Scalable Production of 2-Amino-4-Acetaminoanisole via Optimized DNCB Reduction and Acylation Technology
The evolution of dye intermediate manufacturing has long been driven by the necessity to balance raw material costs with process stability and environmental compliance. A pivotal advancement in this sector is documented in patent CN1146450A, which details a robust technological process for the preparation of 2-amino-4-acetaminoanisole utilizing 2,4-dinitrochlorobenzene as the primary feedstock. Historically, the industry relied heavily on p-anisidine-based routes, which, while mature, suffered from prohibitive material costs and significant waste generation. This new methodology represents a paradigm shift, leveraging the economic advantages of chloronitrobenzene derivatives while solving the critical technical challenge of intermediate instability through innovative reductive acylation techniques. For global procurement leaders, this transition signifies a move towards more sustainable and cost-effective supply chains for critical disperse dye precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for 2-amino-4-acetyl-anisidine predominantly utilized p-anisidine as the starting material, a route that, despite its operational maturity, presents severe economic and environmental drawbacks for modern large-scale manufacturing. The primary constraint lies in the exorbitant cost of p-anisidine relative to alternative nitro-chloro precursors, which drastically inflates the overall production cost of the final dye intermediate. Furthermore, conventional processes often struggle with lower product yields and generate substantial quantities of hazardous three wastes, complicating disposal and increasing the environmental footprint of the facility. The instability of intermediates in older methods frequently led to quality inconsistencies, such as discoloration and variable purity, necessitating costly downstream purification steps that further eroded profit margins. Consequently, the industry has urgently required a technological breakthrough that could decouple high quality from high raw material expenses.
The Novel Approach
The innovative process described in the patent data fundamentally restructures the synthesis logic by initiating the reaction sequence with 2,4-dinitrochlorobenzene, a significantly more economical raw material available in bulk quantities globally. This novel approach integrates methanol etherification followed by a specialized reduction and partial acylation sequence that eliminates the need for isolating the highly unstable 2,4-diaminoanisole intermediate. By maintaining the reaction mixture in a protected state throughout the transition from reduction to acylation, the process effectively mitigates oxidation risks that previously plagued similar synthetic routes. This seamless integration not only simplifies the operational workflow but also ensures a consistent total yield of approximately 80% with a product content exceeding 96.7%. The strategic elimination of intermediate isolation steps reduces solvent consumption and processing time, offering a compelling value proposition for cost-sensitive dye manufacturing operations.
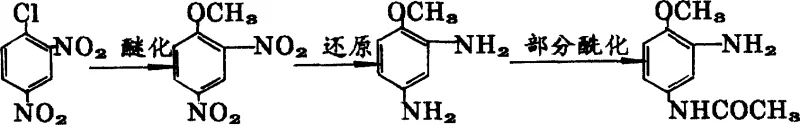
Mechanistic Insights into Protective Reductive Acylation
The core chemical innovation of this technology lies in the meticulous management of the 2,4-diaminoanisole intermediate, which is notoriously susceptible to oxidative degradation upon exposure to atmospheric oxygen. In standard reduction protocols, separating the diamine from the reduction catalyst or iron mud exposes the fresh amine groups to air, leading to rapid formation of tarry byproducts and a significant drop in assay. The patented mechanism circumvents this by leveraging the reducing environment inherent in the reaction mixture itself, effectively using the residual iron species or added metallic powders as sacrificial antioxidants. This creates a kinetic barrier against oxidation, allowing the subsequent acylation with acetic anhydride to proceed selectively on one amino group while preserving the integrity of the molecule. Such mechanistic control is critical for maintaining the pale color and high purity required for high-performance disperse dyes like Disperse Blue BGL.
Furthermore, the selectivity of the partial acylation is enhanced by precise temperature control and the presence of magnesium oxide as an acid scavenger within this protected matrix. The reaction conditions, typically maintained between 0-5°C during the addition of acetic anhydride, ensure that mono-acylation occurs preferentially over di-acylation, minimizing the formation of the 2,4-diacetylamino impurity. The subsequent warming to 60°C facilitates the completion of the reaction and aids in the coagulation of residual iron sludge, which can then be easily filtered off. This delicate balance of redox potential and thermal management ensures that the final product meets the rigorous melting point and purity specifications necessary for downstream dye synthesis, demonstrating a sophisticated understanding of process chemistry that translates directly to commercial reliability.
