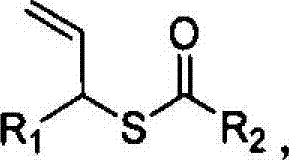
Advanced Iridium-Catalyzed Synthesis of Chiral Allyl Thiocarboxylates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing chiral sulfur-containing motifs, which are prevalent in bioactive molecules ranging from antibiotics to agrochemicals. Patent CN103896816A introduces a groundbreaking approach for the preparation of chiral allyl thiocarboxylates, utilizing a highly efficient iridium-catalyzed allylic substitution reaction. This technology addresses the longstanding challenge of sulfur-induced catalyst poisoning that has historically plagued transition metal-catalyzed thiol functionalizations. By employing a specific iridium complex generated in situ from [Ir(COD)Cl]2 and chiral ligands, the process achieves exceptional levels of stereocontrol under remarkably mild conditions. The versatility of this method allows for a broad substrate scope, accommodating various alkyl and aryl groups, thereby positioning it as a critical tool for the synthesis of high-value pharmaceutical intermediates. For procurement and R&D teams, understanding the mechanistic advantages of this patent is key to securing a reliable supply chain for complex sulfur-based building blocks.
Historically, the synthesis of optically active sulfur compounds relied heavily on asymmetric sulfur-Michael additions or stoichiometric chiral auxiliary methods, which often suffered from significant limitations in terms of atom economy and operational complexity. Conventional transition metal-catalyzed allylation reactions were frequently hindered by the strong coordination of sulfur nucleophiles to the metal center, leading to catalyst deactivation and poor turnover numbers. This necessitated the use of excessive catalyst loading or harsh reaction conditions that were incompatible with sensitive functional groups. In contrast, the novel approach detailed in the patent data utilizes a specialized iridium catalytic system that resists sulfur poisoning while maintaining high reactivity. This breakthrough enables the direct coupling of potassium thiocarboxylates with allyl carbonate compounds without the need for protective group strategies or extreme temperatures. The shift from traditional stoichiometric methods to this catalytic cycle represents a paradigm shift in process chemistry, offering a more sustainable and economically viable pathway for industrial production.
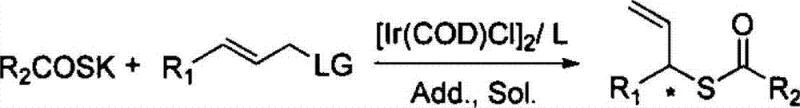
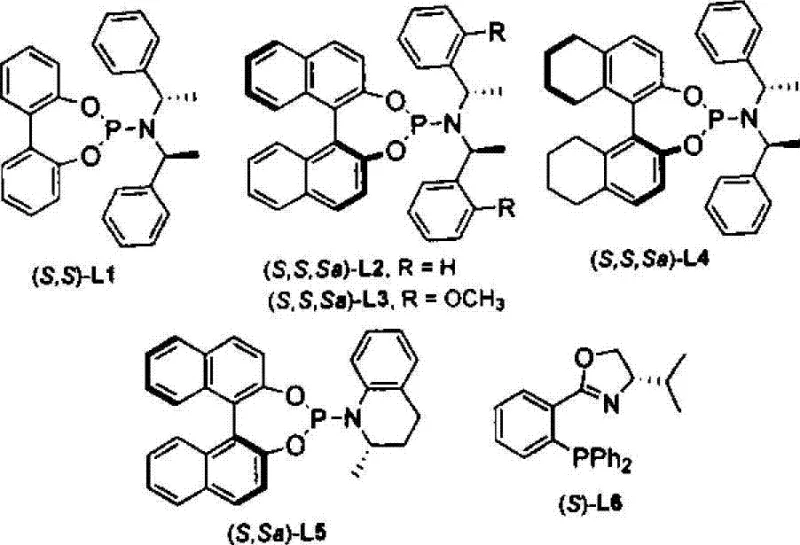
The core of this technological advancement lies in the precise engineering of the catalytic cycle, where the interplay between the iridium precursor and the chiral ligand dictates the stereochemical outcome. The patent specifies the use of phosphine-amine type ligands, such as those depicted in the structural diagrams, which create a chiral environment around the metal center to enforce high enantioselectivity. Mechanistic studies suggest that the iridium complex facilitates the formation of a pi-allyl intermediate that is selectively attacked by the sulfur nucleophile at the branched position. This regioselectivity is crucial, as linear isomers are often difficult to separate and represent a significant yield loss in traditional processes. Furthermore, the inclusion of specific additives like potassium acetate or cesium fluoride plays a vital role in modulating the reactivity of the nucleophile and stabilizing the catalytic species. The result is a process that consistently delivers enantiomeric excess (ee) values reaching up to 95%, ensuring that the resulting chiral allyl thiocarboxylates meet the stringent purity requirements demanded by modern drug discovery programs.
