Advanced D-A Type Photochromic Materials: Scalable Synthesis for Next-Gen Optical Switches
The landscape of smart optical materials is undergoing a significant transformation with the emergence of advanced Donor-Acceptor (D-A) type photochromic systems, as detailed in the groundbreaking patent CN116375722A. This technology addresses the longstanding limitations of traditional spiropyran derivatives, which often suffer from poor reversibility and negligible activity in the solid state due to steric hindrance and aggregation. By strategically constructing a methyl-substituted spiropyran molecular skeleton and subsequently coupling diverse aromatic conjugated units via robust Suzuki chemistry, this invention achieves a remarkable extension of molecular conjugation length. This structural modification effectively regulates the HOMO/LUMO energy levels and band gaps, resulting in materials that exhibit rapid open-close ring rates and distinct color changes even in pure solid films. The ability to tune these properties through simple variation of the boronic acid coupling partner offers unprecedented flexibility for applications ranging from camouflage coatings to high-density optical data storage devices.
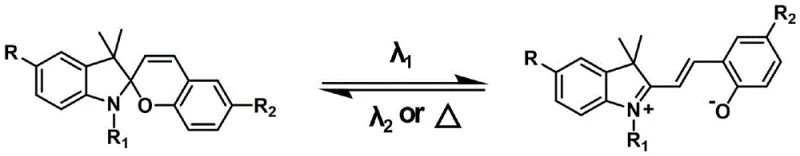
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of spiropyran-based photochromic materials has been constrained by their inherent tendency to aggregate in solid matrices, which severely inhibits the conformational changes required for the photochromic process. Conventional methods typically rely on simple functional group modifications or physical blending with polymer matrices, which yield only marginal improvements in fatigue resistance and switching speed. In many cases, the rigid environment of a solid film prevents the spiro-carbon bond from breaking efficiently, leading to materials that are only active in dilute solution states. Furthermore, traditional synthetic routes often lack modularity, requiring entirely new pathways to achieve different absorption wavelengths, which complicates the supply chain and increases the cost of goods for specialized optical applications. These factors have collectively hindered the widespread commercial adoption of spiropyrans in high-performance solid-state devices where reliability and consistency are paramount.
The Novel Approach
The innovative methodology presented in this patent overcomes these barriers by introducing bulky aromatic conjugated units directly onto the spiropyran backbone through a late-stage palladium-catalyzed cross-coupling reaction. This D-A architectural strategy not only extends the pi-electron delocalization to redshift the absorption spectrum but also physically increases the free volume around the photoactive center. By selecting specific heterocyclic units such as thiophene, pyridine, or benzofuran, manufacturers can precisely engineer the intramolecular charge transfer (ICT) characteristics to enhance both the color contrast and the thermal stability of the open-ring merocyanine form. This approach transforms the spiropyran from a solution-phase curiosity into a robust solid-state material capable of withstanding repeated cycling without significant degradation. The modularity of this synthesis allows for a single common intermediate to be diversified into a library of high-performance materials, streamlining the production process and reducing inventory complexity for industrial buyers.
Mechanistic Insights into Suzuki-Miyaura Functionalization of Spiropyrans
The core of this technological advancement lies in the precise construction of the D-A conjugated system, which fundamentally alters the electronic landscape of the spiropyran molecule. The mechanism begins with the formation of a stable brominated spiropyran intermediate (SP-Br), which serves as an electrophilic handle for subsequent transition metal catalysis. When subjected to Suzuki coupling conditions, the carbon-bromine bond undergoes oxidative addition with the palladium catalyst, followed by transmetallation with the organoboron species and reductive elimination to forge the new carbon-carbon bond. This process effectively links the electron-rich or electron-deficient aromatic unit to the spiropyran core, creating a push-pull electronic system that lowers the energy barrier for the photo-induced ring-opening reaction. The resulting extended conjugation stabilizes the zwitterionic merocyanine form, leading to the observed bathochromic shift and enhanced color intensity upon UV irradiation.
Furthermore, the steric bulk introduced by these conjugated appendages plays a critical mechanistic role in suppressing non-radiative decay pathways that typically plague solid-state photochromics. In the absence of these groups, close packing of molecules in the solid state restricts the necessary planarization of the merocyanine form, effectively locking the molecule in its closed, colorless state. The new D-A structures disrupt this crystalline packing, creating nanoscopic voids or free volume that accommodate the significant geometric rearrangement from the sp3-hybridized spiro-carbon to the planar sp2-hybridized open form. This steric engineering ensures that the photoisomerization kinetics remain fast and reversible, even in the absence of solvent plasticization. Consequently, the material exhibits superior fatigue resistance, as the reduced intermolecular friction minimizes the mechanical stress on the molecular framework during repeated switching cycles.
