Advanced Synthesis of 2-Methyl-7-Benzimidazolylspirobifluorene for High-Performance OLEDs
The landscape of organic optoelectronics is continuously evolving, driven by the demand for materials that offer superior thermal stability and simplified device architectures. Patent CN102659688A introduces a significant breakthrough in this domain with the disclosure of 2-methyl-7-benzimidazolylspirobifluorene, a novel compound designed to address the limitations of traditional doping methods in white organic light-emitting diodes (WOLEDs). Unlike conventional approaches that rely on blending multiple emitters, which often suffer from phase separation and spectral instability over time, this single-molecule white light emitter leverages the rigid three-dimensional architecture of the spirobifluorene core. This structural innovation not only ensures high color purity and operational stability but also simplifies the manufacturing process by eliminating the need for complex host-guest engineering. As a reliable OLED material supplier, understanding the nuances of such advanced molecular designs is crucial for developing next-generation display technologies that meet the rigorous demands of the global market.
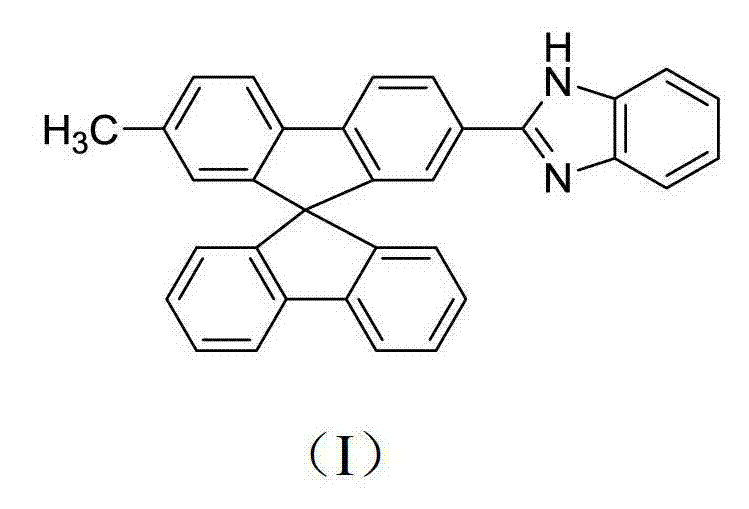
The strategic value of this compound extends beyond its electroluminescent properties; it also serves as a highly sensitive fluorescent probe for pH detection, demonstrating versatility in chemical sensing applications. The synthesis pathway outlined in the patent utilizes readily available starting materials like p-bromotoluene, which significantly lowers the barrier to entry for commercial production. By employing a sequence of coupling, bromination, Grignard reactions, and oxidative functionalization, the process achieves a balance between chemical complexity and industrial feasibility. For procurement managers and supply chain heads, this translates into a robust sourcing strategy where raw material availability is not a bottleneck. The ability to produce high-purity spirobifluorene derivatives through a scalable route positions this technology as a cornerstone for cost reduction in electronic chemical manufacturing, ensuring a steady supply of critical components for the burgeoning flat-panel display industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for achieving white light in OLEDs predominantly rely on the physical blending of red, green, and blue emitters or the use of excimer complexes, both of which present inherent stability challenges. In doped systems, the different rates of degradation among the various emissive species often lead to a shift in color coordinates over the device's lifetime, resulting in poor spectral stability and inconsistent performance. Furthermore, the phase separation of mixed materials under the stress of electrical driving and thermal load can cause a dramatic increase in driving voltage and a subsequent reduction in device lifespan. These issues necessitate complex encapsulation and precise control over layer thicknesses, driving up manufacturing costs and complicating the scale-up process. Additionally, many conventional blue-emitting spirobifluorene derivatives lack the functional groups necessary for broad-spectrum emission, limiting their utility as standalone white light sources and requiring additional energy-consuming layers to achieve the desired chromaticity.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by integrating a benzimidazole unit directly onto the spirobifluorene scaffold, creating a single-molecule system capable of emitting relatively pure white light. This molecular design exploits the intramolecular charge transfer interactions between the electron-donating spirobifluorene core and the electron-withdrawing benzimidazole group, facilitating a broad emission spectrum without the need for external dopants. The synthetic route is remarkably efficient, starting from commodity chemicals and proceeding through well-understood reaction mechanisms such as Grignard coupling and transition-metal catalyzed oxidation. This simplicity allows for tighter control over impurity profiles, which is critical for maintaining high fluorescence quantum yields. By avoiding the pitfalls of phase separation and leveraging the thermal robustness of the spiro linkage, this method offers a streamlined path to high-performance WOLEDs that are easier to manufacture and more reliable in operation, representing a significant leap forward in organic semiconductor technology.
