Scalable Synthesis of 1-BOC-3-piperidone: A High-Efficiency Route for Pharmaceutical Intermediates
The pharmaceutical industry continuously demands robust and efficient synthetic routes for key heterocyclic intermediates, and the synthesis of 1-BOC-3-piperidone stands as a critical example of process optimization. As detailed in patent CN103304472A, a novel methodology has been established that transforms the production landscape for this valuable building block, moving away from inefficient legacy processes toward a streamlined, high-yield protocol. This technical insight report analyzes the proprietary three-step sequence originating from 3-hydroxypyridine, which leverages a combination of selective reduction, robust nitrogen protection, and catalytic Oppenauer oxidation. By integrating these specific chemical transformations, the process achieves a total yield exceeding 80% and purity levels greater than 98%, addressing the stringent quality requirements of modern drug discovery and development pipelines. The structural integrity of the target molecule, characterized by the tert-butoxycarbonyl protecting group on the piperidine nitrogen and the ketone functionality at the 3-position, is preserved throughout this gentle yet effective synthetic pathway.
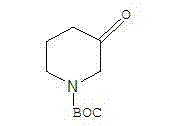
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-BOC-3-piperidone and related piperidone derivatives has been plagued by significant inefficiencies that hinder large-scale commercial viability. Traditional routes often suffer from convoluted operational procedures that require multiple purification steps, leading to substantial material loss at each stage. The patent literature explicitly notes that prior art methods frequently result in overall yields hovering around merely 40%, which represents a massive economic burden when scaling to metric ton quantities. Furthermore, these older methodologies are environmentally detrimental, generating copious amounts of wastewater and solid residues that complicate disposal and increase the environmental footprint of the manufacturing facility. The reliance on harsh reaction conditions in some conventional paths also poses safety risks and limits the choice of compatible functional groups, thereby restricting the versatility of the intermediate for downstream applications in complex API synthesis.
The Novel Approach
In stark contrast, the innovative route described in CN103304472A introduces a paradigm shift by prioritizing atom economy and operational simplicity without compromising on product quality. This novel approach utilizes 3-hydroxypyridine as a readily available and cost-effective starting material, initiating a cascade of reactions that are both chemoselective and high-yielding. The strategy employs a mild reduction followed by a standard protection step, culminating in an Oppenauer oxidation that elegantly installs the required carbonyl group. By optimizing reaction parameters such as temperature, solvent systems, and catalyst loading, this method drastically simplifies the workup procedure, replacing complex chromatographic separations with straightforward extraction and distillation techniques. The result is a green chemistry protocol that not only boosts the total yield to over 80% but also significantly reduces the generation of hazardous waste, aligning perfectly with the sustainability goals of modern chemical manufacturing.
Mechanistic Insights into the Three-Step Synthetic Sequence
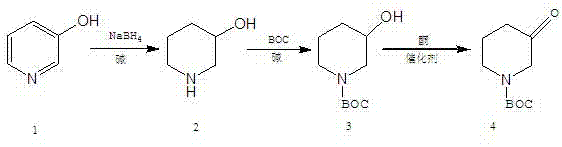
The core of this synthetic breakthrough lies in the precise execution of three distinct chemical transformations, each optimized to maximize conversion and minimize by-product formation. The sequence begins with the reduction of the aromatic pyridine ring of 3-hydroxypyridine using sodium borohydride in an alkaline medium. This step is critical as it converts the stable aromatic system into the saturated piperidine ring, specifically yielding 3-hydroxypiperidine. The use of an alkaline environment stabilizes the borohydride species, preventing premature decomposition and ensuring a controlled release of hydride ions, which is essential for achieving the reported 95% yield in this initial step. Following this, the secondary amine nitrogen is selectively protected using di-tert-butyl dicarbonate (BOC anhydride) under basic conditions. This protection is vital for preventing side reactions during the subsequent oxidation phase and ensures that the nitrogen lone pair does not interfere with the aluminum-based catalyst used later.
The final and most mechanistically interesting step is the Oppenauer oxidation, which converts the secondary alcohol of the protected intermediate into the target ketone. This reversible reaction is driven to completion by using a large excess of a sacrificial ketone, typically cyclohexanone, in the presence of an aluminum alkoxide catalyst such as aluminum isopropoxide. The mechanism involves the formation of a six-membered transition state where the hydride is transferred from the substrate alcohol to the sacrificial ketone. This metal-catalyzed hydride transfer is highly specific for secondary alcohols and operates under relatively mild thermal conditions (around 80°C), preserving the acid-sensitive BOC group which might be compromised under stronger oxidative conditions. The entire reaction pathway is depicted below, illustrating the flow from the aromatic precursor to the final saturated ketone.
