Advanced Blue Light-Emitting Anthracene Derivatives for High-Performance OLED Displays
Introduction to Next-Generation Blue OLED Emitters
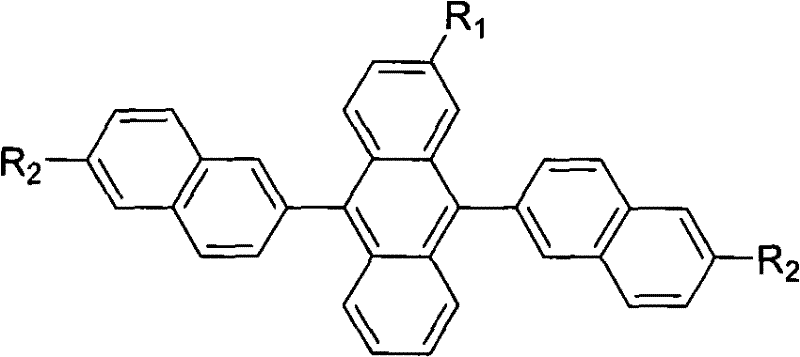
The rapid evolution of the organic light-emitting display (OLED) industry has created an urgent demand for high-performance blue light-emitting materials that offer superior stability and color purity. As detailed in patent CN101698796A, a novel class of substituted 9,10-dinaphthyl anthracene derivatives has been developed to address the critical limitations of existing blue emitters, such as film instability and low quantum efficiency. This breakthrough technology involves a strategic molecular design where rigid radicals, including methyl, ethyl, isopropyl, and tert-butyl groups, are introduced into the core anthracene structure. By modifying the steric environment of the molecule, this innovation effectively disrupts molecular symmetry and increases intermolecular spacing, thereby solving the persistent problem of crystallization in thin films. The result is a material that maintains an amorphous state even after annealing, delivering near-dark blue emission with significantly enhanced brightness and longevity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional blue light-emitting materials, particularly the standard 9,10-di(2-naphthyl)anthracene (ADN), have long suffered from inherent structural weaknesses that compromise device performance over time. Under prolonged operation in an electric field or during thermal annealing processes at temperatures around 95°C, these conventional materials exhibit unstable film morphology and a strong tendency to crystallize. This crystallization leads to the formation of non-uniform films, which creates defects that act as quenching sites for excitons, drastically reducing the lifespan and efficiency of the OLED device. Furthermore, the photochromic properties of classical blue materials often deviate towards the green region of the spectrum, with CIE coordinates typically around x=0.20, y=0.26, making it difficult to achieve the high color purity required for true dark blue displays. Additionally, the fluorescence quantum efficiency of these legacy materials remains suboptimal, limiting the overall power efficiency of the display panel.
The Novel Approach
The innovative approach described in the patent overcomes these challenges through precise steric engineering of the anthracene core. By substituting the hydrogen atoms on the anthracene or naphthalene rings with bulky alkyl groups such as tert-butyl or trifluoromethyl, the new materials introduce significant steric hindrance. This structural modification physically prevents the molecules from packing too closely together, effectively inhibiting the crystallization process that plagues traditional ADN. Consequently, the resulting thin films remain amorphous and stable even after thermal treatment, ensuring consistent device performance. Moreover, this molecular redesign not only stabilizes the film morphology but also shifts the emission spectrum closer to pure dark blue while boosting the fluorescence quantum efficiency. The patent data confirms that these substituted derivatives achieve an efficiency increase of 1.2 to 1.5 times compared to traditional materials, marking a substantial leap forward in blue OLED technology.
Mechanistic Insights into Steric Engineering and Aromatization
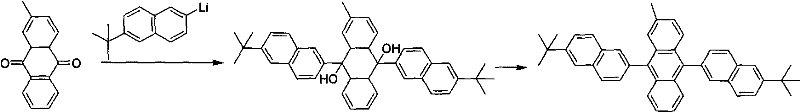
The synthesis and function of these advanced materials rely on a sophisticated understanding of organic reaction mechanisms and solid-state physics. The core synthetic strategy involves the nucleophilic addition of lithiated naphthalene derivatives to substituted anthraquinones. In this critical step, the organolithium species, generated at cryogenic temperatures to ensure stability, attacks the carbonyl carbons of the anthraquinone. This forms a diol intermediate, specifically a 9,10-dihydroxy-9,10-dihydroanthracene derivative. The presence of the bulky substituents on the naphthalene rings during this addition is crucial, as it dictates the final conformation of the molecule. Following the formation of the diol, an aromatization step is required to restore the conjugated anthracene system. This is typically achieved through reduction using stannous chloride in an acidic medium, which removes the hydroxyl groups and establishes the fully aromatic, planar central core while retaining the bulky peripheral groups that provide the necessary steric bulk.
Beyond the synthesis, the mechanism of improved performance lies in the suppression of concentration quenching. In conventional OLED materials, molecules tend to aggregate in the solid state, leading to non-radiative decay pathways that waste energy as heat. The rigid radicals introduced in this patent, such as the tert-butyl groups, act as physical spacers between the emissive anthracene cores. This increased intermolecular distance minimizes pi-pi stacking interactions, which are the primary cause of concentration quenching. As a result, the material can be used as a host or even a dopant at higher concentrations without suffering from efficiency roll-off. The disruption of symmetry also plays a vital role in preventing the ordered packing that leads to crystallization, ensuring that the material forms a uniform, pinhole-free amorphous film upon vacuum deposition. This combination of steric hindrance and electronic tuning allows for the realization of high-efficiency, stable dark blue emission that was previously unattainable with standard ADN derivatives.