Mechanistic Insights into FeCl3-Catalyzed Coupling and Oxidative Functionalization
The synthesis of the spirobifluorene core relies heavily on the precision of Grignard chemistry and subsequent acid-catalyzed cyclization, a process that demands rigorous control over reaction conditions to minimize byproduct formation. The initial coupling of p-bromotoluene involves the formation of a Grignard reagent in the presence of ferric chloride, which acts as a catalyst to facilitate the homocoupling reaction efficiently. This step is critical as it establishes the biphenyl backbone required for the subsequent spiro-formation. Following bromination, the second Grignard reaction with 9-fluorenone generates a tertiary alcohol intermediate, which undergoes dehydration and cyclization in the presence of acetic and hydrochloric acids to close the spiro ring. The mechanistic elegance lies in the orthogonality of the resulting structure, where the two fluorene units are perpendicular to each other, effectively suppressing pi-pi stacking interactions that typically lead to fluorescence quenching in planar aromatic systems.
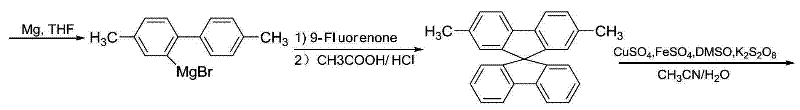
Further functionalization of the spiro core involves a sophisticated oxidative transformation using a copper and iron catalytic system with potassium persulfate as the oxidant. This step selectively converts one of the methyl groups into an aldehyde functionality, which is then condensed with o-phenylenediamine to form the benzimidazole ring. The choice of this catalytic system is particularly advantageous for industrial applications as it avoids the use of expensive noble metals like palladium or platinum, thereby reducing the overall cost of goods sold. Impurity control is managed through careful optimization of reaction temperatures, typically maintained between 110°C and 125°C during oxidation, and subsequent purification via column chromatography. This ensures that the final product meets the stringent purity specifications required for electronic applications, where even trace metal contaminants can act as quenching sites and degrade device performance. The result is a material with high thermal stability and excellent solubility, ready for vacuum deposition or solution processing.
How to Synthesize 2-Methyl-7-Benzimidazolylspirobifluorene Efficiently
The synthesis of this advanced OLED material requires a systematic approach that balances reaction efficiency with product purity, leveraging the detailed protocols established in the patent literature. The process begins with the preparation of the biphenyl precursor, followed by the construction of the spiro skeleton and final functionalization, each step requiring specific attention to stoichiometry and temperature control to maximize yield. Operators must ensure anhydrous conditions during the Grignard steps to prevent reagent decomposition, while the oxidation phase requires precise dosing of the oxidant to avoid over-oxidation to the carboxylic acid. The detailed standardized synthesis steps provided below outline the critical parameters for replicating this high-value chemical transformation in a production environment, ensuring consistency and reliability for commercial scale-up of complex organic semiconductors.
- Perform Grignard coupling of p-bromotoluene to form 4,4'-dimethylbiphenyl, followed by selective bromination.
- React the brominated biphenyl with 9-fluorenone via Grignard addition and acid-catalyzed dehydration to form the spirobifluorene core.
