Advanced Palladium-Catalyzed Synthesis of Chiral 3-(2,3-Butadienyl)Oxindoles for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust methodologies for constructing complex molecular architectures, particularly those containing quaternary carbon centers which are prevalent in bioactive natural products and drug candidates. Patent CN113548999A introduces a groundbreaking approach for the preparation of racemic and chiral 3-(2,3-butadienyl)oxindolinone compounds via a palladium-catalyzed coupling reaction. This technology represents a significant leap forward in synthetic efficiency, enabling the direct assembly of these valuable scaffolds from readily available 2,3-butadienyl carbonates and oxindolinones. The process is notable not only for its operational simplicity but also for its exceptional ability to control stereochemistry, achieving high enantioselectivity and chemical selectivity in a single transformation. Furthermore, the resulting compounds have shown promising binding capabilities with the SARS-CoV-2 main protein 3CL hydrolase, highlighting their potential utility in the urgent development of antiviral therapeutics.
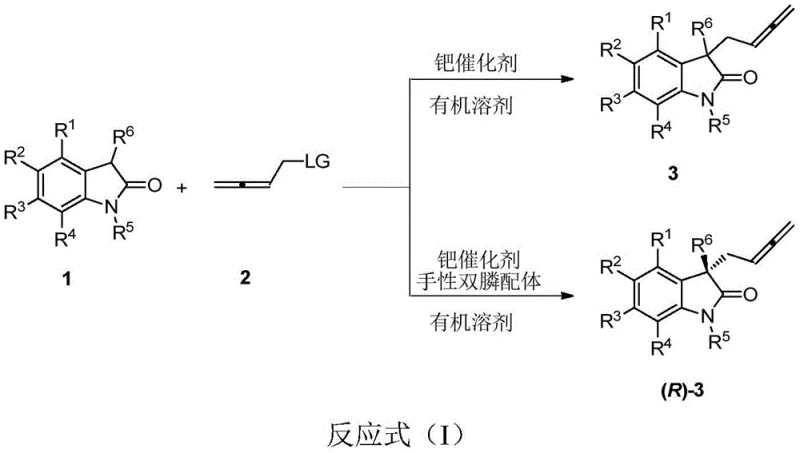
For R&D directors evaluating new synthetic routes, the mechanistic elegance of this transformation offers a compelling alternative to legacy methods. Conventional strategies for synthesizing 3,3-disubstituted oxindoles often involve multiple steps, protecting group manipulations, and struggle to achieve high levels of stereocontrol at the quaternary center. These traditional pathways can lead to complex impurity profiles and reduced overall yields, complicating the purification process and increasing production costs. In contrast, the novel approach detailed in this patent utilizes a transition metal-catalyzed asymmetric coupling that bypasses many of these hurdles. By leveraging specific chiral phosphine ligands in conjunction with palladium catalysts, the reaction directly constructs the desired allene-containing oxindole structure with remarkable precision. This shift from multi-step sequences to a concise, one-step coupling significantly reduces the process mass intensity and simplifies the workflow for process chemists aiming to scale up these critical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of optically active allene compounds containing quaternary carbon centers has been a formidable challenge in organic synthesis. Traditional methods often rely on the use of racemic 2,3-butadienol derivatives coupled with nucleophiles, which frequently results in mixtures of stereoisomers that are difficult to separate. Additionally, the inherent reactivity of oxindolinones can lead to unwanted side reactions, such as the formation of 1,3-conjugated diene products instead of the desired allene structure, due to enol tautomerization issues under transition metal catalysis. These limitations necessitate extensive optimization of reaction conditions and often require expensive chiral auxiliaries or resolution steps that drastically lower the overall atom economy. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, while supply chain heads face risks associated with the availability of specialized reagents required for these convoluted synthetic pathways.
