Advanced Manufacturing of 3-Ethoxy-4-Ethoxycarbonyl Phenylacetic Acid for Global Pharma Supply Chains
Introduction to Next-Generation Repaglinide Intermediate Synthesis
The global demand for effective antidiabetic medications continues to surge, placing immense pressure on the supply chains responsible for producing critical active pharmaceutical ingredients (APIs) and their precursors. At the forefront of this chemical evolution is the synthesis of 3-ethoxy-4-ethoxycarbonylphenylacetic acid, a pivotal intermediate in the manufacture of Repaglinide, a premier non-sulfonylurea insulin secretagogue. A groundbreaking patent, identified as CN101891621A, has introduced a transformative compounding method that addresses long-standing inefficiencies in this sector. This innovative approach streamlines the production workflow into a concise two-step sequence, leveraging double alkylation followed by a precise lithiation-carboxylation protocol. By shifting away from hazardous reagents and complex purification requirements, this technology offers a robust pathway for reliable pharmaceutical intermediate supplier networks to enhance both product quality and operational safety. The method achieves a remarkable product purity exceeding 99.4% with a melting point range of 78 to 80 degrees Celsius, setting a new benchmark for quality in the fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key intermediate has been plagued by significant technical and environmental hurdles that hindered efficient commercial scale-up of complex intermediates. Legacy pathways, such as those described in U.S. Pat 6686497B1, relied heavily on solvent systems like tetrahydrofuran (THF) and dimethyl sulfoxide (DMSO) which were difficult to recover, leading to inflated raw material costs and substantial waste generation. Furthermore, other established routes, including those detailed in U.S. Pat 20040249188A1, necessitated the use of highly toxic substances such as sodium cyanide and methyl sulfate. These hazardous reagents not only posed severe safety risks to personnel but also required elaborate waste treatment protocols to neutralize toxic byproducts before disposal. The reliance on chromatographic purification in earlier academic routes further compounded these issues, rendering them impractical for high-volume industrial application due to prohibitive time and cost factors.
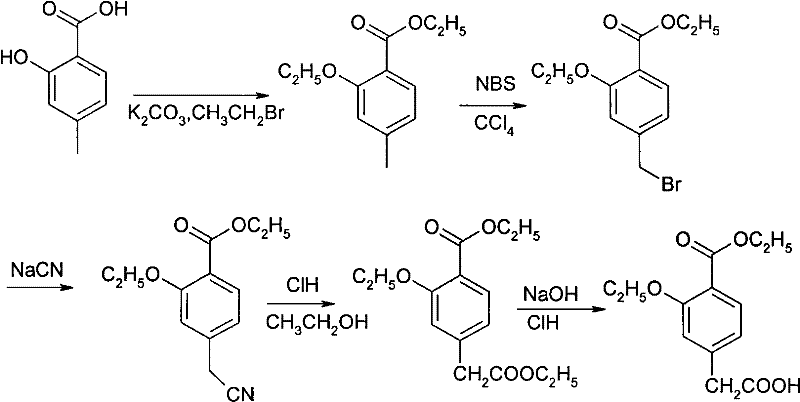
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel process outlined in patent CN101891621A introduces a streamlined and environmentally benign strategy that radically simplifies the manufacturing landscape. This approach initiates with a double alkylation reaction using 4-methyl salicylate and bromoethane in N,N-dimethylformamide, efficiently generating the requisite ester intermediate without the need for isolation. The subsequent step involves a direct dissolution of the crude product into 2-methyl tetrahydrofuran, where it undergoes lithiation with lithium diisopropylamide at cryogenic temperatures followed by carbon dioxide introduction. This elegant sequence bypasses the need for toxic cyanide reagents entirely and eliminates the requirement for energy-intensive chromatographic separation. The result is a highly efficient workflow that utilizes recyclable solvents and common industrial feedstocks, thereby achieving cost reduction in API manufacturing while maintaining exceptional product integrity.
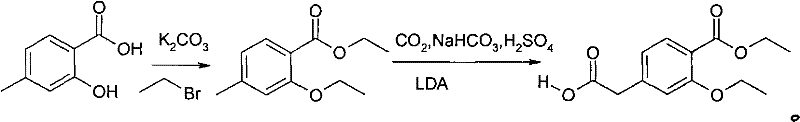
Mechanistic Insights into Lithiation-Carboxylation Chemistry
The core chemical innovation driving this process lies in the precise execution of the lithiation-carboxylation mechanism, which ensures high regioselectivity and yield. The reaction utilizes lithium diisopropylamide (LDA) as a strong, non-nucleophilic base to deprotonate the benzylic position of the 2-ethyoxyl-4-ethyl methylbenzoate intermediate. This deprotonation must be conducted at strictly controlled low temperatures, specifically around -80°C, to prevent side reactions and ensure the formation of the stable benzylic anion. Once the anion is generated, the introduction of carbon dioxide gas facilitates a nucleophilic attack, effectively installing the carboxylic acid functionality at the desired position. This direct carboxylation strategy is superior to older nitrile hydrolysis methods because it avoids the formation of amide byproducts and eliminates the harsh acidic or basic conditions typically required to convert nitriles to acids, thus preserving the integrity of the sensitive ester groups elsewhere in the molecule.