- Oxidize the methyl group to an aldehyde using a copper-iron catalytic system, then condense with o-phenylenediamine to finalize the benzimidazole structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this synthetic route offers compelling economic and logistical benefits that extend far beyond the laboratory scale. The reliance on p-bromotoluene as a primary feedstock ensures a stable and cost-effective supply chain, as this chemical is produced in massive quantities globally for various industrial applications, mitigating the risk of raw material shortages. Furthermore, the elimination of precious metal catalysts in the key oxidation step significantly reduces the cost burden associated with catalyst recovery and metal scavenging, which are often hidden expenses in fine chemical manufacturing. The robustness of the reaction conditions, which tolerate standard industrial solvents and equipment, facilitates a smoother transition from pilot scale to full commercial production, reducing the lead time for high-purity electroluminescent materials to reach the market. This operational efficiency translates directly into improved margins and a more resilient supply network for downstream device manufacturers.
- Cost Reduction in Manufacturing: The synthetic pathway is designed to minimize the use of expensive reagents and complex purification steps, leading to substantial cost savings in the overall production budget. By utilizing a copper-iron catalytic system instead of noble metals, the process avoids the high capital expenditure associated with precious metal recovery infrastructure. Additionally, the high yield of the cyclization and condensation steps reduces the volume of waste solvent generated per kilogram of product, further lowering disposal costs and environmental compliance fees. These factors combine to create a highly competitive cost structure that allows for aggressive pricing strategies without compromising on quality or profitability.
- Enhanced Supply Chain Reliability: The use of commodity chemicals and standard reaction types ensures that the supply chain is not vulnerable to the bottlenecks often associated with exotic or proprietary intermediates. The multi-step synthesis allows for the stocking of stable intermediates such as 2,7-dimethylspirobifluorene, providing a buffer against demand fluctuations and ensuring continuous availability for customers. This modularity in production planning enhances the reliability of delivery schedules, a critical factor for electronics manufacturers who operate on tight just-in-time inventory models. The ability to source raw materials from multiple vendors further de-risks the supply chain, ensuring uninterrupted production even in volatile market conditions.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction conditions that are easily adapted to large-scale reactors without significant loss of efficiency or selectivity. The aqueous workup procedures and the use of common organic solvents simplify waste treatment and recycling, aligning with increasingly stringent environmental regulations. The absence of heavy metal residues in the final product reduces the regulatory burden related to hazardous substance restrictions in electronic devices, such as RoHS compliance. This environmental stewardship not only protects the brand reputation of the manufacturer but also streamlines the approval process for new device certifications, accelerating time-to-market for innovative display technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel spirobifluorene compound, based on the detailed data provided in the patent documentation. Understanding these aspects is vital for stakeholders evaluating the material for integration into their product lines, as it clarifies the performance capabilities and manufacturing feasibility. The answers reflect a deep dive into the chemical properties and process advantages that distinguish this technology from existing alternatives in the organic electronics sector.
Q: What are the thermal stability characteristics of this spirobifluorene derivative?
A: The compound exhibits exceptional thermal stability with a decomposition temperature (Td) of 204.5°C and a melting point exceeding 250°C, making it highly suitable for vacuum deposition processes in OLED manufacturing.
Q: How does the spiro structure improve device performance?
A: The three-dimensional orthogonal structure of the spirobifluorene core effectively prevents close packing of chromophores, reducing fluorescence quenching and enhancing the fluorescence quantum yield for brighter, more efficient white light emission.
Q: Is this material suitable for applications beyond electroluminescence?
A: Yes, due to the benzimidazole moiety, the compound functions as a highly sensitive fluorescent pH probe with a wide response range in both acidic and alkaline environments, offering dual-use potential for sensing applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-7-Benzimidazolylspirobifluorene Supplier
As the demand for high-efficiency white OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis capabilities and rigorous quality assurance. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to seamlessly transition novel compounds from the research bench to the factory floor. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 2-methyl-7-benzimidazolylspirobifluorene meets the exacting standards required for high-performance display applications. We understand the critical nature of supply continuity in the electronics industry and are committed to delivering consistent quality that empowers your innovation.
We invite you to collaborate with our technical team to explore how this advanced material can enhance your product portfolio and drive down your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Let us be your strategic partner in navigating the complexities of organic semiconductor supply chains and achieving your commercial goals.
