Advanced Synthesis of Photo-Responsive Triphenylethylene Derivatives for Commercial Scale-Up of Complex Functional Materials
The landscape of smart functional materials is undergoing a significant transformation with the advent of multi-responsive organic systems capable of converting mechanical energy into light while maintaining optical switchability. Patent CN115745875A introduces a groundbreaking class of light-controlled organic mechanoluminescent materials containing a triphenylethylene group, which addresses the critical challenge of controllability in stress-sensing applications. This technology leverages a robust Suzuki-Miyaura cross-coupling strategy to integrate aromatic heterocycles onto a triphenylethylene scaffold, resulting in compounds that emit strong fluorescence upon mechanical stimulation yet can be reversibly switched off via ultraviolet irradiation. For R&D directors and procurement specialists in the electronic chemical sector, this represents a pivotal advancement in creating high-purity OLED material precursors and stress sensors that offer both sensitivity and tunability without compromising on synthetic feasibility.
Furthermore, the patent elucidates a synthesis pathway that balances high luminous quantum efficiency with operational simplicity, a rare combination in the field of aggregation-induced emission (AIE) active materials. The ability to restore mechanoluminescent performance through visible light irradiation after UV exposure creates a bistable system ideal for reusable security tags and high-precision damage monitoring devices. By focusing on the modular assembly of these complex structures, the invention provides a clear roadmap for the commercial scale-up of complex functional materials, ensuring that supply chains can meet the growing demand for intelligent responsive polymers and coatings in next-generation optoelectronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing mechanoluminescent materials often suffer from rigid molecular architectures that lack the dynamic responsiveness required for smart sensing applications. Many existing triboluminescent compounds rely on simple crystallization processes that yield materials with fixed emission properties, making it impossible to modulate their output once fabricated. Furthermore, conventional synthetic routes frequently involve multi-step sequences with harsh reaction conditions, leading to low overall yields and difficult purification protocols that generate substantial chemical waste. The inability to effectively control the intermolecular interactions in these legacy materials often results in poor crystal packing, which diminishes the efficiency of mechanical-to-optical energy conversion and limits their practical utility in high-sensitivity stress detection scenarios.
The Novel Approach
In contrast, the methodology outlined in CN115745875A employs a strategic molecular design that couples a triphenylethylene core with specific aromatic heterocycles to create a distorted configuration with a large molecular dipole moment. This structural distortion prevents tight pi-stacking in the ground state, thereby enhancing the mechanoluminescent efficiency upon external stimulation. The novelty lies not just in the emission capability but in the integration of a photochromic switch directly into the mechanoluminescent framework, allowing for external optical control over the material's activity. This dual-functionality is achieved through a streamlined one-pot coupling reaction that significantly simplifies the manufacturing process compared to traditional multi-step syntheses, offering a distinct advantage for cost reduction in electronic chemical manufacturing by minimizing unit operations and solvent usage.

The core innovation is encapsulated in the general structural formula where the triphenylethylene moiety serves as the mechanophore while the appended heterocycle acts as the photo-switchable unit. This modular design allows chemists to fine-tune the electronic properties of the final product by varying the heterocyclic component, providing a versatile platform for developing a wide range of specialized electronic chemicals tailored to specific wavelength requirements or sensitivity thresholds.
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling for Functional Materials
The synthesis relies on the palladium-catalyzed cross-coupling between a halogenated aromatic heterocycle and a boron-functionalized triphenylethylene derivative, a reaction known for its high tolerance of functional groups and mild conditions. The catalytic cycle initiates with the oxidative addition of the aryl halide to the zero-valent palladium species, forming an organopalladium intermediate that is crucial for the subsequent transmetallation step. In this specific application, the use of tetrakis(triphenylphosphine)palladium ensures efficient activation of the carbon-halogen bond even in sterically hindered environments typical of triphenylethylene derivatives. The presence of potassium carbonate as a base facilitates the formation of the reactive boronate species, driving the equilibrium towards the formation of the new carbon-carbon bond that links the heterocycle to the triphenylethylene core.
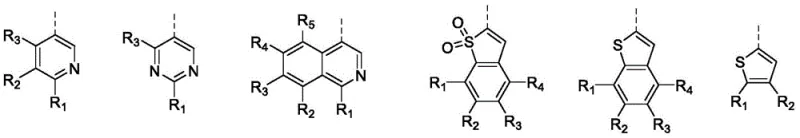
Crucially, the selection of the aromatic heterocycle is not arbitrary; it is engineered to influence the Highest Occupied Molecular Orbital (HOMO) and Lowest Unoccupied Molecular Orbital (LUMO) energy levels of the final molecule. As illustrated by the variety of heterocyclic options such as fluorinated pyridines and thiophenes, the introduction of electronegative heteroatoms creates an electron-deficient environment that stabilizes the excited states necessary for photochromism. This electronic modulation ensures that the molecule can undergo reversible cyclization or conformational changes under UV irradiation without degrading the mechanoluminescent backbone. For quality control teams, understanding this mechanism is vital for impurity profiling, as side reactions like homocoupling of the boronic acid or dehalogenation of the heterocycle must be strictly monitored to maintain the high purity specifications required for optoelectronic applications.