Furthermore, the impurity profile of the final product is meticulously managed through the choice of solvents and reaction conditions. By employing 2-methyl tetrahydrofuran, which offers excellent solubility for organolithium species, the reaction mixture remains homogeneous, promoting consistent kinetics and minimizing localized hot spots that could lead to degradation. The subsequent acidification step using sulfuric acid is carefully controlled to a pH of 2, ensuring complete precipitation of the target acid while keeping inorganic salts in the aqueous phase. This precise control over the workup procedure allows for the removal of residual starting materials and inorganic impurities simply through filtration and washing, obviating the need for complex recrystallization or column chromatography. The resulting white powder exhibits a sharp melting point and high assay value, confirming the efficacy of this mechanistic approach in delivering high-purity repaglinide intermediate suitable for downstream pharmaceutical synthesis.
How to Synthesize 3-Ethoxy-4-Ethoxycarbonyl Phenylacetic Acid Efficiently
Implementing this advanced synthetic route requires strict adherence to temperature controls and reagent stoichiometry to maximize the benefits of the patented methodology. The process begins with the preparation of the alkylated ester, followed immediately by the lithiation step in a moisture-free environment to prevent quenching of the reactive organolithium species. Operators must ensure that the carbon dioxide feed is maintained at optimal pressure and flow rates to drive the carboxylation to completion. The detailed standardized synthesis steps, including specific reagent quantities, stirring rates, and safety protocols for handling pyrophoric reagents like n-butyllithium, are outlined below to guide technical teams in replicating this high-efficiency process.
- Perform a double alkylation reaction using 4-methyl salicylate and bromoethane in N,N-dimethylformamide with potassium carbonate to generate 2-ethyoxyl-4-ethyl methylbenzoate.
- Dissolve the crude intermediate directly into 2-methyl tetrahydrofuran without purification to prepare for the subsequent lithiation step.
- Add lithium diisopropylamide at -80°C, introduce carbon dioxide gas for carboxylation, and finalize with sulfuric acid acidification to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route presents a compelling value proposition centered on risk mitigation and operational efficiency. By eliminating the dependency on highly regulated and toxic reagents such as sodium cyanide, manufacturers can significantly reduce the regulatory burden and insurance costs associated with hazardous material handling. The ability to recycle solvents like N,N-dimethylformamide and 2-methyl tetrahydrofuran repeatedly creates a closed-loop system that minimizes raw material consumption and waste disposal fees. This shift towards a greener chemistry platform not only aligns with increasingly stringent environmental regulations but also stabilizes production costs against fluctuations in raw material pricing. Consequently, this leads to substantial cost savings and a more resilient supply chain capable of withstanding market volatility.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the unit operations and the elimination of expensive purification technologies. Traditional methods often required multiple isolation steps and chromatographic purification, which are labor-intensive and consume vast amounts of silica and solvents. In contrast, this new method achieves high purity through simple crystallization and filtration, drastically reducing the operational expenditure per kilogram of product. Additionally, the use of common industrial raw materials like bromoethane and 4-methyl salicylate ensures a stable and cost-effective supply base, avoiding the premiums associated with specialty reagents.
- Enhanced Supply Chain Reliability: From a logistics perspective, removing toxic cyanide salts from the bill of materials simplifies transportation and storage requirements. Hazardous chemicals often require specialized shipping containers and dedicated storage facilities, which can introduce bottlenecks and delays in the supply chain. By substituting these with safer alternatives, the lead time for high-purity intermediates can be effectively reduced, ensuring a continuous flow of materials to downstream API manufacturers. This reliability is crucial for meeting the rigorous delivery schedules demanded by global pharmaceutical clients who operate on just-in-time inventory models.
- Scalability and Environmental Compliance: The robustness of this synthetic route makes it ideally suited for scaling from pilot plant to full commercial production. The reaction conditions are straightforward and do not require exotic equipment, allowing for easy integration into existing manufacturing infrastructure. Furthermore, the reduced generation of hazardous waste simplifies compliance with environmental protection standards, lowering the risk of regulatory fines or shutdowns. This environmental friendliness enhances the corporate social responsibility profile of the manufacturer, making it a preferred partner for sustainability-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms historical precedents. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines.
Q: How does this novel synthesis route improve upon traditional methods involving sodium cyanide?
A: Traditional routes often rely on highly toxic reagents like sodium cyanide and complex multi-step sequences involving nitrile hydrolysis. This patented method eliminates the need for cyanide entirely by utilizing a direct lithiation-carboxylation strategy, significantly enhancing operational safety and reducing hazardous waste disposal costs.
Q: What represents the key advantage regarding solvent usage in this process?
A: Unlike previous methodologies that utilized non-recyclable solvent systems like DMSO or required chromatographic purification, this process employs N,N-dimethylformamide and 2-methyl tetrahydrofuran, both of which can be recovered and reused repeatedly. This circular solvent usage drastically lowers raw material consumption and environmental impact.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the process is specifically designed for industrial scalability. It utilizes common industrial raw materials, avoids tedious purification steps like column chromatography, and achieves a total recovery of over 61% with high purity (99.4%), making it economically viable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Ethoxy-4-Ethoxycarbonyl Phenylacetic Acid Supplier
As the pharmaceutical industry evolves towards more sustainable and efficient manufacturing practices, partnering with a technically proficient CDMO is essential for success. NINGBO INNO PHARMCHEM stands at the forefront of this transformation, leveraging deep expertise in process chemistry to deliver superior intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-ethoxy-4-ethoxycarbonylphenylacetic acid meets the exacting standards required for antidiabetic drug synthesis.
We invite global partners to collaborate with us to optimize their supply chains and reduce time-to-market for critical therapies. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how our advanced processes can lower your overall cost of goods. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, taking the first step towards a more efficient and reliable partnership.
