Advanced Synthetic Route for Suvorexant Intermediates: Scalable Manufacturing Solutions
Advanced Synthetic Route for Suvorexant Intermediates: Scalable Manufacturing Solutions
The pharmaceutical landscape for Central Nervous System (CNS) disorders continues to evolve, with Suvorexant (MK-4305) standing out as a pivotal orexin receptor antagonist for treating insomnia. The efficient production of its key building block, 2-(4-methyl-2-carboxyphenyl)-1,2,3-triazole, remains a critical bottleneck for generic manufacturers and CDMOs aiming to enter this market. Patent CN103012293A introduces a transformative synthetic methodology that addresses these challenges by utilizing 4-methyl-2-carboxyphenylhydrazine hydrochloride as a foundational substrate. This novel approach bypasses the economic and technical hurdles associated with traditional routes, offering a streamlined pathway through hydrazone formation, oximation, cyclization, and deoxygenation. For R&D directors and procurement strategists, understanding this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials at competitive margins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(4-methyl-2-carboxyphenyl)-1,2,3-triazole has relied heavily on coupling reactions involving aryl iodides and triazole precursors, as documented in earlier patents such as WO2011050200. These legacy processes are fraught with significant commercial disadvantages, primarily driven by the exorbitant cost of iodinated starting materials and the necessity for expensive chiral resolving agents like L1 (e.g., (1S,2S)-(+)-1,2-cyclohexanediamine). Furthermore, the purification protocols in these conventional methods are notoriously arduous, often requiring repeated cycles of acidification, alkalization, and recrystallization to achieve acceptable purity levels. This not only inflates the operational expenditure but also drastically reduces the overall throughput, creating substantial supply chain vulnerabilities for downstream API manufacturers who require consistent, large-volume deliveries.
The Novel Approach
In stark contrast, the methodology disclosed in CN103012293A presents a robust alternative that fundamentally restructures the synthetic logic. By initiating the sequence with affordable hydrazine derivatives and employing a strategic four-step cascade, the process eliminates the need for precious metal catalysts or chiral auxiliaries. The route proceeds through the formation of a hydrazone intermediate, followed by conversion to an oxime, copper-catalyzed ring closure, and final deoxygenation. This sequence is not only chemically elegant but also operationally superior, as it avoids the generation of difficult-to-separate isomers. The result is a process that is inherently safer, more cost-effective, and ideally suited for industrial scale-up, providing a clear pathway for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Copper-Catalyzed Cyclization and Deoxygenation
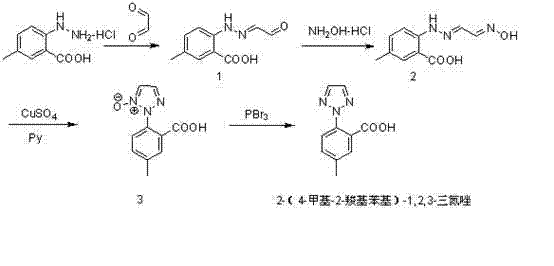
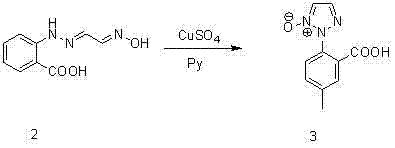
The core innovation of this synthetic strategy lies in the third step, where the triazole ring is constructed via a copper-mediated cyclization. In this critical transformation, the oxime intermediate reacts with copper sulfate in the presence of an organic base, such as pyridine or piperidine, at elevated temperatures ranging from 60°C to 100°C. This step facilitates the intramolecular closure to form the N-oxide triazole species (Compound 3). The use of copper sulfate is particularly advantageous from a commercial perspective, as it is a ubiquitous, low-cost commodity chemical compared to specialized palladium or rhodium catalysts often seen in cross-coupling reactions. The mechanism ensures high regioselectivity, effectively preventing the formation of unwanted positional isomers that typically plague triazole synthesis, thereby simplifying the downstream purification burden significantly.
Following the cyclization, the final deoxygenation step utilizes phosphorus halides, specifically phosphorus tribromide (PBr3) or phosphorus trichloride (PCl3), to reduce the N-oxide to the target triazole. This reaction is conducted under reflux conditions, typically between 50°C and 100°C, and is followed by a straightforward aqueous workup. The efficiency of this deoxygenation is evidenced by the high yields reported in the patent examples, often exceeding 90%. From a process chemistry standpoint, the combination of these two steps creates a powerful 'one-two punch' that builds the heterocyclic core with precision and removes the oxygen functionality without compromising the integrity of the carboxylic acid group, ensuring the final product meets stringent quality specifications required for API synthesis.
