Advanced Synthesis of 3-Substituted Glutaric Acid Diesters for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for constructing five-membered ring systems, which serve as critical scaffolds in numerous active pharmaceutical ingredients (APIs). Patent CN108358783B introduces a transformative preparation method for 3-substituted glutaric acid diesters and glutaconic acid diesters, addressing long-standing safety and efficiency bottlenecks in intermediate synthesis. This technology leverages the classic reactivity of malonic diesters but optimizes the workflow to eliminate hazardous gaseous reagents, marking a significant evolution in process safety. By shifting from unstable gas feeds to stable liquid and solid precursors, the method not only enhances operator safety but also simplifies the engineering requirements for commercial manufacturing. For R&D directors and procurement specialists, this patent represents a viable alternative to legacy processes that rely on dangerous feedstocks like ketene or high-pressure hydrogenation, offering a pathway to more reliable supply chains for complex organic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted glutaric derivatives has been plagued by significant safety hazards and operational complexities that hinder efficient commercial scale-up. Prior art, such as German patent DE1200279, relies on the reaction of dichloromethyl methyl ether with ketene; however, ketene is a highly toxic, unstable gas that requires rigorous containment and poses severe inhalation risks, making work-up procedures difficult and dangerous. Other approaches, like those reported by Yamamoto et al., utilize expensive starting materials such as methyl 3-hydroxyglutarate and methyl iodide, driving up the raw material costs prohibitively for large-scale production. Furthermore, methods involving boron trifluoride catalysis generate extremely toxic fluoride smoke upon contact with moisture, necessitating specialized scrubbing systems and corrosion-resistant reactors that increase capital expenditure. Even catalytic hydrogenation routes described in Chinese patent CN101143824A require high-pressure equipment (10-30 kg) to handle hydrogen gas, introducing explosion risks and demanding heavy-duty reactor infrastructure that many fine chemical plants lack.
The Novel Approach
The methodology disclosed in CN108358783B fundamentally reengineers the synthesis by utilizing malonic acid diesters and halogenated methanes as the primary building blocks, effectively bypassing the need for gaseous ketene or high-pressure hydrogen. This novel approach employs a substitution reaction under alkaline conditions followed by decarboxylation and esterification, all of which can be conducted in standard enamel or stainless steel reactors under mild thermal conditions. By replacing hazardous gases with stable liquids and solids, the process drastically reduces the safety risk profile, allowing for easier operation and simpler post-reaction work-up procedures. The use of conventional bases like sodium alkoxides and common solvents ensures that the reaction is accessible to a wide range of manufacturing facilities without the need for exotic catalysts or specialized high-pressure vessels. This shift not only improves the safety culture within the plant but also streamlines the regulatory compliance process, as the handling of less hazardous materials simplifies environmental and health safety reporting.
Mechanistic Insights into Malonic Ester Alkylation and Decarboxylation
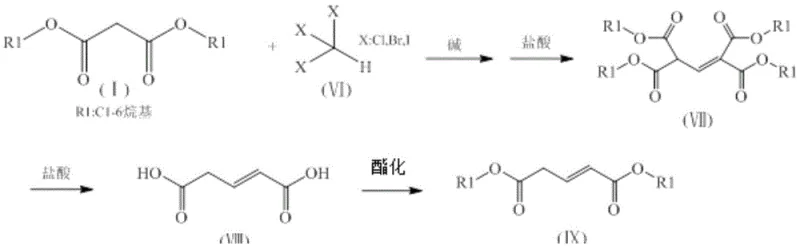
The core of this synthetic strategy relies on the well-established acidity of the alpha-protons in malonic diesters, which allows for the formation of a nucleophilic enolate species upon treatment with a strong base. In the presence of bases such as sodium hydride, potassium hydride, or sodium alkoxides, the malonic diester (I) is deprotonated to form a reactive intermediate that attacks the electrophilic carbon of the halogenated methane. When synthesizing 3-substituted glutaric diesters, a monosubstituted halogenated methane (II) acts as the alkylating agent, introducing the desired substituent at the central carbon of the glutaric backbone. Conversely, for glutaconic diesters, a trihalomethane (VI) is employed, leading to a different substitution pattern that ultimately results in an unsaturated double bond after subsequent elimination and decarboxylation steps. The precision of this alkylation is critical, as it determines the regiochemistry of the final product and minimizes the formation of over-alkylated byproducts.
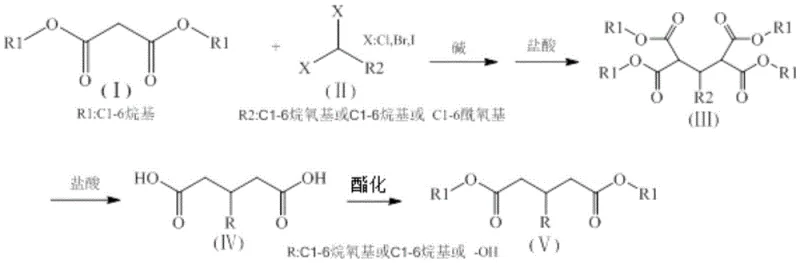
Following the initial alkylation, the intermediate undergoes a carefully controlled hydrolysis and decarboxylation sequence. The reaction mixture is treated with hydrochloric acid, which facilitates the cleavage of the ester groups and the subsequent loss of carbon dioxide from the beta-keto acid intermediate. This step is thermally driven, typically occurring at temperatures between 80-100°C, ensuring complete conversion to the dicarboxylic acid. The final stage involves Fischer esterification, where the dicarboxylic acid is refluxed with an alcohol in the presence of an acid catalyst such as concentrated sulfuric acid. This last transformation regenerates the ester functionality, yielding the target 3-substituted glutaric diester (V) or glutaconic diester (IX) with high purity. The entire mechanistic pathway is designed to maximize atom economy while minimizing the generation of hazardous waste streams, aligning with modern green chemistry principles.
