purity and efficiency of green emitting layers in OLED devices.
How to Synthesize Naphthoperylene Fluorescent Compounds Efficiently
The synthesis of these high-value fluorescent compounds follows a logical progression from simple ketones to complex fused ring systems, balancing chemical efficiency with operational simplicity. The process is divided into distinct stages: precursor preparation, skeleton construction, functionalization (optional), and final aromatization. Each stage has been optimized in the patent examples to maximize yield and minimize impurity formation, making it an ideal candidate for technology transfer to pilot and commercial scales. The detailed standardized synthesis steps below outline the specific reagents, stoichiometry, and conditions required to replicate the high yields reported in the patent documentation, ensuring consistency for quality control laboratories.
- Perform aluminum amalgam reduction of 7-methoxy-1-tetralone followed by acid-catalyzed cyclization to form the binaphthyl precursor.
- Execute diazonium coupling with phthalic acid diazonium chloride to construct the benzo[5]helicene skeleton.
- Conduct final oxidative cyclodehydrogenation using DDQ and Lewis acid to aromatize the system into the target naphthoperylene compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits that extend beyond mere technical feasibility. The reliance on commodity chemicals such as 7-methoxy-1-tetralone, aluminum foil, and mercuric chloride ensures that raw material sourcing is stable and不受 geopolitical fluctuations that often affect specialized reagents. Moreover, the high yields observed in critical steps, such as the 97% yield in the DDQ oxidation and 100% yield in the final cyclization, significantly reduce the volume of waste generated per kilogram of product. This efficiency directly correlates to lower disposal costs and a reduced environmental footprint, aligning with the increasingly stringent sustainability mandates faced by modern chemical manufacturers. For supply chain heads, this means a more resilient production pipeline capable of meeting fluctuating market demands without compromising on delivery schedules.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts in the final ring-closing steps, relying instead on DDQ and Lewis acids, drastically lowers the direct material costs associated with production. Additionally, the high atom economy of the cyclization reactions minimizes the loss of valuable carbon skeletons, ensuring that the majority of the input mass is converted into the desired high-purity OLED material. This economic efficiency allows for competitive pricing strategies while maintaining healthy profit margins, a crucial factor for procurement managers evaluating long-term supplier contracts.
- Enhanced Supply Chain Reliability: The robustness of the synthetic protocol, which tolerates standard laboratory glassware and common solvents like dichloromethane and xylene, simplifies the equipment requirements for manufacturing. This reduces the capital expenditure needed for specialized reactors and allows for faster scale-up from gram to ton quantities. Consequently, suppliers can offer shorter lead times for high-purity electronic chemicals, providing downstream OLED panel manufacturers with the agility needed to respond to rapid changes in consumer electronics trends.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic or volatile reagents that would necessitate complex abatement systems, thereby streamlining regulatory compliance. The ability to perform reactions at moderate temperatures (e.g., reflux in xylene or room temperature for coupling) further enhances safety profiles and reduces energy consumption. These factors collectively support the commercial scale-up of complex polymer additives and small molecule emitters, ensuring that production can be expanded seamlessly to meet global demand without encountering significant environmental bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these naphthoperylene derivatives. The answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the suitability of this technology for their specific product lines and quality requirements.
Q: What is the quantum yield of the synthesized naphthoperylene compounds?
A: According to patent CN101792376B, the compounds exhibit high quantum yields, reaching up to 0.78 in solution, with excitation at 450nm and emission at 478nm.
Q: Can the aromatic substituents on the naphthoperylene core be customized?
A: Yes, the synthetic route allows for extensive derivatization via Suzuki coupling at the 5,12-positions, enabling the introduction of various aryl or heteroaryl groups.
Q: Is this synthesis suitable for large-scale commercial production?
A: The process utilizes cheap raw materials and achieves high yields in key steps (up to 100% in final cyclization), indicating strong potential for scalable industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthoperylene Fluorescent Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the naphthoperylene scaffold described in CN101792376B for the future of display technology. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready product is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed analysis of impurity profiles and optical properties such as quantum yield and emission wavelength. We are committed to delivering high-purity OLED material that meets the exacting standards of the global electronics industry.
We invite you to collaborate with our technical procurement team to explore how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this efficient manufacturing route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that your next generation of green fluorescent emitters is built on a foundation of chemical excellence and supply chain reliability.
