Advanced Visible-Light Regulated Dithienylethylene Fluorescent Molecular Switches for High-Performance Optoelectronics
Advanced Visible-Light Regulated Dithienylethylene Fluorescent Molecular Switches for High-Performance Optoelectronics
The field of optoelectronic materials is undergoing a significant transformation driven by the demand for safer, more efficient, and higher-resolution imaging and storage technologies. Patent CN112300142B introduces a groundbreaking class of dithienylethylene (DTE) fluorescent molecular switches that are regulated and controlled entirely by visible light. Unlike traditional photochromic materials that rely on high-energy ultraviolet (UV) radiation, these novel derivatives utilize an aniline-alkynyl photosensitizing group conjugated with a perylene monoimide (PMI) fluorescent unit. This strategic molecular design not only shifts the response wavelength into the visible spectrum but also enhances thermal stability and fatigue resistance. For R&D directors and procurement specialists in the electronic chemicals sector, this represents a pivotal shift towards materials that offer superior performance without the operational hazards of UV sources.
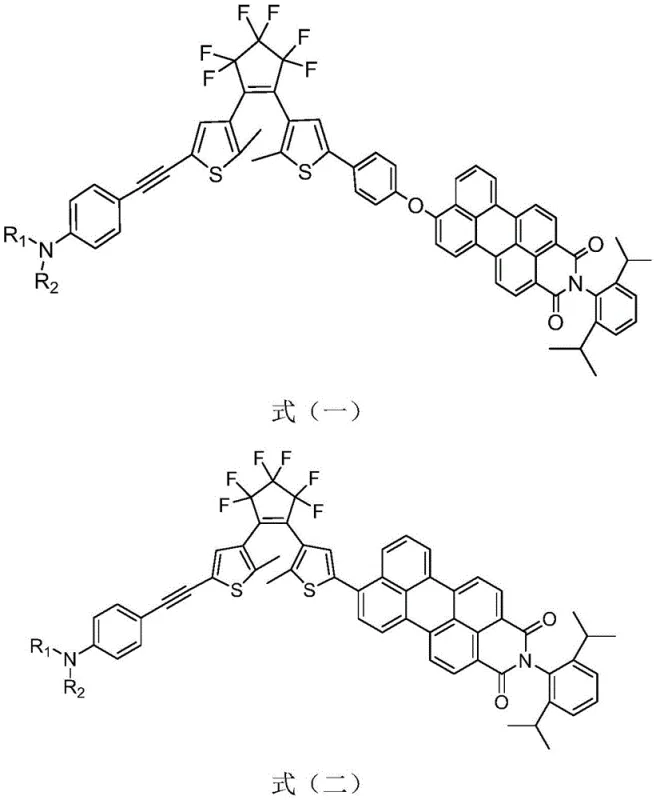
The core innovation lies in the specific structural architecture where the photochromic unit is modified to interact efficiently with visible photons. The patent details compounds represented by general formulas (I) and (II), where the R1 and R2 groups can be varied to fine-tune solubility and electronic properties. This modularity allows for the customization of the material for specific applications ranging from ultra-high-density optical storage to biological super-resolution imaging. By replacing the conventional UV trigger with visible light in the 400-440 nm range, the technology mitigates the risk of photodegradation to biological samples and improves operator safety, making it a highly attractive candidate for next-generation display and sensing applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, diarylethene (DAE) and dithienylethylene (DTE) molecular switches have been constrained by their reliance on ultraviolet light for the ring-closing reaction. While early iterations demonstrated high quantum yields, the requirement for UV excitation poses severe limitations for practical deployment, particularly in biological environments or sensitive polymer matrices. High-energy UV photons can cause irreversible damage to organic tissues and degrade the polymer hosts themselves, leading to poor device longevity. Furthermore, many existing visible-light-responsive switches suffer from slow response times, low photocyclization conversion rates, and inadequate fluorescence quenching efficiency. The inability to achieve a true dark state with high contrast ratios limits their utility in high-precision optical storage where binary data integrity is paramount.
