Advanced Synthesis of Asymmetric Spirobifluorenes for High-Performance OLED Applications
Advanced Synthesis of Asymmetric Spirobifluorenes for High-Performance OLED Applications
The rapid evolution of organic light-emitting diode (OLED) technology demands materials that offer superior thermal stability, solubility, and charge transport properties. Patent CN102633610A introduces a groundbreaking class of 'vertically' unsymmetrical spirobifluorene compounds derived from the strategic conversion of methyl groups on the fluorene loop. Unlike traditional symmetric analogues, these asymmetric structures allow for precise tuning of electronic properties by attaching different functional groups to the upper and lower fluorene units. This innovation addresses a critical bottleneck in the industry where synthesizing such asymmetry was previously prohibitively complex and costly. By leveraging a unique synthetic pathway starting from inexpensive 4,4'-dimethylbiphenyl, this technology enables the production of high-purity OLED material intermediates with exceptional spiro-conjugation effects.
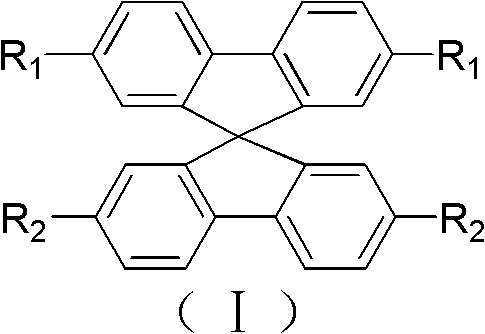
The core structural advantage lies in the three-dimensional orthogonal arrangement provided by the spiro-carbon center, which effectively inhibits molecular aggregation and enhances amorphous film formation. This patent specifically targets the synthesis of compounds where R1 and R2 are distinct, allowing for the independent optimization of hole and electron transport characteristics within a single molecule. Such molecular engineering is essential for developing next-generation display and optoelectronic materials that require balanced charge injection. The versatility of this chemical platform extends beyond OLEDs, finding applications in organic nonlinear optical materials and fluorescent probes, thereby opening diverse revenue streams for manufacturers adopting this proprietary synthesis route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spirobifluorene derivatives has relied heavily on the Grignard reaction of 2-bromobiphenyl with fluorenone. However, 2-bromobiphenyl is typically prepared via coupling reactions involving o-dibromobenzene, a raw material that is not only expensive but also subject to significant supply chain volatility. Furthermore, achieving vertical asymmetry using conventional routes often necessitates intricate protection-deprotection sequences to differentiate the reactivity of the two fluorene rings. These additional synthetic steps drastically reduce overall process efficiency, increase solvent consumption, and complicate purification protocols, leading to higher production costs and longer lead times. The inability to easily functionalize the 2,7-positions with different groups has limited the exploration of asymmetric spirobifluorenes in commercial applications despite their theoretical advantages.
The Novel Approach
The methodology disclosed in CN102633610A circumvents these challenges by utilizing 4,4'-dimethylbiphenyl as a cost-effective starting scaffold. This approach leverages the inherent reactivity of the methyl groups, which can be selectively transformed into bromomethyl, aldehyde, carboxyl, and other functional moieties post-cyclization. This 'post-functionalization' strategy allows for the construction of the spiro core first, followed by the divergent modification of the upper and lower rings to achieve the desired asymmetry. By avoiding the need for expensive pre-functionalized halogenated biphenyls, this route significantly lowers the raw material cost basis. The simplicity of the reaction conditions, primarily involving standard Grignard chemistry and radical substitutions, ensures that the process is robust and amenable to large-scale industrial production without requiring specialized high-pressure or cryogenic equipment.
Mechanistic Insights into Grignard-Mediated Spiro Cyclization and Methyl Transformation
The heart of this synthesis lies in the efficient construction of the spirobifluorene core followed by the precise manipulation of the methyl substituents. The process initiates with the formation of a Grignard reagent from 4,4'-dimethyl-2-bromobiphenyl, which then undergoes nucleophilic addition to a fluorenone derivative. This step is critical as it establishes the quaternary spiro-carbon center. Following the addition, an acid-catalyzed dehydration cyclization occurs, typically using acetic acid and hydrochloric acid, to close the five-membered ring and finalize the spiro structure. The use of methyl groups at the 4,4' positions of the biphenyl precursor is strategic; they remain inert during the Grignard formation and cyclization, preserving their potential for later modification. This orthogonality is key to the success of the asymmetric synthesis, ensuring that the core formation does not interfere with the future functionalization sites.
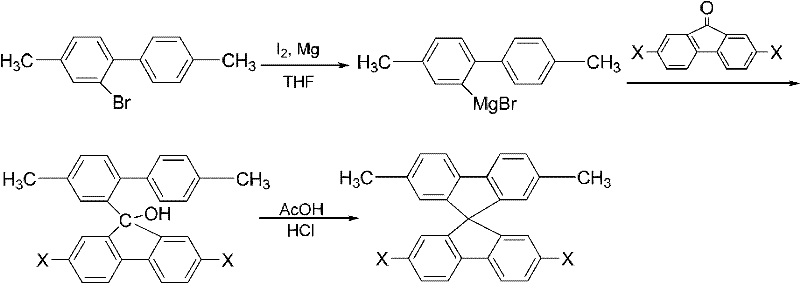
Once the spiro core containing methyl groups is established, the pathway diverges to introduce asymmetry through a series of functional group transformations. The methyl groups are first activated via radical bromination using N-bromosuccinimide (NBS) and a radical initiator like benzoyl peroxide. This converts the stable methyl groups into reactive bromomethyl intermediates. Subsequently, these bromomethyl groups are oxidized to aldehyde functionalities using sodium bicarbonate in dimethyl sulfoxide (DMSO). The resulting dialdehyde intermediate serves as a versatile hub for further derivatization. For instance, reaction with hydroxylamine hydrochloride yields dicyano derivatives, while condensation with active methylene compounds or amines can produce alkenyl or heterocyclic substituents. This modular approach allows chemists to attach electron-donating or electron-withdrawing groups independently to the top and bottom rings, fine-tuning the HOMO-LUMO energy levels for specific device applications.
