Revolutionizing Aromatic Heterocyclic Formate Production with Metal-Free Organocatalysis
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective manufacturing processes. Patent CN110292948B introduces a groundbreaking methodology for the synthesis of aromatic heterocyclic formate compounds, utilizing a novel imines-functionalized imidazole chloride salt as an organocatalyst. This technology represents a paradigm shift away from the reliance on precious transition metals, offering a robust pathway for producing critical pharmaceutical and agrochemical intermediates. By leveraging carbon dioxide as a C1 source under mild conditions, this invention addresses long-standing challenges in heterocyclic functionalization, providing a reliable aromatic heterocyclic formate supplier with a distinct competitive edge in green chemistry. The technical implications of this patent extend far beyond simple yield improvements, fundamentally altering the economic and safety profiles of large-scale carboxylation reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the carboxylation of aromatic heterocyclic compounds to form valuable formate esters has been heavily dependent on transition metal catalysis, specifically utilizing gold(I) or copper(I) N-heterocyclic carbene complexes. These conventional methodologies suffer from severe inherent drawbacks that hinder their scalability and commercial viability. Firstly, the reliance on gold catalysts imposes a prohibitive cost burden on the manufacturing process, making the final intermediates economically unfeasible for high-volume production. Secondly, copper-based systems, while slightly cheaper, introduce significant toxicity concerns that necessitate rigorous and expensive purification steps to meet stringent pharmaceutical purity specifications. Furthermore, prior art techniques typically require elevated pressures of carbon dioxide, often around 1.4 atmospheres or higher, which mandates the use of specialized pressure-rated reactors, thereby increasing capital expenditure and operational complexity. Additionally, these metal-catalyzed reactions often demand high temperatures exceeding 100°C to achieve acceptable conversion rates, leading to energy inefficiency and potential thermal degradation of sensitive substrates.
The Novel Approach
In stark contrast to the limitations of metal-dependent pathways, the novel approach disclosed in the patent utilizes an imines-functionalized imidazole chloride salt to catalyze the reaction under remarkably mild conditions. This organocatalytic system operates effectively at normal atmospheric pressure, completely eliminating the safety risks and equipment costs associated with pressurized gas handling. The reaction temperatures are significantly reduced to a range of 50-85°C, with optimal performance observed at 80°C, which drastically lowers energy consumption compared to the >100°C requirements of previous methods. This method not only matches but often exceeds the catalytic activity of metal complexes, achieving gas-phase yields up to 95% for benzoxazole substrates. Crucially, this technology expands the substrate scope to include challenging molecules like benzothiazoles and benzimidazoles, which were previously difficult to functionalize efficiently. 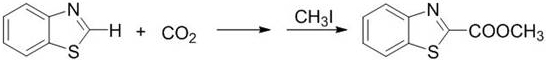
Mechanistic Insights into Imidazole-Catalyzed Carboxylation
The core of this technological advancement lies in the unique electronic properties of the imines-functionalized imidazole chloride salt, which acts as a potent nucleophilic catalyst. The mechanism involves the activation of the aromatic heterocyclic substrate through a transient interaction with the imidazolium species, facilitating the insertion of carbon dioxide into the C-H bond. Unlike metal centers that rely on d-orbital interactions, this organocatalyst leverages steric and electronic tuning provided by the bulky 2,6-di-isopropylphenyl groups on the imine functionality to stabilize the transition state. This stabilization allows the reaction to proceed with high selectivity and efficiency even at lower temperatures. The presence of the imine moiety enhances the Lewis basicity of the catalyst, promoting the deprotonation of the heterocycle by the base (potassium tert-butoxide or cesium carbonate) to generate the reactive nucleophile in situ. This mechanistic pathway avoids the formation of metal-carbene intermediates, thereby sidestepping the issues of metal leaching and contamination that plague traditional synthesis routes.
From an impurity control perspective, the metal-free nature of this catalytic cycle offers profound advantages for downstream processing. In conventional metal-catalyzed reactions, trace amounts of gold or copper can persist in the final product, requiring complex scavenging treatments that reduce overall yield and increase waste. The organocatalytic approach ensures that the primary impurities are organic in nature and can be easily removed through standard extraction and chromatography techniques. The patent data indicates that simple column chromatography using ethyl acetate and petroleum ether is sufficient to achieve high purity, as evidenced by the clean NMR spectra of the products. This simplifies the purification workflow significantly, reducing the number of unit operations required to bring the material to specification. The robustness of the catalyst also means that side reactions such as polymerization or over-alkylation are minimized, resulting in a cleaner crude reaction profile that facilitates easier isolation of the target aromatic heterocyclic formate.
