Advanced Benzimidazole Bisphosphine Copper Complexes for High-Efficiency OLED Display Manufacturing
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that balance exceptional photophysical performance with economic viability. Patent CN111848676A introduces a groundbreaking class of luminescent copper compounds based on benzimidazole bisphosphine ligands, representing a significant leap forward in display chemical manufacturing. Unlike traditional phosphorescent emitters reliant on scarce and expensive heavy metals like iridium or platinum, these novel Cu(I) complexes leverage earth-abundant copper to achieve high internal quantum efficiency. The core innovation lies in the specific molecular architecture, designated as Formula I, which stabilizes the copper center while optimizing spin-orbit coupling for efficient triplet harvesting. 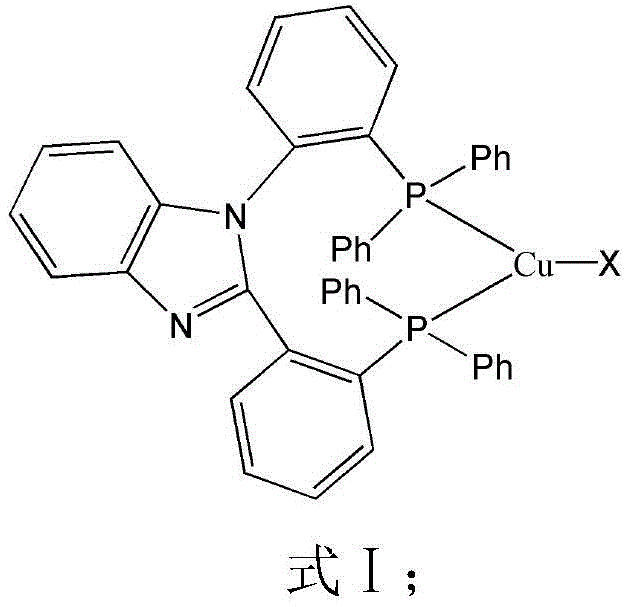 As detailed in the patent, these compounds exhibit strong absorption near 230nm and demonstrate remarkable solid-state photoluminescence quantum efficiencies reaching up to 71.53%, positioning them as prime candidates for next-generation energy-saving lighting and flexible display technologies.
As detailed in the patent, these compounds exhibit strong absorption near 230nm and demonstrate remarkable solid-state photoluminescence quantum efficiencies reaching up to 71.53%, positioning them as prime candidates for next-generation energy-saving lighting and flexible display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the OLED sector has been dominated by organometallic complexes utilizing d6 and d8 transition metals, specifically Iridium(I), Platinum(II), and Osmium(III). While these materials offer excellent color purity and efficiency due to strong spin-orbit coupling, their widespread adoption faces severe bottlenecks regarding resource availability and cost. Iridium, in particular, is one of the rarest elements in the Earth's crust, leading to volatile pricing and supply chain vulnerabilities that threaten the scalability of mass-produced consumer electronics. Furthermore, the synthesis of these heavy metal complexes often requires harsh reaction conditions and intricate purification steps to remove toxic metal residues, complicating the manufacturing workflow and increasing the environmental footprint. The reliance on such precious metals creates a significant barrier to entry for cost-sensitive applications, limiting the potential for OLED technology to penetrate broader markets beyond high-end mobile devices and televisions.
The Novel Approach
The methodology outlined in CN111848676A circumvents these challenges by substituting precious metals with monovalent copper, a strategy that fundamentally alters the cost structure of OLED material production. This novel approach utilizes a rationally designed benzimidazole bisphosphine ligand system that rigidifies the coordination environment around the copper atom, thereby suppressing non-radiative decay pathways that typically plague Cu(I) emitters. By carefully tuning the steric and electronic properties of the ligand, the inventors have achieved emission lifetimes exceeding 61 microseconds and quantum efficiencies comparable to state-of-the-art iridium complexes. This shift not only drastically reduces raw material costs but also simplifies the regulatory landscape regarding heavy metal disposal. The ability to tune the emission color from orange-red to deep red simply by changing the halide counterion (I, Cl, or Br) adds a layer of versatility that is crucial for full-color display applications, offering a robust alternative to conventional phosphorescent dopants.
Mechanistic Insights into Cu(I)-Benzimidazole Bisphosphine Coordination
The exceptional performance of these luminescent copper compounds stems from the unique electronic interplay between the Cu(I) center and the bulky benzimidazole bisphosphine ligand. In the ground state, the copper atom adopts a distorted tetrahedral geometry, coordinated by two nitrogen atoms from the benzimidazole moiety and two phosphorus atoms from the diphenylphosphine groups. This specific coordination geometry is critical for minimizing structural flattening in the excited state, a phenomenon known as pseudo-Jahn-Teller distortion, which often leads to thermal quenching in copper complexes. The rigid backbone of the ligand, synthesized through a multi-step pathway involving key intermediates, effectively locks the copper center in place, preserving the emissive triplet state. 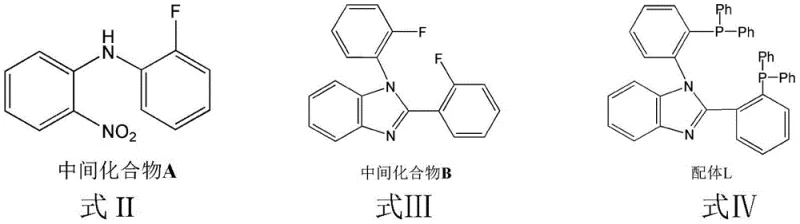 The presence of the benzimidazole ring enhances the electron-withdrawing character of the ligand, facilitating charge transfer transitions that are responsible for the observed luminescence. Furthermore, the choice of halide (X = I, Cl, Br) influences the crystal packing and intermolecular interactions, allowing for fine-tuning of the emission wavelength and lifetime without altering the core ligand structure.