How to Synthesize 2-(4-methyl-2-carboxyphenyl)-1,2,3-triazole Efficiently
The synthesis of this critical intermediate is achieved through a logical progression of functional group transformations that maximize atom economy and minimize waste. The process begins with the condensation of the hydrazine salt with glyoxal, followed by oximation, setting the stage for the ring-closing event. Detailed operational parameters, including specific molar ratios and solvent choices like acetic acid or ethanol, are optimized to ensure reproducibility. For laboratory and pilot plant teams looking to implement this technology, the following guide outlines the standardized procedural framework derived directly from the patent embodiments, ensuring that the high yields and purity profiles demonstrated in the intellectual property can be replicated in your own facilities.
- React 4-methyl-2-hydrazinobenzoic acid hydrochloride with glyoxal to form the hydrazone intermediate (Compound 1).
- Convert Compound 1 to the oxime derivative (Compound 2) using hydroxylamine hydrochloride under basic conditions.
- Perform copper sulfate-catalyzed cyclization in the presence of an organic base to form the triazole N-oxide (Compound 3).
- Execute deoxygenation using phosphorus halides (PBr3 or PCl3) to yield the final 2-(4-methyl-2-carboxyphenyl)-1,2,3-triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the raw material portfolio; by replacing expensive iodinated aromatics and chiral resolving agents with commodity chemicals like copper sulfate and hydroxylamine, the direct material costs are significantly reduced. This shift not only lowers the Cost of Goods Sold (COGS) but also mitigates the risk of supply disruptions associated with niche reagents. Furthermore, the elimination of complex purification loops—such as repeated recrystallizations—translates into shorter cycle times and higher equipment utilization rates, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of high-value inputs with low-cost alternatives. By avoiding the use of proprietary chiral resolving agents and expensive aryl iodides, the raw material expenditure is drastically lowered. Additionally, the high yields observed in each step, particularly the near-quantitative conversion in the initial hydrazone formation and the final deoxygenation, mean that less starting material is wasted. This efficiency, combined with the use of standard solvents and reagents, results in substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final API in the generic marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, non-proprietary reagents. Chemicals such as glyoxal, hydroxylamine hydrochloride, and copper sulfate are produced globally in massive quantities, ensuring that sourcing is never a bottleneck. This contrasts sharply with legacy methods that depend on specialized catalysts or custom-synthesized intermediates which may have long lead times. By securing a supply line based on commodity chemicals, manufacturers can guarantee continuous production schedules, reducing the risk of stockouts and ensuring a steady flow of high-purity pharmaceutical intermediates to downstream partners.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for commercial expansion. The process avoids the generation of heavy metal waste streams associated with palladium or other precious metal catalysts, simplifying effluent treatment and reducing environmental compliance costs. The workup procedures, which primarily involve extraction and drying rather than energy-intensive distillation or chromatography, are easily transferable from kilogram to multi-ton scales. This inherent scalability ensures that as demand for Suvorexant grows, the production capacity for its intermediates can be ramped up seamlessly without requiring significant capital investment in new specialized infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Whether you are evaluating the technical feasibility for your R&D pipeline or assessing the commercial viability for your procurement strategy, these answers offer the clarity needed to make informed decisions about integrating this intermediate into your supply chain.
Q: What are the key advantages of this synthesis route over prior art methods?
A: Unlike conventional methods that rely on expensive aryl iodides and chiral resolving agents, this route utilizes low-cost hydrazine derivatives and avoids complex resolution steps, significantly lowering production costs and simplifying purification.
Q: Does this method produce isomeric impurities?
A: No, the patented process is specifically designed to avoid the formation of isomers, ensuring high regioselectivity and purity of the final 1,2,3-triazole intermediate without the need for extensive chromatographic separation.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method employs readily available reagents like copper sulfate and phosphorus halides, and features simple workup procedures such as extraction and drying, making it highly scalable for commercial API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-methyl-2-carboxyphenyl)-1,2,3-triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex CNS drugs like Suvorexant depends on the availability of high-quality intermediates produced via robust, scalable routes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 2-(4-methyl-2-carboxyphenyl)-1,2,3-triazole meets the highest industry standards, providing you with the confidence needed to advance your drug development programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and cost-effective supply of this critical pharmaceutical intermediate.