How to Synthesize Substituted Dinaphthyl Anthracene Efficiently
The preparation of these high-value electronic chemicals follows a robust two-stage synthetic protocol that is amenable to optimization for various substituents. The process begins with the generation of a highly reactive organolithium intermediate from a halogenated naphthalene precursor, followed by its addition to a functionalized anthraquinone. This nucleophilic addition yields a diol intermediate which is subsequently aromatized to the final anthracene product. The versatility of this route allows for the incorporation of diverse functional groups, including alkyl chains and fluorinated moieties, simply by selecting the appropriate starting anthraquinone. For a comprehensive guide on the specific reaction conditions, stoichiometry, and workup procedures required to achieve high purity and yield, please refer to the standardized synthesis protocol outlined below.
- Perform lithiation of 6-tert-butyl-2-bromonaphthalene using n-Butyl Lithium at -78°C under nitrogen protection.
- Add the lithiated intermediate slowly to a solution of 2-substituted anthraquinone at low temperature to form the diol intermediate.
- Reduce the diol intermediate using stannous chloride dihydrate in glacial acetic acid at elevated temperatures to obtain the final anthracene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers distinct strategic advantages over traditional methods that rely on complex cross-coupling reactions. The elimination of expensive transition metal catalysts, such as palladium or iridium complexes which are often required for C-N or C-C bond formations in other OLED materials, translates directly into substantial cost savings. The reliance on commodity chemicals like n-butyl lithium, stannous chloride, and common solvents ensures that the raw material supply chain is robust and less susceptible to geopolitical fluctuations or shortages of precious metals. Furthermore, the solution-phase nature of the synthesis allows for flexible batch processing, enabling manufacturers to scale production from pilot kilograms to multi-ton commercial volumes without requiring specialized high-pressure or high-vacuum equipment typically associated with more exotic chemistries.
- Cost Reduction in Manufacturing: The synthetic pathway described in the patent utilizes cost-effective reagents and avoids the use of noble metal catalysts, which significantly lowers the bill of materials for the final product. The reduction step employs stannous chloride, a widely available and inexpensive reducing agent, rather than costly hydride sources or catalytic hydrogenation setups. This simplification of the reagent profile reduces both the direct material costs and the downstream costs associated with removing trace metal impurities, a critical requirement for electronic grade materials. Consequently, the overall manufacturing cost per kilogram is drastically reduced, improving the margin profile for downstream display manufacturers.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted anthraquinones and bromonaphthalenes, are established intermediates in the fine chemical industry with multiple global suppliers. This diversity in the supply base mitigates the risk of single-source dependency, ensuring a continuous and reliable flow of raw materials. Additionally, the reaction conditions, while requiring low temperatures for the lithiation step, do not demand extreme pressures or hazardous gases, simplifying the logistics of transportation and storage. This operational simplicity enhances the resilience of the supply chain, allowing for faster turnaround times and more predictable delivery schedules for high-purity electroluminescent materials.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard batch reactor operations commonly found in fine chemical plants. The absence of heavy metal catalysts simplifies waste treatment protocols, as there is no need for complex remediation processes to remove ppm-levels of palladium or other toxic metals from the final product or effluent streams. This aligns well with increasingly stringent environmental regulations regarding heavy metal discharge. The ability to produce high-quality material with a lower environmental footprint not only ensures regulatory compliance but also appeals to end-users who are prioritizing sustainable manufacturing practices in their electronics supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these substituted anthracene derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this material into your current OLED manufacturing workflow.
Q: How does the introduction of rigid radicals improve the material performance?
A: Introducing rigid radicals such as methyl, ethyl, or tert-butyl groups increases steric hindrance within the molecular structure. This breaks the symmetry of the traditional ADN molecule, increasing the intermolecular distance and preventing crystallization, which results in superior amorphous film morphology and enhanced thermal stability.
Q: What is the efficiency improvement compared to conventional blue light materials?
A: The patented substituted anthracene derivatives demonstrate a remarkable improvement in fluorescence quantum efficiency. Experimental data indicates that the efficiency of these new materials is approximately 1.2 to 1.5 times higher than that of traditional blue light-emitting materials, addressing the historical issue of low efficiency in blue OLEDs.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the synthesis utilizes standard organic chemistry techniques such as lithiation and reduction with stannous chloride, avoiding the need for expensive transition metal catalysts like palladium. The use of common solvents like THF and acetic acid, along with robust reaction conditions, makes the process highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Dinaphthyl Anthracene Supplier
As the demand for high-resolution, energy-efficient displays continues to surge, securing a stable supply of advanced blue emitting materials is paramount for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the rigorous purity specifications required for electronic chemicals, utilizing rigorous QC labs to ensure every batch meets the stringent standards necessary for OLED applications. We understand that consistency is key in display manufacturing, and our robust quality management systems guarantee the lot-to-lot reproducibility essential for high-yield panel production.
We invite you to collaborate with us to optimize your material sourcing strategy and accelerate your time-to-market for next-generation displays. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in organic synthesis can drive value and reliability in your supply chain.