How to Synthesize 2-Amino-4-Acetaminoanisole Efficiently
The synthesis of this critical dye intermediate is structured around three distinct yet interconnected unit operations that maximize throughput while minimizing handling risks. The process begins with the etherification of 2,4-dinitrochlorobenzene in a methanol solvent system, followed immediately by a reduction step that can utilize either iron powder or catalytic hydrogenation depending on facility capabilities. The crucial final stage involves the direct partial acylation of the crude reduction mixture, bypassing the dangerous isolation of the free diamine. For R&D teams looking to implement this technology, the following guide outlines the standardized operational parameters derived from the patent examples to ensure reproducible high-yield results.
- Perform methanol etherification of 2,4-dinitrochlorobenzene under alkaline conditions to form 2,4-dinitrobenzene methyl ether.
- Conduct reduction using either iron powder or catalytic hydrogenation to generate the unstable 2,4-diaminoanisole intermediate without isolation.
- Execute partial acylation directly in the presence of protective agents (iron mud or added zinc/iron powder) to prevent oxidation and yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this 2,4-dinitrochlorobenzene-based route offers transformative advantages that extend far beyond simple laboratory yield improvements. The fundamental shift in raw material sourcing from expensive p-anisidine to commodity-grade nitro-chlorobenzenes creates a structural cost advantage that insulates the supply chain from volatile amine market fluctuations. Additionally, the simplification of the process flow, specifically the removal of intermediate isolation and drying steps, significantly reduces energy consumption and labor hours per kilogram of output. These operational efficiencies translate into a more resilient supply capability, allowing manufacturers to respond faster to market demand without the bottlenecks associated with complex multi-step purifications. Ultimately, this technology enables a reliable dye intermediate supplier to offer competitive pricing while maintaining strict quality standards.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the substitution of high-cost p-anisidine with 2,4-dinitrochlorobenzene, which is available at a fraction of the price, thereby drastically lowering the bill of materials. By eliminating the need to separate and dry the unstable diaminoanisole intermediate, the process removes entire unit operations, reducing utility costs associated with heating, cooling, and vacuum drying. The ability to recycle methanol solvent with high recovery rates further diminishes the variable costs associated with solvent procurement and waste treatment. These cumulative savings allow for a substantial reduction in the overall cost of goods sold, enhancing margin potential for downstream dye producers.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like 2,4-dinitrochlorobenzene ensures a more stable and diversified supply base compared to specialty amines which may have limited global suppliers. The robustness of the process, characterized by its tolerance to standard industrial equipment and conditions, minimizes the risk of batch failures that could disrupt delivery schedules. Furthermore, the high stability of the final product ensures that inventory can be held for longer periods without degradation, providing a buffer against demand spikes. This reliability is essential for maintaining continuous production lines in the textile and dyeing industries where interruptions are financially detrimental.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-thousand-liter reactors, which facilitates easy technology transfer from pilot to commercial scale without significant re-engineering. The reduction in waste generation, particularly the avoidance of complex aqueous waste streams from intermediate workups, simplifies effluent treatment and lowers compliance costs. The use of iron powder or reusable hydrogenation catalysts aligns with green chemistry principles by reducing the reliance on stoichiometric heavy metal reagents. This environmental profile makes the technology future-proof against tightening regulatory frameworks regarding industrial emissions and chemical safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent specifications and industrial application data. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this process into existing manufacturing portfolios. The answers provided reflect the specific operational parameters and quality outcomes observed in the documented examples, ensuring accuracy for decision-making purposes.
Q: Why is the 2,4-diaminoanisole intermediate difficult to handle in traditional synthesis?
A: The 2,4-diaminoanisole intermediate generated after reduction is extremely unstable and prone to rapid oxidation when exposed to air, leading to darkened products and low yields if not immediately protected.
Q: How does the new process prevent oxidation during acylation?
A: The process utilizes the residual iron mud from reduction or adds specific amounts of zinc/iron powder as a protective agent during the acylation step, creating a reducing environment that stabilizes the amine.
Q: What are the purity specifications achievable with this method?
A: This optimized technological process consistently achieves a product content of 96.7% with a total yield reaching approximately 80%, meeting stringent requirements for disperse dye synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Acetaminoanisole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final disperse dye products, and we are committed to delivering excellence through advanced process engineering. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2-amino-4-acetaminoanisole meets the exacting standards required for premium dye synthesis. Our commitment to process optimization allows us to consistently deliver materials that support your downstream manufacturing goals with minimal variability.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic advantages of switching to this DNCB-based technology for your operations. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a partnership built on transparency, quality, and mutual growth.