How to Synthesize Aromatic Heterocyclic Formate Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for implementing this technology in a laboratory or pilot plant setting. The process begins with the preparation of the reaction mixture under an inert atmosphere, ensuring that moisture and oxygen do not interfere with the sensitive catalytic species. The sequential addition of the catalyst, base, solvent, and substrate is critical to maintaining the homogeneity of the reaction mixture, particularly when using polar aprotic solvents like DMF. Once the carbon dioxide is introduced, the system is maintained at a controlled temperature to drive the carboxylation to completion before proceeding to the esterification step. 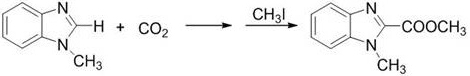
- Prepare the reaction mixture by sequentially adding the imidazole catalyst (1-5 mol%), alkali base (1.0-1.5 equiv), solvent (DMF), and the aromatic heterocyclic substrate under an inert gas atmosphere.
- Introduce carbon dioxide gas into the system and stir the reaction at 50-85°C under normal atmospheric pressure for 12-24 hours to complete the carboxylation.
- Cool the mixture, add a halogenated hydrocarbon (e.g., methyl iodide) for esterification at 45-75°C, then quench with water, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this imidazole-catalyzed process translates into tangible strategic benefits that directly impact the bottom line and operational resilience. The elimination of precious metal catalysts removes a major source of price volatility and supply risk, as the organocatalyst can be synthesized from readily available commodity chemicals. This shift significantly reduces the raw material cost basis for the final intermediate, allowing for more competitive pricing in the global market without sacrificing quality. Furthermore, the ability to operate at atmospheric pressure simplifies the engineering requirements for production facilities, enabling the use of standard glass-lined or stainless steel reactors rather than expensive autoclaves. This flexibility enhances the agility of the supply chain, allowing for faster scale-up and the ability to utilize a broader range of manufacturing partners.
- Cost Reduction in Manufacturing: The removal of expensive gold and copper catalysts results in substantial cost savings, as the organocatalyst is inexpensive to produce and stable in air. The process eliminates the need for costly metal scavengers and extensive purification steps required to remove trace heavy metals, further driving down the cost of goods sold. Additionally, the lower reaction temperatures reduce energy consumption for heating and cooling, contributing to overall operational efficiency. The high yields achieved, often exceeding 90% for key substrates, minimize raw material waste and maximize the throughput of existing production assets.
- Enhanced Supply Chain Reliability: By relying on a metal-free catalyst system, the supply chain becomes less vulnerable to geopolitical fluctuations in the availability of precious metals. The simplicity of the reaction conditions allows for production in a wider variety of facilities, reducing the risk of bottlenecks caused by specialized equipment shortages. The stability of the catalyst in air simplifies storage and handling logistics, reducing the need for specialized containment or inert atmosphere shipping. This robustness ensures consistent supply continuity even in challenging logistical environments, securing the flow of critical intermediates to downstream customers.
- Scalability and Environmental Compliance: The use of carbon dioxide as a feedstock aligns with global sustainability goals, utilizing a greenhouse gas as a valuable resource rather than a waste product. The absence of toxic heavy metals simplifies wastewater treatment and waste disposal, ensuring compliance with increasingly stringent environmental regulations. The process generates minimal hazardous waste, reducing the environmental footprint of the manufacturing operation. The mild conditions and high selectivity make the process inherently safer, reducing the risk of accidents and ensuring a sustainable long-term production capability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel carboxylation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and reliability. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing portfolios. The information covers catalyst loading, reaction conditions, and product isolation strategies to provide a comprehensive overview of the process capabilities.
Q: What are the primary advantages of using imidazole chloride salt over traditional metal catalysts?
A: Unlike traditional gold or copper catalysts which are expensive and toxic, the imidazole chloride salt is metal-free, environmentally benign, air-stable, and significantly lower in cost, facilitating large-scale industrial application.
Q: Does this process require high-pressure equipment for carbon dioxide fixation?
A: No, a key breakthrough of this patent is the ability to perform the carboxylation reaction effectively under normal atmospheric pressure, eliminating the need for specialized high-pressure reactors and enhancing operational safety.
Q: What is the substrate scope for this carboxylation method?
A: The method demonstrates excellent universality, successfully catalyzing benzoxazoles, benzothiazoles, and even difficult-to-react benzimidazoles, achieving high yields where prior art methods often failed or required harsh conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Heterocyclic Formate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the imidazole-catalyzed carboxylation technology disclosed in CN110292948B and are fully equipped to leverage it for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facility is outfitted with state-of-the-art rigorous QC labs capable of verifying the stringent purity specifications required for pharmaceutical intermediates, guaranteeing that every batch meets the highest international standards. We understand that consistency and quality are paramount, and our team is dedicated to optimizing this metal-free route to deliver maximum value.
We invite forward-thinking organizations to collaborate with us to unlock the full commercial potential of this sustainable synthesis method. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Together, we can drive innovation in the fine chemical sector while achieving significant economic and environmental benefits through the adoption of this advanced organocatalytic technology.