How to Synthesize 3-Substituted Glutaric Diester Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to ensure high yields and minimal impurity formation. The process begins with the activation of the malonic diester, followed by the controlled addition of the halogenated species to manage exotherms. Detailed operational parameters, including specific molar ratios and heating profiles, are essential for reproducing the high purity levels demonstrated in the patent examples. For process chemists looking to implement this route, understanding the nuances of the work-up and purification stages is just as critical as the reaction itself. The detailed standardized synthesis steps for implementing this technology are provided in the guide below.
- React malonic diester with a base (such as sodium alkoxide) in a solvent like THF or alcohol at 70-110°C to form the enolate.
- Add monosubstituted halogenated methane or trihalomethane to the reaction mixture, maintaining temperature control to facilitate substitution.
- Perform acid hydrolysis and decarboxylation followed by esterification with alcohol and acid catalyst to yield the final diester product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits by mitigating risks associated with volatile raw material markets and complex logistics. The shift away from gaseous reagents like ketene and boron trifluoride eliminates the need for specialized cylinder handling, gas monitoring systems, and emergency response protocols dedicated to toxic inhalation hazards. This simplification of the raw material profile allows procurement managers to source inputs from a broader base of chemical suppliers, enhancing competition and stabilizing pricing. Furthermore, the reliance on commodity chemicals such as malonic esters and common halogenated solvents ensures a consistent supply flow, reducing the likelihood of production stoppages due to precursor shortages. The overall process design supports a more resilient supply chain capable of withstanding market fluctuations and regulatory changes regarding hazardous substance transport.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the elimination of expensive and specialized catalysts, such as zinc isobutyrate or boron trifluoride, which often require complex recovery systems or single-use disposal. By utilizing inexpensive bases like sodium methoxide and readily available halogenated methanes, the direct material costs are drastically lowered compared to legacy routes that depend on high-value starting materials like methyl 3-hydroxyglutarate. Additionally, the ability to conduct reactions in standard reactors without high-pressure ratings reduces capital depreciation costs and maintenance expenses associated with specialized equipment. The simplified work-up procedure, which avoids complex gas scrubbing and high-pressure filtration, further contributes to operational expenditure savings by reducing labor hours and utility consumption.
- Enhanced Supply Chain Reliability: The use of liquid and solid raw materials at ambient conditions greatly simplifies logistics and storage requirements, removing the complexities associated with managing pressurized gas cylinders or cryogenic feeds. This stability ensures that inventory can be held safely on-site for extended periods without degradation, providing a buffer against supply disruptions. The widespread availability of malonic diesters and halogenated solvents in the global chemical market means that multiple qualified suppliers can be vetted, preventing single-source dependency. Consequently, lead times for raw material delivery are minimized, and the overall agility of the manufacturing schedule is improved, allowing for faster response to customer demand spikes.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gas emissions make this process inherently easier to scale from pilot plant to multi-ton commercial production without encountering the engineering bottlenecks typical of high-pressure or gas-phase reactions. The reduction in hazardous waste generation, particularly the avoidance of fluoride-containing byproducts and heavy metal residues, simplifies wastewater treatment and lowers the cost of environmental compliance. This alignment with stricter environmental regulations future-proofs the manufacturing asset, reducing the risk of shutdowns due to non-compliance. The straightforward purification via rectification further ensures that the final product meets stringent quality standards with minimal solvent waste, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios. Comprehensive responses to specific technical queries can be found in the section below.
Q: What are the safety advantages of this new synthesis method compared to traditional ketene routes?
A: Traditional methods often utilize ketene, a highly toxic and unstable gas, or boron trifluoride which generates hazardous fluoride smoke upon hydrolysis. The patented method replaces these gaseous hazards with stable liquid or solid raw materials like malonic diesters and halogenated methanes, significantly reducing operational risk and eliminating the need for specialized high-pressure gas reactors.
Q: Can this process be scaled for industrial production without specialized equipment?
A: Yes, the process is designed for scalability using conventional chemical reactors. It operates under mild conditions (70-110°C) and atmospheric or low pressure, avoiding the extreme pressures (10-30 kg) required by catalytic hydrogenation methods. This allows for deployment in standard fine chemical manufacturing facilities.
Q: What represents the primary cost driver reduction in this manufacturing route?
A: The primary cost reduction stems from the use of commodity chemicals such as dimethyl malonate and common halogenated solvents, which are widely available and inexpensive compared to specialized precursors like methyl 3-hydroxyglutarate. Additionally, the elimination of expensive noble metal catalysts and complex gas handling infrastructure lowers both CAPEX and OPEX.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted Glutaric Diester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of downstream pharmaceutical applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 3-substituted glutaric diester meets the exacting standards required for API synthesis. Our facility is equipped to handle the specific thermal and chemical requirements of this alkylation-based process, delivering consistent quality and reliability to our global partners.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall production costs.