The Novel Approach
The methodology disclosed in patent CN113548999A overcomes these historical barriers by employing a highly selective palladium-catalyzed system. By utilizing 2,3-butadienyl carbonates as electrophiles and functionalized oxindolinones as nucleophiles, the reaction proceeds through a well-defined catalytic cycle that favors the formation of the target 3-(2,3-butadienyl)oxindolinone structure. The use of tailored chiral phosphine ligands, such as the (R)-L series described in the patent, ensures that the stereochemical outcome is tightly controlled, delivering products with high enantiomeric excess. This approach not only streamlines the synthesis but also exhibits broad substrate scope, accommodating various electron-donating and electron-withdrawing substituents on the aromatic rings. Such versatility is crucial for medicinal chemistry campaigns where rapid analog generation is required to optimize biological activity without being constrained by synthetic feasibility.
Mechanistic Insights into Palladium-Catalyzed Asymmetric Coupling
Understanding the catalytic cycle is essential for appreciating the robustness of this technology. The reaction initiates with the oxidative addition of the palladium catalyst to the 2,3-butadienyl carbonate, generating a pi-allyl palladium intermediate. This species is highly reactive and undergoes rapid isomerization to form a more stable configuration. Subsequently, the enol form of the oxindolinone coordinates to the palladium center, facilitating a ligand exchange process. The stereochemical fate of the product is determined during the final reductive elimination step, where the chiral environment provided by the phosphine ligand directs the formation of the new carbon-carbon bond with high fidelity. This mechanism effectively suppresses the formation of thermodynamic byproducts like conjugated dienes, ensuring that the kinetic product—the desired allene—is obtained with high chemical selectivity. The ability to tune the electronic and steric properties of the ligand allows for fine adjustments to the reaction profile, making it adaptable to a wide range of substrate combinations.
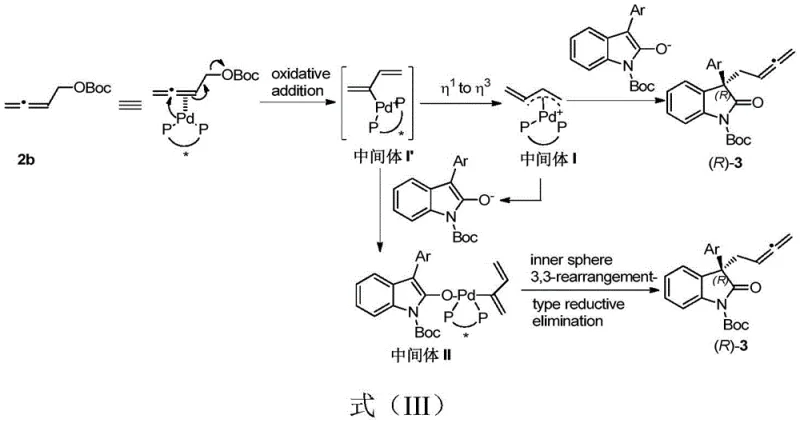
From an impurity control perspective, this mechanism offers distinct advantages. The high chemoselectivity of the palladium catalyst minimizes the generation of structural isomers and oligomeric byproducts that are common in less controlled coupling reactions. The specific interaction between the catalyst and the substrates ensures that the reaction pathway is directed exclusively towards the formation of the quaternary carbon center at the 3-position of the oxindole ring. This purity profile is critical for pharmaceutical applications, where regulatory standards demand stringent control over impurity levels. By reducing the burden on downstream purification processes, this technology enhances the overall efficiency of the manufacturing process. For quality assurance teams, the predictability of the impurity profile simplifies validation efforts and supports the establishment of robust control strategies for commercial production.
How to Synthesize 3-(2,3-Butadienyl)Oxindoles Efficiently
The practical implementation of this synthesis involves careful control of reaction parameters to maximize yield and enantioselectivity. The process typically begins with the preparation of a reaction vessel under an inert atmosphere to prevent catalyst deactivation. Specific ratios of palladium source and chiral ligand are introduced, followed by the substrates dissolved in an appropriate organic solvent such as acetonitrile or DMPU. Temperature control is paramount, with reactions often conducted at low temperatures ranging from -30°C to -25°C to maintain high stereocontrol. The detailed standardized synthesis steps are outlined in the guide below, providing a clear roadmap for laboratory execution and subsequent scale-up activities.