How to Synthesize 1-BOC-3-piperidone Efficiently
Implementing this synthesis requires careful attention to the specific stoichiometric ratios and thermal profiles outlined in the patent to ensure reproducibility and safety. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors by maintaining strict control over exothermic events, particularly during the borohydride reduction phase. Operators must adhere to the specified addition rates and temperature ramps to prevent runaway reactions and ensure the formation of the desired regioisomer. While the general workflow is straightforward, the nuances of the workup—specifically the pH adjustments and solvent choices during extraction—are critical for isolating the intermediate in high purity before the final oxidation. For a comprehensive, step-by-step technical guide including exact reagent quantities and safety protocols, please refer to the standardized synthesis instructions provided below.
- Reduce 3-hydroxypyridine using sodium borohydride in an alkaline solution to form 3-hydroxypiperidine.
- Protect the nitrogen atom of 3-hydroxypiperidine with di-tert-butyl dicarbonate (BOC anhydride) under basic conditions.
- Oxidize the protected intermediate using aluminum isopropoxide and a ketone (Oppenauer oxidation), followed by vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend far beyond simple chemical curiosity. The primary value proposition lies in the dramatic improvement of mass balance; by nearly doubling the yield compared to traditional methods, the consumption of raw materials per kilogram of finished product is significantly reduced. This efficiency directly translates to a lower cost of goods sold (COGS), as less starting material is wasted, and the throughput of existing reactor capacity is effectively doubled without requiring capital expenditure on new equipment. Furthermore, the elimination of complex purification steps such as column chromatography in favor of vacuum distillation streamlines the production timeline, reducing labor costs and energy consumption associated with solvent handling and recovery.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the high atom economy and the use of commodity chemicals like sodium borohydride and aluminum isopropoxide. By avoiding expensive transition metal catalysts or specialized reagents that require rigorous removal to meet pharmaceutical standards, the downstream processing costs are drastically simplified. The ability to achieve high purity through distillation rather than recrystallization or chromatography means that solvent recovery rates are higher and waste disposal fees are lower, contributing to a leaner and more cost-effective manufacturing model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reliance on 3-hydroxypyridine as the starting material ensures a stable and secure supply chain, as this feedstock is widely produced and commercially available from multiple global vendors. The robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without catastrophic failure, reduces the risk of batch failures that can disrupt supply continuity. This reliability allows for more accurate demand forecasting and inventory planning, ensuring that critical timelines for API production are met without the delays often associated with low-yield or finicky synthetic routes.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing unit operations that are standard in any GMP-compliant facility, such as stirred tank reactors and falling film evaporators. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, minimizing the regulatory burden and potential fines associated with effluent discharge. This 'green' profile not only future-proofs the supply chain against tightening environmental laws but also enhances the corporate social responsibility profile of the final drug product, a factor that is becoming increasingly important to stakeholders and end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1-BOC-3-piperidone synthesized via this specific route. These answers are derived directly from the experimental data and claims presented in the source patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating route feasibility and procurement teams assessing vendor capabilities. The responses cover aspects of yield optimization, impurity profiles, and the specific advantages of the Oppenauer oxidation step in this context.
Q: What is the primary advantage of the Oppenauer oxidation route for 1-BOC-3-piperidone?
A: The primary advantage is the significant increase in total yield, reaching over 80% compared to conventional methods which often yield around 40%, alongside a much cleaner reaction profile that minimizes waste generation.
Q: How is the purity of the final 1-BOC-3-piperidone ensured?
A: High purity (>98%) is achieved through a rigorous final purification step involving vacuum distillation at controlled temperatures (104-105°C) and low pressure (60 Pa), effectively removing residual solvents and by-products.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is explicitly designed for industrial scalability, utilizing common reagents like sodium borohydride and aluminum isopropoxide, and avoiding complex chromatographic separations in favor of standard distillation techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-BOC-3-piperidone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the lab are faithfully reproduced on an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of 1-BOC-3-piperidone meets the exacting standards required for pharmaceutical intermediate applications. Our infrastructure is designed to handle the specific thermal and safety requirements of reactions involving borohydrides and aluminum alkoxides, guaranteeing a safe and consistent supply.
We invite you to collaborate with us to leverage this efficient synthesis technology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield route for your specific volume requirements. We encourage you to contact us today to discuss your needs,索取 specific COA data for our current inventory, and review our detailed route feasibility assessments to ensure seamless integration into your supply chain.