How to Synthesize Asymmetric Spirobifluorene Efficiently
The synthesis of these high-value electronic chemicals follows a logical progression from commodity chemicals to specialized intermediates. The process begins with the bromination of 4,4'-dimethylbiphenyl, followed by Grignard reagent preparation and coupling with fluorenone. The subsequent steps involve the activation of the methyl groups and their conversion into the desired functional handles. This standardized workflow minimizes the need for custom catalysts and allows for the use of common industrial solvents like THF, dichloromethane, and DMSO. The detailed标准化 synthesis steps see the guide below ensure reproducibility and high purity, which are paramount for electronic grade materials.
- Prepare 4,4'-dimethyl-2-bromobiphenyl by brominating 4,4'-dimethylbiphenyl using liquid bromine and iodine catalyst in dichloromethane at low temperature.
- Form the spiro core by reacting the brominated biphenyl Grignard reagent with 2,7-disubstituted fluorenone in THF, followed by acid-catalyzed dehydration cyclization.
- Functionalize the methyl groups on the spiro core via radical bromination with NBS, oxidation to aldehydes, and subsequent conversion to target groups like cyano or heterocycles.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical performance. The primary advantage is the drastic reduction in raw material costs achieved by substituting expensive halogenated biphenyls with commodity-grade dimethylbiphenyl. This shift decouples production costs from the volatile pricing of specialized halogenated aromatics, providing greater budget predictability. Additionally, the synthetic route relies on well-established unit operations such as reflux, filtration, and distillation, which reduces the capital expenditure required for plant retrofitting. The robustness of the chemistry ensures consistent batch-to-batch quality, minimizing the risk of supply disruptions caused by failed runs or complex purification bottlenecks.
- Cost Reduction in Manufacturing: The elimination of expensive 2-bromobiphenyl precursors results in substantial cost savings throughout the production lifecycle. By utilizing 4,4'-dimethylbiphenyl, which is derived from widely available toluene feedstocks, the material cost basis is significantly lowered. Furthermore, the high atom economy of the methyl transformation steps reduces waste generation and solvent usage, contributing to lower operational expenditures. The avoidance of complex protection-deprotection sequences also shortens the overall cycle time, increasing throughput capacity without additional infrastructure investment.
- Enhanced Supply Chain Reliability: Sourcing 4,4'-dimethylbiphenyl is far more reliable than procuring specialized halogenated intermediates, as it is produced by multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and ensures continuity of supply even during market fluctuations. The simplified synthetic pathway also reduces the number of critical intermediates that need to be stocked, streamlining inventory management and reducing working capital requirements. The scalability of the process ensures that demand surges can be met rapidly without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The process utilizes standard reagents and solvents that are easily managed within existing environmental, health, and safety (EHS) frameworks. The absence of heavy metal catalysts or exotic reagents simplifies waste treatment and disposal, aligning with increasingly stringent global environmental regulations. The high yields observed in key steps, such as the initial bromination and final functionalization, minimize the volume of chemical waste generated per kilogram of product. This green chemistry profile not only reduces disposal costs but also enhances the sustainability credentials of the final electronic materials, a growing priority for downstream OEMs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these asymmetric spirobifluorene compounds. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of this technology.
Q: Why is the asymmetric spirobifluorene synthesis difficult conventionally?
A: Conventional methods typically rely on 2-bromobiphenyl, which is expensive and derived from costly o-dibromobenzene coupling. Furthermore, achieving vertical asymmetry (different substituents on the top and bottom fluorene units) requires complex multi-step protection and deprotection strategies that lower overall yield and increase purification burdens.
Q: How does the methyl transformation strategy improve cost efficiency?
A: This novel route utilizes 4,4'-dimethylbiphenyl as a starting material, which is significantly cheaper and more readily available than traditional halogenated biphenyl precursors. The methyl groups serve as versatile handles that can be sequentially transformed into various functional groups, eliminating the need for expensive pre-functionalized starting materials.
Q: What are the thermal stability characteristics of these compounds?
A: The resulting asymmetric spirobifluorene compounds exhibit high thermal stability, with melting points often exceeding 250°C. The orthogonal sp3-hybridized spiro carbon prevents close packing of chromophores, enhancing solubility and preventing crystallization, which is critical for the longevity and performance of OLED devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Spirobifluorene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of electronic chemical synthesis, including stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards of the OLED industry. We understand the critical nature of material consistency in display manufacturing and have implemented robust quality assurance protocols to guarantee the performance of our spirobifluorene derivatives in final devices.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our methyl-transformation based intermediates. We encourage you to contact us for specific COA data and route feasibility assessments tailored to your specific R&D requirements, ensuring a seamless transition from laboratory discovery to commercial reality.