- Prepare a Schlenk reaction tube under argon protection and add palladium catalyst along with the specific chiral phosphine ligand.
- Introduce the functionalized oxindolinone and 2,3-butadienyl carbonate substrates into the organic solvent system.
- Maintain the reaction at low temperatures between -30°C to -25°C for extended periods to ensure high enantioselectivity before purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers substantial strategic benefits beyond mere technical performance. The reliance on commercially available starting materials, such as substituted oxindoles and butadienyl carbonates, mitigates the risk of supply disruptions associated with exotic or custom-synthesized reagents. The one-step nature of the reaction significantly reduces the number of unit operations required, leading to a drastic simplification of the manufacturing process. This reduction in complexity translates directly into lower operational expenditures and a smaller physical footprint for production facilities. Furthermore, the high conversion rates and selectivity minimize waste generation, aligning with modern sustainability goals and reducing the costs associated with waste disposal and environmental compliance.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps and protecting group manipulations inherently lowers the cost of goods sold. By avoiding the need for expensive chiral resolution techniques or auxiliary reagents, the process achieves cost efficiency through intrinsic stereocontrol. The high atom economy of the coupling reaction ensures that a greater proportion of raw materials are converted into the final product, reducing material costs. Additionally, the simplified workup procedures decrease the consumption of solvents and purification media, further contributing to overall cost savings in large-scale production environments.
- Enhanced Supply Chain Reliability: The use of robust and widely available catalysts and ligands ensures a stable supply chain. Unlike proprietary enzymes or fragile biocatalysts that may require cold chain logistics, the palladium systems described are stable and easy to handle. The broad substrate scope means that variations in raw material specifications can often be accommodated without significant re-optimization of the process. This flexibility allows procurement teams to source materials from multiple vendors, reducing dependency on single suppliers and enhancing the resilience of the supply network against market fluctuations.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not require extreme pressures or temperatures, making the process inherently safer and easier to scale from kilogram to tonne quantities. The reduced generation of hazardous byproducts simplifies effluent treatment and lowers the environmental impact of the manufacturing process. This alignment with green chemistry principles facilitates regulatory approval and supports corporate sustainability initiatives. The ability to produce high-purity intermediates efficiently positions manufacturers to meet the growing demand for antiviral and other therapeutic agents without compromising on environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis technology. These answers are derived from the detailed experimental data and mechanistic studies presented in the patent documentation. They provide clarity on the feasibility, scope, and potential applications of the described methodology for stakeholders involved in drug development and manufacturing.
Q: What is the primary chemical challenge addressed by this patent?
A: The patent addresses the difficult construction of tetra-substituted optical active quaternary carbon centers, specifically within the 3,3-disubstituted oxindolinone framework, which is crucial for bioactivity but hard to synthesize with high stereoselectivity.
Q: How does this method improve upon traditional synthesis routes?
A: Unlike traditional multi-step approaches that often suffer from poor stereocontrol or require harsh conditions, this method achieves direct one-step construction using a palladium-catalyzed coupling reaction with excellent functional group compatibility and high conversion rates.
Q: What are the potential therapeutic applications of these compounds?
A: The synthesized 3-(2,3-butadienyl)oxindolinone compounds and their derivatives have demonstrated binding affinity to the SARS-CoV-2 main protein 3CL hydrolase, indicating significant potential for developing broad-spectrum antiviral medications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(2,3-Butadienyl)Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality intermediates for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from discovery to market. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee the consistency and reliability of every batch we deliver. Our expertise in palladium-catalyzed transformations allows us to optimize this specific technology for your unique requirements, delivering cost-effective solutions without compromising on quality.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain insights into the potential economic advantages of adopting this method for your specific targets. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us partner with you to accelerate the development of vital antiviral medications and other life-saving therapies through superior chemical manufacturing.
