Advanced Manufacturing of Sartan Intermediates: A Safe, Scalable Tetrazole Cyclization Strategy
Advanced Manufacturing of Sartan Intermediates: A Safe, Scalable Tetrazole Cyclization Strategy
The global demand for angiotensin II receptor antagonists, commonly known as Sartans, continues to surge as these medications remain the gold standard for managing hypertension and protecting target organs. At the heart of synthesizing blockbuster drugs like Losartan, Valsartan, and Irbesartan lies the critical building block: 2-(1-hydro-4-tetrazole)-4'-methylbiphenyl and its diverse derivatives. 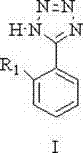 Recent intellectual property developments, specifically patent CN102060798A, have unveiled a transformative synthetic methodology that addresses long-standing safety and purity challenges in producing these essential pharmaceutical intermediates. This technical insight report analyzes the breakthrough disclosed in the patent, offering a comprehensive evaluation for R&D directors and supply chain leaders seeking to optimize their production of high-purity pharmaceutical intermediates. By shifting away from hazardous azide chemistry toward a hydrazine-based [3+2] cycloaddition strategy, this route promises not only enhanced operational safety but also significant improvements in impurity profiles and cost structures for commercial scale-up of complex pharmaceutical intermediates.
Recent intellectual property developments, specifically patent CN102060798A, have unveiled a transformative synthetic methodology that addresses long-standing safety and purity challenges in producing these essential pharmaceutical intermediates. This technical insight report analyzes the breakthrough disclosed in the patent, offering a comprehensive evaluation for R&D directors and supply chain leaders seeking to optimize their production of high-purity pharmaceutical intermediates. By shifting away from hazardous azide chemistry toward a hydrazine-based [3+2] cycloaddition strategy, this route promises not only enhanced operational safety but also significant improvements in impurity profiles and cost structures for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the tetrazole ring, a pharmacophore essential for the biological activity of Sartan drugs, has relied heavily on the use of sodium azide (NaN3) as the nitrogen source. While chemically effective, the reliance on azides introduces severe logistical and safety bottlenecks for large-scale manufacturing. Sodium azide is classified as a highly explosive and toxic substance, necessitating rigorous and costly safety protocols for its transportation, storage, and handling within a production facility. Furthermore, traditional catalytic systems designed to facilitate this azide coupling often employ organotin reagents, such as trimethyltin chloride (Me3SnCl) or tributyltin chloride. These heavy metal catalysts are notoriously difficult to remove completely from the final product, often leaving trace residues that fail to meet the stringent purity specifications required for active pharmaceutical ingredients (APIs). Even alternative methods utilizing zinc or lithium salts, while an improvement, still fundamentally depend on the hazardous azide reagent, leaving the process vulnerable to regulatory scrutiny and environmental compliance issues regarding the disposal of azide-containing waste streams.
The Novel Approach
The methodology detailed in patent CN102060798A represents a paradigm shift by completely eliminating sodium azide from the synthetic sequence. Instead, this innovative route utilizes commercially available ortho-cyanobiphenyl derivatives as the starting material, reacting them initially with alcohols in a protic solvent under acidic conditions to form an imidate intermediate. This is followed by a nucleophilic substitution with hydrazine hydrate to generate an amidrazone precursor, which finally undergoes a diazotization-driven [3+2] cycloaddition to close the tetrazole ring. This approach leverages mild reaction conditions and avoids the use of toxic organotin catalysts entirely, relying instead on accessible inorganic acids or Lewis acids. By replacing high-risk reagents with safer alternatives like hydrazine hydrate and sodium nitrite, the process significantly reduces the environmental footprint and simplifies the regulatory burden associated with hazardous material management, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Hydrazine-Mediated Tetrazole Formation
The core of this synthetic innovation lies in the stepwise construction of the heterocyclic ring through a carefully orchestrated sequence of nucleophilic additions and cyclizations. The process begins with the activation of the nitrile group on the biphenyl scaffold. Under the influence of a strong inorganic acid catalyst, such as hydrochloric acid or hydrobromic acid, the nitrogen atom of the cyano group is protonated, thereby increasing the electrophilicity of the adjacent carbon atom. This activation allows a protic solvent molecule, typically an alcohol like methanol or ethanol, to attack the carbon center, resulting in the formation of an imidate ester intermediate (Compound III). This step is crucial as it converts the relatively inert nitrile into a more reactive species capable of undergoing subsequent transformation with hydrazine. The reaction is typically conducted at moderate temperatures, often ranging from 5°C to 35°C initially, followed by heating to ensure complete conversion, demonstrating a high degree of control over the reaction kinetics to minimize side products.
Following the formation of the imidate, the second stage involves the displacement of the alkoxy group by hydrazine hydrate under reflux conditions. This hydrazinolysis reaction yields the key amidrazone intermediate (Compound IV), which serves as the direct precursor for ring closure. 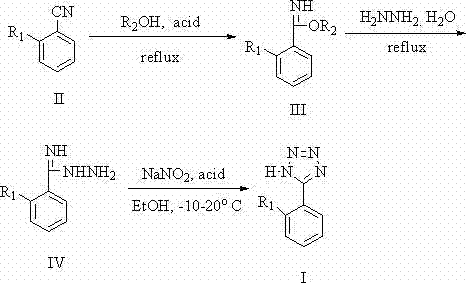 The final and most critical step is the [3+2] cycloaddition, driven by the in situ generation of a nitrosating agent. By introducing sodium nitrite (NaNO2) in the presence of acid at controlled low temperatures between -10°C and 50°C, the amidrazone undergoes diazotization. This generates a reactive diazonium species that immediately cyclizes with the adjacent nitrogen to form the stable tetrazole ring (Compound I). This low-temperature control is vital for suppressing the formation of decomposition byproducts and ensuring high regioselectivity. The result is a clean reaction profile that facilitates easier downstream purification, directly contributing to the high purity and yield (>60% overall) reported in the patent examples, which is a critical metric for any reliable pharmaceutical intermediates supplier.