How to Synthesize Chiral Allyl Thiocarboxylates Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalytic species and the control of reaction parameters to maximize yield and selectivity. The process begins with the generation of the active iridium complex by mixing the dimeric precursor with the chiral ligand in a suitable solvent, followed by the addition of the substrates and additives. Maintaining the temperature within the optimal range of -20°C to 30°C is critical to balance reaction rate and stereocontrol, while the choice of solvent, such as dichloromethane or THF, influences the solubility of the ionic intermediates. Detailed standardized synthetic steps see the guide below.
- Prepare the reaction mixture by combining potassium thiocarboxylate and allyl carbonate compounds in an organic solvent such as dichloromethane.
- Add the iridium catalyst precursor [Ir(COD)Cl]2 along with the specific chiral ligand and necessary additives like potassium acetate.
- Maintain the reaction temperature between -20°C and 30°C for 2 to 48 hours, then isolate the product via chromatography or distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-catalyzed methodology offers substantial benefits for supply chain stability and cost management in the manufacturing of fine chemicals. The ability to operate under mild conditions reduces energy consumption and minimizes the risk of thermal runaway, which enhances overall process safety and lowers insurance and infrastructure costs. Moreover, the high selectivity of the reaction reduces the burden on downstream purification units, as fewer byproducts and isomers are generated, leading to significant savings in solvent usage and waste disposal. For procurement managers, this translates to a more predictable production timeline and a reduced risk of batch failures due to impurity profile deviations. The robustness of the catalyst system also implies that raw material variability can be better tolerated, ensuring consistent output quality even when sourcing bulk chemicals from different suppliers.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the use of low catalyst loading significantly decrease the raw material costs associated with each batch. Additionally, the simplified workup procedure reduces the requirement for extensive chromatographic separations, which are often the most expensive step in fine chemical production. By avoiding the use of toxic heavy metal scavengers required for other transition metals, the process further lowers waste treatment expenses and regulatory compliance costs.
- Enhanced Supply Chain Reliability: The mild reaction conditions and broad substrate scope allow for flexible manufacturing schedules that can adapt to fluctuating demand without compromising product quality. The availability of the iridium catalyst and ligands from established suppliers ensures that production is not bottlenecked by scarce reagents. This reliability is crucial for long-term contracts with pharmaceutical clients who require guaranteed continuity of supply for their clinical and commercial programs.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents and standard reactor equipment that are readily available in multi-purpose chemical plants. The high atom economy and reduced waste generation align with green chemistry principles, making it easier to obtain environmental permits and meet corporate sustainability goals. This environmental compatibility is increasingly becoming a deciding factor for multinational corporations when selecting contract development and manufacturing organizations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing production portfolios.
Q: What are the advantages of using iridium catalysis for sulfur-containing compounds?
A: Traditional metal catalysts often suffer from poisoning effects due to sulfur nucleophiles. The iridium complex described in CN103896816A overcomes this by maintaining high catalytic activity and achieving excellent enantioselectivity up to 95% ee.
Q: What are the typical reaction conditions for this synthesis?
A: The process operates under mild conditions, typically between -20°C to 30°C, using common organic solvents like DCM or THF, which facilitates easier scale-up and safety management compared to harsher traditional methods.
Q: How does this method impact impurity profiles?
A: The high regioselectivity (up to 90:10) and enantioselectivity significantly reduce the formation of unwanted isomers, simplifying downstream purification and ensuring higher purity specifications for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Allyl Thiocarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation therapeutics and agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of chiral allyl thiocarboxylate meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance means that you can rely on us for consistent material that supports your critical research and manufacturing timelines.
We invite you to engage with our technical procurement team to discuss how this advanced iridium-catalyzed technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this more efficient synthetic route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to be your strategic partner in complex chemical synthesis.
