Scalable Production of High-Purity Ornithine Phytosterol Ester Hydrochloride for Functional Foods
Introduction to Advanced Nutritional Intermediate Synthesis
The development of functional food ingredients often hinges on overcoming inherent physicochemical limitations such as poor solubility and low bioavailability. Patent CN108912201B introduces a groundbreaking preparation method for ornithine phytosterol ester hydrochloride, a novel derivative designed to synergize the health benefits of ornithine and phytosterols. This innovation addresses the critical challenge of phytosterol insolubility by chemically bonding it with ornithine, a water-soluble amino acid known for its role in the urea cycle and liver health. The resulting compound not only exhibits enhanced solubility in both aqueous and lipid systems but also maintains the physiological efficacy of its parent molecules. For manufacturers seeking a reliable nutritional ingredients supplier, this technology represents a significant leap forward in creating high-value additives for the food, pharmaceutical, and cosmetic industries. The process is characterized by mild reaction conditions and a streamlined workflow, making it highly attractive for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies to improve phytosterol solubility have often relied on physical modifications like microencapsulation or simple esterification with fatty acids such as lauric acid. However, these conventional methods frequently suffer from significant drawbacks including high energy consumption, cumbersome multi-step procedures, and uncertain safety profiles regarding residual solvents or reagents. Furthermore, simple physical mixing does not fundamentally alter the chemical nature of the sterol, often resulting in unstable dispersions that separate over time. The lack of a covalent bond means the bioavailability improvement is marginal compared to chemical derivatization. Additionally, many existing chemical methods require harsh conditions that can degrade sensitive functional groups or lead to racemization of chiral amino acids, rendering the final product unsuitable for high-end pharmaceutical or premium food applications. These inefficiencies create bottlenecks in cost reduction in food additive manufacturing and limit the versatility of the final ingredient.
The Novel Approach
The patented methodology offers a sophisticated solution by employing a specific catalytic system to drive the esterification between protected ornithine and phytosterol. This approach utilizes a composite catalyst system comprising DCC, HOBT, and DMAP, which operates effectively at mild temperatures around 25 degrees Celsius. Unlike traditional high-energy processes, this method ensures high conversion rates exceeding 89.99 percent while maintaining the structural integrity of the molecules. The use of protective groups (Boc) during the reaction prevents side reactions, and a subsequent deprotection step yields the pure hydrochloride salt. This two-step strategy allows for precise control over the reaction pathway, minimizing impurities and eliminating the need for extensive downstream purification. 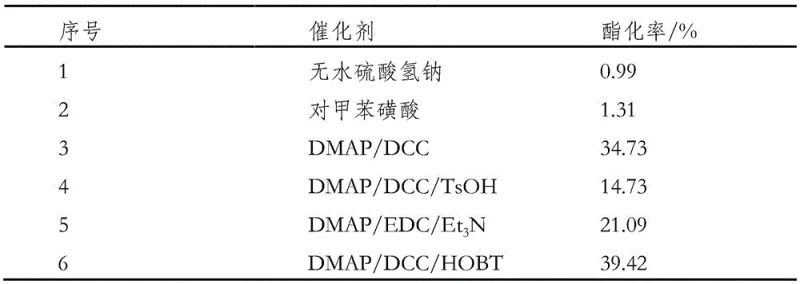 By integrating these advanced catalytic techniques, the process achieves a final product purity of over 99.37 percent, setting a new benchmark for quality in the production of complex nutritional intermediates.
By integrating these advanced catalytic techniques, the process achieves a final product purity of over 99.37 percent, setting a new benchmark for quality in the production of complex nutritional intermediates.
Mechanistic Insights into DCC/HOBT/DMAP Catalyzed Esterification
The core of this synthesis lies in the synergistic action of the ternary catalyst system. Dicyclohexylcarbodiimide (DCC) acts as the primary coupling agent, activating the carboxyl group of the protected ornithine to form an O-acylurea intermediate. However, this intermediate is inherently unstable and prone to intramolecular cyclization, which can lead to racemization and the loss of optical purity. This is where 1-Hydroxybenzotriazole (HOBT) plays a critical role; it reacts with the O-acylurea to form a more stable active ester, effectively suppressing racemization and ensuring the L-configuration of the ornithine moiety is preserved. Finally, 4-Dimethylaminopyridine (DMAP) serves as a potent nucleophilic catalyst, accelerating the attack of the phytosterol hydroxyl group on the activated carbonyl carbon. This mechanistic orchestration ensures high efficiency and stereoselectivity, which is paramount for producing high-purity ornithine derivatives that meet stringent regulatory standards for human consumption.