The final and most critical step is the [3+2] cycloaddition, driven by the in situ generation of a nitrosating agent. By introducing sodium nitrite (NaNO2) in the presence of acid at controlled low temperatures between -10°C and 50°C, the amidrazone undergoes diazotization. This generates a reactive diazonium species that immediately cyclizes with the adjacent nitrogen to form the stable tetrazole ring (Compound I). This low-temperature control is vital for suppressing the formation of decomposition byproducts and ensuring high regioselectivity. The result is a clean reaction profile that facilitates easier downstream purification, directly contributing to the high purity and yield (>60% overall) reported in the patent examples, which is a critical metric for any reliable pharmaceutical intermediates supplier.
How to Synthesize 2-(1-hydro-4-tetrazole)-4'-methylbiphenyl Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and safety. The protocol outlined in the patent provides a robust framework for transitioning from laboratory benchtop to pilot plant operations. The process is divided into three distinct operational units: the acid-catalyzed imidate formation, the hydrazine reflux step, and the final nitrosative cyclization. Each stage utilizes common industrial solvents and reagents, ensuring that the supply chain for raw materials remains stable and cost-effective. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Perform acid-catalyzed nucleophilic addition of o-cyanobiphenyl derivatives in a protic solvent to form the imidate intermediate.
- React the imidate intermediate with hydrazine hydrate under reflux conditions to generate the amidrazone precursor.
- Execute the final [3+2] cycloaddition using sodium nitrite and acid at controlled low temperatures (-10 to 50°C) to close the tetrazole ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this azide-free methodology offers substantial strategic advantages beyond mere chemical elegance. The primary benefit is the drastic simplification of the safety infrastructure required for production. By removing sodium azide and organotin catalysts from the bill of materials, facilities can avoid the exorbitant costs associated with storing and handling Class 1 explosives and toxic heavy metals. This reduction in hazard classification translates directly into lower insurance premiums, reduced waste disposal fees, and minimized downtime related to safety audits. Furthermore, the use of commodity chemicals like hydrazine hydrate and sodium nitrite ensures a resilient supply chain, as these reagents are widely available from multiple global vendors, reducing the risk of supply disruptions that often plague specialty reagent markets.
- Cost Reduction in Manufacturing: The elimination of expensive organotin catalysts removes the need for costly downstream purification steps, such as specialized resin treatments or activated carbon filtration, which are traditionally required to meet heavy metal limits. Additionally, the avoidance of azide waste streams significantly lowers the cost of environmental compliance and hazardous waste incineration. The process operates under mild conditions with high atom economy in the final cyclization step, leading to substantial cost savings in utility consumption and raw material usage compared to legacy methods.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals rather than specialized, hazardous reagents enhances the stability of the supply chain. Raw materials such as ortho-cyanobiphenyls and hydrazine hydrate are produced at massive scales globally, ensuring consistent availability and price stability. This reliability allows for better long-term planning and inventory management, reducing the lead time for high-purity pharmaceutical intermediates and ensuring continuous production schedules without the interruptions often caused by regulatory holds on toxic substances.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of exothermic runaway risks associated with azide chemistry. The reaction temperatures are easily manageable in standard glass-lined or stainless steel reactors, facilitating a smooth transition from kilogram to multi-ton production scales. Moreover, the "green" nature of the process, characterized by the absence of heavy metal residues and explosive waste, aligns perfectly with modern environmental, social, and governance (ESG) goals, making it easier to obtain regulatory approvals in stringent markets like the EU and North America.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the technology's feasibility for your specific application needs.
Q: Why is the hydrazine hydrate route preferred over sodium azide for tetrazole synthesis?
A: Traditional methods utilizing sodium azide (NaN3) pose significant safety hazards regarding transportation and storage due to explosion risks. Furthermore, classic catalytic systems often employ toxic organotin compounds (e.g., Me3SnCl), leading to difficult-to-remove heavy metal residues in the final API. The hydrazine route described in CN102060798A eliminates these toxic catalysts and explosive reagents, ensuring a safer, greener profile suitable for GMP manufacturing.
Q: What yields can be expected from this novel synthetic pathway?
A: According to the patent data, the overall process demonstrates robust efficiency with a total yield exceeding 60%. Specific experimental examples highlight high conversion rates at individual stages, such as 80% for the imidate formation, 87% for the hydrazinolysis step, and up to 90% for the final cyclization, indicating a highly viable process for industrial scale-up.
Q: Does this method require expensive transition metal catalysts?
A: No, this methodology specifically avoids the use of expensive and toxic transition metal catalysts like organotins. Instead, it utilizes readily available inorganic acids (HCl, HBr) or Lewis acids (ZnCl2, FeCl3) and common reagents like sodium nitrite. This substitution drastically reduces raw material costs and simplifies the downstream purification process by removing the need for specialized heavy metal scavenging steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(1-hydro-4-tetrazole)-4'-methylbiphenyl Supplier
The synthetic route described in patent CN102060798A represents a significant advancement in the manufacturing of Sartan intermediates, offering a safer and more efficient pathway to market. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative chemistries for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you require clinical trial materials or full commercial supply, we can deliver with consistency. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, ensuring that every batch of 2-(1-hydro-4-tetrazole)-4'-methylbiphenyl meets the highest industry standards for impurity control and identity.
We invite you to explore how this optimized synthesis can enhance your project's economics and timeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to process innovation can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