The Novel Approach
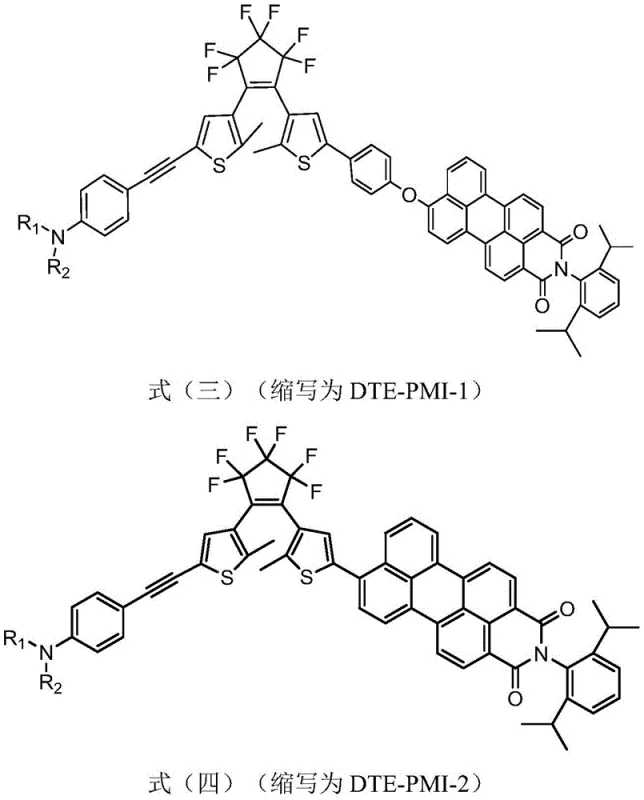
The technology disclosed in CN112300142B overcomes these hurdles through a sophisticated conjugate modification strategy. By introducing an aniline-alkynyl group, the inventors successfully extended the conjugation degree of the visible light-sensitive moiety. This modification facilitates a red-shift in the absorption spectrum, enabling the ring-closing reaction to be triggered by safe, low-energy visible light. Moreover, the patent describes a unique non-conjugated connection mode between the DTE unit and the PMI fluorescent group via an oxygen-bridged bond. This specific linkage preserves the independent performance of both units while maximizing the fluorescence on-off ratio. The result is a material that combines the robustness of perfluorocyclopentene cores with the bright emission of perylene dyes, all controllable within the visible spectrum.
Mechanistic Insights into Aniline-Alkynyl Conjugated Photosensitization
The mechanistic superiority of this system stems from the electronic interplay between the photosensitizer and the photochromic switch. In traditional systems, the energy transfer between the antenna and the switch is often inefficient due to mismatched energy levels or poor orbital overlap. In this invention, the triple bond of the alkynyl group acts as a rigid conduit for electron delocalization, effectively coupling the aniline donor with the dithienylethylene acceptor. This extended pi-system lowers the energy gap required for excitation, allowing 405 nm photons to drive the isomerization process with high efficiency. The patent data indicates that this conjugated approach yields significantly better photochromic response compared to non-conjugated analogues, ensuring rapid switching kinetics essential for real-time imaging applications.
Furthermore, the choice of the perylene monoimide (PMI) as the fluorescent reporter is critical for achieving high contrast. PMI is known for its high fluorescence quantum yield and photostability. When covalently linked to the DTE switch, the open-ring form of the DTE allows the PMI to fluoresce brightly. Upon visible light irradiation, the DTE undergoes cyclization to its closed-ring form, which possesses an absorption band that overlaps with the emission band of the PMI. This spectral overlap triggers a Fluorescence Resonance Energy Transfer (FRET) process or direct electron transfer, effectively quenching the PMI fluorescence. The patent highlights that this quenching mechanism is highly efficient, with some embodiments achieving quenching rates exceeding 98%, thereby creating a distinct binary signal for optical data processing.
How to Synthesize Dithienylethylene Derivatives Efficiently
The synthesis of these advanced molecular switches involves a convergent strategy that ensures high purity and scalability. The process begins with the preparation of a brominated dithienylethylene core, followed by the sequential attachment of the photosensitizer and the fluorophore using palladium-catalyzed cross-coupling reactions. This modular approach allows for the easy variation of substituents to optimize material properties. The detailed standardized synthetic steps, including specific reagent ratios, temperature controls, and purification methods, are outlined in the guide below.