How to Synthesize Triphenylethylene Derivative Efficiently
The patented process offers a reproducible and scalable protocol for generating these advanced materials, utilizing standard laboratory equipment that translates easily to pilot plant operations. The procedure involves dissolving the reactants in tetrahydrofuran, degassing with nitrogen to prevent catalyst oxidation, and heating under reflux to drive the reaction to completion over a 24-hour period. Following the reaction, the workup involves simple filtration and solvent removal, followed by purification via silica gel column chromatography to isolate the target compound with high fidelity. Detailed standardized synthesis steps are provided below to guide process engineers in replicating these results.
- Dissolve the bromo- or iodo-containing aromatic heterocyclic compound and the triphenylethylene compound containing boronic acid or borate ester in a tetrahydrofuran solution.
- Add potassium carbonate as the base and introduce nitrogen gas to stir the mixture at room temperature for approximately 30 minutes to ensure deoxygenation.
- Introduce tetrakis(triphenylphosphine)palladium catalyst, heat the solution to reflux at 85°C for 24 hours, then cool, filter, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers profound benefits regarding raw material availability and process robustness. The starting materials, specifically brominated or iodinated heterocycles and triphenylethylene boronic esters, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This abundance ensures that procurement managers can negotiate favorable terms and maintain consistent inventory levels, which is essential for reducing lead time for high-purity mechanoluminescent compounds in a volatile market. Moreover, the reaction conditions do not require cryogenic temperatures or high-pressure reactors, allowing production to occur in standard glass-lined steel vessels found in most multipurpose chemical manufacturing facilities.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the use of a single catalytic system significantly lower the operational expenditure associated with labor and energy consumption. By avoiding the need for exotic ligands or sensitive organometallic reagents that require strict inert atmosphere handling beyond standard nitrogen blanketing, the process reduces the overhead costs related to specialized equipment and safety protocols. Additionally, the high selectivity of the Suzuki coupling minimizes the formation of difficult-to-separate byproducts, which streamlines the downstream purification process and reduces the volume of silica and solvents required for chromatography, leading to substantial cost savings in waste disposal and material usage.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents such as potassium carbonate and palladium tetrakis ensures that the supply chain is resilient to disruptions often caused by the degradation of sensitive intermediates. The robustness of the reaction means that batch-to-batch variability is minimized, providing reliable electronic chemical supplier partners with the confidence to commit to long-term delivery schedules. This stability is particularly valuable for clients in the display and sensor industries who require consistent material performance to maintain the quality of their end-user devices, thereby strengthening the strategic partnership between the manufacturer and the buyer.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids exothermic hazards associated with rapid addition of reagents, allowing for safe expansion from kilogram to tonne-scale production. The use of tetrahydrofuran as a solvent, while requiring recovery systems, is well-established in the industry with mature recycling technologies that support environmental compliance goals. Furthermore, the solid waste generated is primarily spent silica gel and inorganic salts, which are easier to manage and dispose of compared to the heavy metal sludge often produced by stoichiometric organometallic reactions, aligning with modern green chemistry principles and corporate sustainability mandates.
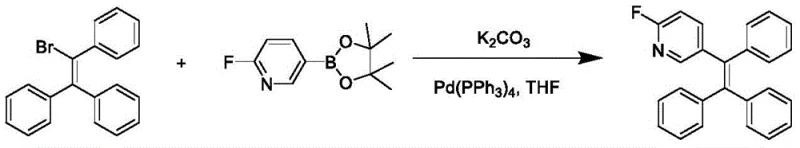
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating the feasibility of integrating these materials into their product lines.
Q: What is the primary advantage of the triphenylethylene derivatives described in CN115745875A?
A: The primary advantage is their dual functionality: they exhibit strong mechanoluminescence under external stress while simultaneously possessing reversible photochromic properties. This allows the material to switch its luminescent state on and off via UV and visible light irradiation, enabling smart sensing applications.
Q: How does the introduction of aromatic heterocycles affect the material's performance?
A: The introduction of heteroatoms in the aromatic heterocyclic groups effectively modulates the aromaticity and the distribution of HOMO and LUMO electron clouds. This structural modification facilitates reversible ring-closing reactions under UV radiation, which is critical for achieving the desired photochromic performance alongside mechanoluminescence.
Q: Is the synthesis process suitable for large-scale production?
A: Yes, the synthesis utilizes standard Suzuki-Miyaura coupling conditions with commercially available reagents like Pd(PPh3)4 and K2CO3 in THF. The process avoids exotic catalysts or extreme pressure conditions, making it highly amenable to commercial scale-up with straightforward purification via column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylethylene Derivative Supplier
As the demand for intelligent responsive materials grows, partnering with an experienced CDMO becomes critical for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from prototype to mass market is seamless and efficient. Our stringent purity specifications and rigorous QC labs guarantee that every batch of triphenylethylene derivative meets the exacting standards required for high-performance optoelectronic applications, minimizing the risk of device failure due to material impurities.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this streamlined manufacturing route. We encourage potential partners to contact us immediately to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for the next generation of smart sensing and display technologies.