The presence of the benzimidazole ring enhances the electron-withdrawing character of the ligand, facilitating charge transfer transitions that are responsible for the observed luminescence. Furthermore, the choice of halide (X = I, Cl, Br) influences the crystal packing and intermolecular interactions, allowing for fine-tuning of the emission wavelength and lifetime without altering the core ligand structure.
Impurity control is paramount in OLED materials, as trace contaminants can act as quenching sites that degrade device performance. The synthetic route described in the patent incorporates rigorous purification protocols at each stage, including solvent extraction, rotary evaporation, and recrystallization, to ensure high chemical purity. The use of palladium-catalyzed cross-coupling in the initial step to form Intermediate A is optimized with specific ligands like BINAP to minimize side reactions and homocoupling byproducts. Subsequent cyclization to form the benzimidazole ring (Intermediate B) utilizes sodium dithionite as a mild reducing agent, avoiding the formation of harsh acidic or basic waste streams that could degrade the sensitive phosphine groups. The final complexation step is performed at ambient temperatures (20-35°C), preventing thermal decomposition of the ligand and ensuring the formation of well-defined crystalline products suitable for vacuum deposition in OLED device fabrication.
How to Synthesize Luminescent Copper Compounds Efficiently
The preparation of these high-performance emitters follows a logical four-step sequence that balances yield with operational simplicity, making it highly attractive for process chemists aiming for commercial scale-up. The process begins with the construction of the nitrogen-containing scaffold, followed by ring closure, phosphination, and finally metalation. Each step has been optimized to use commercially available starting materials and standard laboratory equipment, reducing the need for specialized high-pressure reactors or cryogenic conditions. The detailed standardized synthesis steps, including precise molar ratios, solvent choices, and temperature profiles, are provided in the guide below to assist R&D teams in replicating these results.
- Synthesize Intermediate A via Pd-catalyzed coupling of o-nitroaniline and o-fluoroiodobenzene at 115-120°C.
- Convert Intermediate A to Intermediate B (benzimidazole derivative) using o-fluorobenzaldehyde and sodium dithionite at 130-135°C.
- React Intermediate B with potassium diphenylphosphide in THF under reflux to generate the bisphosphine Ligand L.
- Complex Ligand L with cuprous halide (CuI, CuCl, or CuBr) in dichloromethane/methanol at room temperature to crystallize the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from precious metal-based emitters to copper-based alternatives represents a strategic opportunity to de-risk the supply chain and optimize manufacturing costs. The volatility of the platinum group metal market has long been a pain point for display manufacturers, often leading to unpredictable budget variances and sourcing delays. By adopting the technology described in CN111848676A, companies can secure a more stable supply of raw materials, as copper and the organic precursors (such as o-nitroaniline and o-fluoroiodobenzene) are produced in vast quantities globally. This shift eliminates the geopolitical risks associated with mining concentrated in specific regions, ensuring a continuous flow of materials necessary for uninterrupted production lines.
- Cost Reduction in Manufacturing: The substitution of iridium with copper results in a drastic reduction in raw material expenditure, as copper is orders of magnitude cheaper and more abundant than precious metals. Beyond the metal cost itself, the simplified synthesis route operates under milder conditions, particularly the final complexation step which occurs at room temperature, significantly lowering energy consumption compared to high-temperature processes required for other emitters. The elimination of expensive heavy metal scavengers and the reduced need for ultra-pure solvent systems further contribute to substantial cost savings in the overall production budget, enhancing the profit margin for OLED panel manufacturers.
- Enhanced Supply Chain Reliability: The precursors required for this synthesis, including various fluorinated benzenes and phosphine derivatives, are commodity chemicals with established global supply networks. This abundance ensures that lead times for raw material delivery are minimized, allowing for just-in-time inventory management strategies that reduce warehousing costs. Furthermore, the robustness of the synthetic pathway means that yield fluctuations are minimal, providing predictable output volumes that facilitate accurate demand forecasting and capacity planning for large-scale display production facilities.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with reaction conditions that are easily transferable from gram-scale laboratory synthesis to multi-kilogram pilot plant operations. The use of standard organic solvents like toluene, THF, and dichloromethane allows for the utilization of existing solvent recovery infrastructure, minimizing waste generation and aligning with strict environmental regulations. The absence of highly toxic heavy metals in the final waste stream simplifies effluent treatment processes, reducing the environmental compliance burden and associated disposal fees for chemical manufacturing plants.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these benzimidazole bisphosphine copper complexes in OLED workflows. These answers are derived directly from the experimental data and characterization results presented in the patent documentation, providing a reliable basis for feasibility assessments.
Q: What is the photoluminescence quantum efficiency of these copper complexes?
A: According to patent data, the solid powder of the bromide variant (C43H32CuBrN2P2) achieves a maximum quantum efficiency of 71.53% at 293K, with a lifetime up to 61.08μs.
Q: Why are copper complexes preferred over iridium for OLEDs?
A: Copper is earth-abundant and significantly cheaper than scarce transition metals like Iridium or Platinum, offering a sustainable path for mass production without compromising emission efficiency.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the process utilizes mild reaction conditions (20-35°C for the final step) and standard solvents like toluene and THF, facilitating straightforward scale-up from lab to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Luminescent Copper Compound Supplier
As the demand for high-efficiency, cost-effective OLED materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis capabilities and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that the transition from lab-scale discovery to mass manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the structural integrity and photophysical properties of every batch, ensuring consistent performance in your final display devices.
We invite you to engage with our technical procurement team to discuss how these innovative copper complexes can be integrated into your specific product roadmap. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this earth-abundant emitter technology. Please contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, and let us help you drive the next generation of sustainable display innovation.