- Preparation of the brominated dithienylethylene core (Br-DTE-Br) via lithiation and substitution reactions starting from 3,5-dibromo-2-methylthiophene.
- Mono-substitution of the Br-DTE-Br core with an aniline-alkynyl compound using Pd(PPh3)2Cl2 and CuI catalysts to form the photosensitized intermediate.
- Final coupling of the intermediate with a perylene monoimide borate ester via Suzuki coupling to yield the target fluorescent molecular switch.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light regulated technology offers substantial strategic advantages beyond mere technical performance. The shift away from UV-dependent chemistry simplifies the handling and storage requirements of the final products, reducing the need for specialized UV-shielding packaging and safety protocols. Additionally, the enhanced fatigue resistance of these materials translates directly into longer product lifecycles for end-users, reducing warranty claims and enhancing brand reputation for durability. The synthetic route utilizes commercially available starting materials and standard catalytic systems, which supports a stable and scalable supply chain capable of meeting industrial demand without relying on exotic or restricted precursors.
- Cost Reduction in Manufacturing: The synthetic pathway described in the patent relies on robust palladium-catalyzed coupling reactions, such as Suzuki and Sonogashira couplings, which are well-established in industrial organic synthesis. By avoiding the need for complex multi-step protections or harsh conditions that degrade yield, the overall process efficiency is significantly improved. The elimination of expensive UV-specific stabilization additives further reduces the Bill of Materials (BOM) cost. Moreover, the high conversion rates and selectivity of the reactions minimize the formation of difficult-to-separate impurities, lowering the cost associated with downstream purification and waste treatment.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including thiophene derivatives, aniline compounds, and perylene precursors, are produced by a wide network of global chemical suppliers. This diversity in the supply base mitigates the risk of single-source bottlenecks. The robustness of the chemical bonds formed during synthesis ensures that the final product has a long shelf life, reducing inventory write-offs due to degradation. This stability allows for larger batch production runs and more flexible logistics planning, ensuring consistent availability for downstream manufacturers of optical devices and imaging systems.
- Scalability and Environmental Compliance: The preparation method operates under relatively mild conditions, often at temperatures below 100°C and using common solvents like toluene and tetrahydrofuran. This reduces the energy footprint of the manufacturing process compared to high-temperature alternatives. Furthermore, the high atom economy of the coupling reactions and the ability to recycle catalysts contribute to a greener manufacturing profile. The absence of toxic UV-generating components in the final application aligns with increasingly stringent environmental and safety regulations regarding consumer electronics and biomedical devices, facilitating easier market entry and regulatory approval.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these visible-light fluorescent switches. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the material's capabilities and limitations.
Q: What is the primary advantage of the aniline-alkynyl modification in these molecular switches?
A: The introduction of the aniline-alkynyl group extends the pi-conjugation system, which significantly red-shifts the absorption wavelength from the ultraviolet region to the visible light region (400-440 nm). This allows for full visible-light regulation, avoiding the sample damage and safety hazards associated with UV irradiation.
Q: How does the fatigue resistance of these new derivatives compare to traditional DTE switches?
A: The novel derivatives exhibit superior fatigue resistance, maintaining stable fluorescence switching performance over multiple cycles. For instance, specific embodiments showed less than 5% fluorescence loss after ten alternating irradiation cycles, ensuring long-term reliability for optical storage applications.
Q: Can these materials be used for super-resolution imaging?
A: Yes, the star-shaped triplet structures (e.g., 3DTE-PMI) described in the patent demonstrate exceptional performance in super-resolution imaging. They achieve a fluorescence on-off ratio as high as 659:1 and enable reconstructed imaging with resolutions down to 24 nm, breaking the diffraction limit of conventional optical microscopy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dithienylethylene Derivative Supplier
As the demand for high-performance optoelectronic materials continues to surge, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and stringent purity specifications guarantee that every batch of dithienylethylene derivative meets the exacting standards required for advanced applications like super-resolution imaging and optical data storage.
We invite you to collaborate with our technical team to explore how this visible-light regulated technology can enhance your product portfolio. Whether you require a Customized Cost-Saving Analysis for your specific formulation or need to evaluate the feasibility of large-scale production, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the rapidly evolving electronic materials market.