Furthermore, the reaction conditions are meticulously optimized to balance kinetics and thermodynamics. The initial activation step is conducted under ice bath conditions to control the exothermic formation of the active intermediate, preventing thermal degradation. Subsequent esterification proceeds at ambient temperature (25 to 30 degrees Celsius), which is energetically favorable compared to high-temperature reflux methods. The molar ratios of reagents are also critical; a ratio of 1.2:1.2:1.0 for DCC:HOBT:Ornithine derivative has been identified as optimal for maximizing yield. This precise control over stoichiometry minimizes waste and reduces the burden on waste treatment systems. For R&D teams, understanding this mechanism is key to troubleshooting and scaling the process, as deviations in catalyst loading or temperature can significantly impact the esterification rate and the final impurity profile.
How to Synthesize Ornithine Phytosterol Ester Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high yield. The process begins with the activation of the amino acid derivative, followed by the coupling with the sterol, and concludes with deprotection and purification. Each stage demands careful monitoring of reaction progress, typically via TLC or HPLC, to determine the exact endpoint and prevent over-reaction or hydrolysis. The detailed standardized synthesis steps see the guide below, which outlines the specific reagent quantities, solvent choices, and workup procedures necessary for successful execution. Following these protocols ensures that the commercial scale-up of complex nutritional intermediates can be achieved with consistent quality and minimal batch-to-batch variation.
- Activate N-tert-butoxycarbonyl-N'-tert-butoxycarbonyl-L-ornithine using DCC and HOBT in anhydrous organic solvent under ice bath conditions for 1 to 2 hours.
- Add phytosterol and DMAP catalyst, raise temperature to 25 degrees Celsius, and stir for 4 to 8 hours to complete esterification.
- Remove solvent, purify via silica gel column chromatography, and perform deprotection using hydrogen chloride in ethyl acetate to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial advantages that directly address the pain points of procurement and supply chain management in the fine chemical sector. The mild reaction conditions and high selectivity translate into a more robust and predictable manufacturing process, reducing the risk of batch failures and supply disruptions. By eliminating the need for harsh reagents and extreme temperatures, the process also lowers the barrier for equipment requirements, allowing for production in standard stainless steel reactors without specialized high-pressure or high-temperature capabilities. This flexibility enhances supply chain reliability and enables faster response times to market demand fluctuations. Moreover, the high purity of the crude product reduces the load on purification units, shortening the overall production cycle time and increasing throughput capacity.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available organic reagents significantly lower the raw material costs. Furthermore, the high conversion rate minimizes the amount of unreacted starting material that needs to be recovered or disposed of, leading to substantial cost savings in waste management. The simplified purification process, driven by the high selectivity of the catalyst system, reduces solvent consumption and energy usage associated with extensive chromatography or recrystallization steps. These factors collectively contribute to a more economical production model, allowing for competitive pricing in the global market for functional food ingredients.
- Enhanced Supply Chain Reliability: The reagents used in this synthesis, such as DCC, HOBT, and DMAP, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The robustness of the reaction against minor variations in conditions ensures consistent output quality, which is critical for maintaining long-term contracts with major food and pharmaceutical clients. Additionally, the scalability of the process from gram to ton scale has been demonstrated, providing confidence in the ability to meet large-volume orders without compromising on purity or lead time. This reliability is essential for building trust with partners who require just-in-time delivery for their own production schedules.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of dicyclohexylurea (DCU) which can be filtered off easily, and aqueous waste streams that are easier to treat than those containing heavy metals. The use of common organic solvents like dichloromethane and ethyl acetate allows for efficient recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint. This compliance with environmental regulations simplifies the permitting process for new production facilities and reduces the risk of regulatory penalties. The ease of scale-up ensures that increasing production capacity to meet growing market demand can be achieved with minimal capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of ornithine phytosterol ester hydrochloride. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this ingredient into their product portfolios. For further technical specifications or custom synthesis requests, please refer to the contact information provided at the end of this report.
Q: Why is ornithine phytosterol ester hydrochloride superior to free phytosterols?
A: Free phytosterols suffer from poor water and oil solubility, limiting their application. This esterified derivative significantly improves solubility and bioavailability while retaining the physiological functions of both ornithine and phytosterols.
Q: What is the purity level achievable with this synthesis method?
A: The patented process achieves a product purity of over 99.37 percent without the need for complex further separation, ensuring high quality for food and pharmaceutical grade applications.
Q: Does the catalytic system prevent racemization during synthesis?
A: Yes, the inclusion of HOBT in the DCC/HOBT/DMAP catalytic system converts active O-acylurea intermediates into stable species, effectively inhibiting racemization and preserving the L-configuration of ornithine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ornithine Phytosterol Ester Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex laboratory innovations into commercially viable manufacturing processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We understand that reducing lead time for high-purity nutritional ingredients is critical for your product launch timelines. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of ornithine phytosterol ester hydrochloride meets the highest industry standards. We are committed to being a partner that not only supplies chemicals but also provides technical solutions to optimize your formulation.
We invite you to collaborate with us to explore the full potential of this advanced ingredient in your applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact us today to request specific COA data and route feasibility assessments for your next project. Let us help you accelerate your development cycle and bring high-quality functional products to market faster.