How to Synthesize D-A Type Photochromic Spiropyrans Efficiently
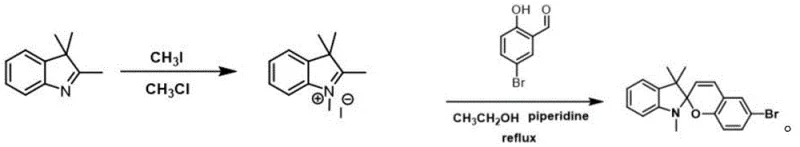
The synthesis of these high-value optical materials follows a streamlined three-step protocol that balances high yield with operational simplicity, making it highly attractive for commercial scale-up. The process initiates with the quaternization of a commercially available indole derivative, followed by a condensation reaction to establish the core spiropyran scaffold bearing a reactive bromine handle. The final and most critical step involves the palladium-catalyzed coupling with various boronic acids, a reaction known for its tolerance to functional groups and mild conditions. Detailed standardized synthetic steps for the preparation of the key intermediate and final derivatives are provided in the guide below, ensuring reproducibility and quality control for manufacturing teams.
- Quaternization of 2,3,3-trimethylindole with methyl iodide in chloroform under reflux to form the indolium salt intermediate.
- Condensation of the indolium salt with 5-bromosalicylaldehyde in ethanol using piperidine as a base to construct the brominated spiropyran backbone (SP-Br).
- Suzuki-Miyaura coupling of SP-Br with various aryl or heteroaryl boronic acids using Pd(PPh3)4 catalyst to introduce conjugated donor-acceptor units.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented methodology offers substantial strategic advantages by decoupling the complexity of the core scaffold synthesis from the diversification of the final product properties. The ability to utilize a single, common intermediate (SP-Br) to generate multiple distinct end-products means that manufacturers can maintain a leaner inventory of bulk precursors while responding rapidly to specific customer requirements for different absorption wavelengths. This modularity significantly reduces the working capital tied up in raw materials and minimizes the risk of obsolescence for specialized SKUs. Additionally, the reliance on well-established Suzuki coupling chemistry ensures that the process can be easily transferred to existing multipurpose reactors without the need for exotic equipment or hazardous reagents, thereby lowering the barrier to entry for contract manufacturing organizations.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for complex, multi-step custom syntheses for each new variant, as the diversity is introduced in the final step using readily available commodity boronic acids. This convergence strategy drastically simplifies the supply chain logistics and reduces the overall cost of goods sold by maximizing the utility of the expensive spiropyran intermediate. Furthermore, the high selectivity of the palladium-catalyzed reaction minimizes the formation of difficult-to-remove byproducts, reducing the burden on downstream purification processes like chromatography and crystallization. These efficiencies translate directly into margin improvements for suppliers and more competitive pricing for downstream integrators in the optical materials sector.
- Enhanced Supply Chain Reliability: The starting materials for this process, including substituted indoles, salicylaldehydes, and various boronic acids, are widely produced commodities with robust global supply chains. This availability mitigates the risk of supply disruptions that often plague specialty chemical manufacturing reliant on obscure or single-source reagents. The use of standard solvents such as ethanol, THF, and dioxane further ensures that production can be sustained across different geographic regions without logistical bottlenecks. For supply chain directors, this translates to predictable lead times and the confidence to commit to long-term supply agreements with major electronics or coating manufacturers who demand consistent quality and volume.
- Scalability and Environmental Compliance: The reaction conditions described, primarily involving reflux in common organic solvents and standard aqueous workups, are inherently scalable from gram to tonne quantities without significant re-engineering. The process avoids the use of highly toxic heavy metals beyond the catalytic amounts of palladium, which can be effectively scavenged and recycled, aligning with increasingly stringent environmental regulations in the fine chemical industry. The solid-state activity of the final products also implies that less material is needed to achieve the desired optical effect in application formulations, contributing to a lower overall environmental footprint. This combination of scalability and compliance makes the technology a sustainable choice for long-term industrial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of these novel D-A type photochromic materials. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the technology's value proposition. Understanding these nuances is essential for R&D teams evaluating the material for specific device architectures and for procurement officers assessing the total cost of ownership.
Q: How does the D-A structure improve solid-state photochromism?
A: The introduction of bulky aromatic conjugated units increases the free volume within the solid matrix, preventing aggregation-caused quenching and allowing the necessary molecular rotation for the ring-opening process to occur even in pure solid films.
Q: What are the key advantages of using Suzuki coupling for these materials?
A: Suzuki coupling allows for the modular attachment of diverse electron-donating or withdrawing groups, enabling precise tuning of the HOMO/LUMO energy levels and absorption wavelengths without redesigning the entire synthetic route.
Q: Is the photochromic switching reversible in these new derivatives?
A: Yes, the patent data indicates significantly improved reversibility and faster switching rates compared to traditional spiropyrans, attributed to the optimized electronic push-pull effects within the D-A conjugated system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photochromic Material Supplier
As the demand for intelligent optical materials continues to surge in sectors like wearable electronics and anti-counterfeiting technologies, having a manufacturing partner with deep expertise in complex heterocyclic synthesis is critical. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready product is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of spiropyran derivative meets the exacting standards required for high-performance optoelectronic applications. We understand the nuances of handling air-sensitive intermediates and optimizing palladium removal to meet residual metal limits for electronic grade chemicals.
We invite you to engage with our technical procurement team to discuss how this patented D-A spiropyran technology can be integrated into your product roadmap. By requesting a Customized Cost-Saving Analysis, you can gain visibility into how our optimized synthetic routes can reduce your overall material costs while enhancing product performance. We are prepared to provide specific COA data and route feasibility assessments tailored to your unique volume requirements, ensuring a partnership built on transparency, technical excellence, and mutual growth in the advanced materials marketplace.
