Advanced Manufacturing of 3-Substituted Glutaric Acid Diesters for Pharmaceutical Applications
Advanced Manufacturing of 3-Substituted Glutaric Acid Diesters for Pharmaceutical Applications
The landscape of fine chemical synthesis is constantly evolving to prioritize safety, efficiency, and scalability, particularly for critical building blocks used in drug discovery. A significant breakthrough in this domain is detailed in Chinese patent CN108358783B, which outlines a robust preparation method for 3-substituted glutaric acid diesters and glutaconic acid diesters. These compounds serve as pivotal precursors for constructing various five-membered ring systems, which are ubiquitous motifs in bioactive molecules and agrochemical agents. The innovation lies in shifting away from hazardous gaseous reagents toward a liquid-phase alkylation strategy using malonic diesters. This transition not only mitigates severe safety risks associated with traditional ketene chemistry but also streamlines the operational workflow, making it an attractive option for a reliable pharmaceutical intermediate supplier seeking to optimize their manufacturing portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glutaric acid derivatives has been plagued by significant engineering and safety challenges that hinder efficient commercial scale-up of complex esters. For instance, German patent DE1200279 describes a route involving the reaction of dichloromethyl methyl ether with ketene catalyzed by zinc isobutyrate. Ketene is a highly toxic, unstable gas that demands rigorous containment protocols and specialized reactor designs to prevent leakage and exposure. Similarly, international patent WO2010/121769 relies on boron trifluoride catalysis with ketene and trimethyl orthoformate; boron trifluoride is notorious for generating extremely toxic fluoride smoke upon contact with moisture, creating severe environmental and worker safety liabilities. Furthermore, alternative hydrogenation routes reported in CN101143824A require high-pressure equipment (10-30 kg) to handle hydrogen gas, significantly increasing capital expenditure and operational complexity. These legacy methods impose heavy burdens on cost reduction in fine chemical manufacturing due to the need for exotic materials of construction and extensive safety mitigation systems.
The Novel Approach
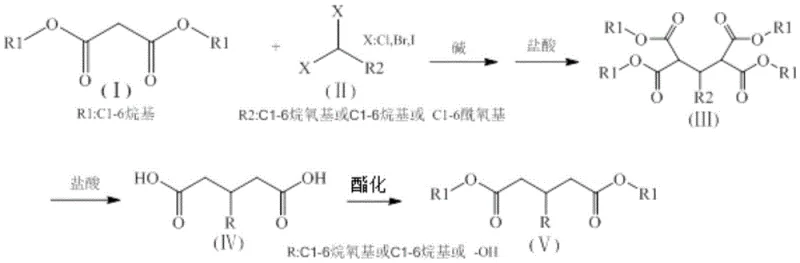
In stark contrast, the methodology disclosed in CN108358783B leverages the classic reactivity of malonic esters to achieve the same structural outcomes under remarkably mild conditions. By utilizing malonic diester as the starting scaffold and reacting it with halogenated methane derivatives in the presence of a base, the process avoids gaseous feedstocks entirely. The raw materials, such as dimethyl malonate and various haloalkanes, are typically liquids or solids at room temperature, allowing for standard dosing and handling procedures in conventional enamel reactors. This fundamental shift in feedstock state dramatically simplifies the process engineering requirements. As illustrated in the reaction scheme below, the substitution reaction proceeds smoothly to form the intermediate, which is subsequently converted to the target diester through hydrolysis and esterification. This approach effectively bypasses the need for high-pressure hydrogenation or toxic Lewis acid catalysts, offering a streamlined pathway that enhances both operator safety and process economics.
Mechanistic Insights into Malonic Ester Alkylation and Decarboxylation
The core of this synthetic strategy relies on the high acidity of the alpha-protons in malonic diesters, which allows for facile deprotonation by strong bases such as sodium alkoxides or metal hydrides. Once the enolate is formed, it acts as a potent nucleophile, attacking the electrophilic carbon of the halogenated methane (either mono-substituted or trihalomethane). This alkylation step is critical for introducing the desired substituent at the 3-position of the glutaric backbone. Following alkylation, the intermediate undergoes a carefully controlled hydrolysis and decarboxylation sequence. Treatment with hydrochloric acid at elevated temperatures (80-100°C) cleaves the ester groups and induces the loss of carbon dioxide from one of the carboxyl groups. This decarboxylation is thermodynamically driven and serves to shorten the carbon chain appropriately while establishing the glutaric acid framework. The precision of this step is vital for impurity control, as incomplete decarboxylation can lead to tricarboxylic acid byproducts that are difficult to separate.
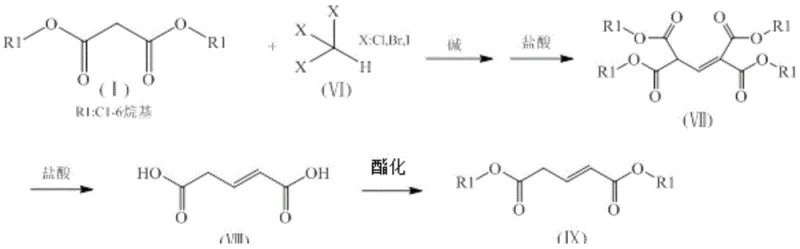
For the synthesis of glutaconic acid diesters, the mechanism diverges slightly by employing trihalomethanes, which introduce multiple halogen atoms that facilitate elimination reactions to form the double bond characteristic of the glutaconate structure. The subsequent esterification step converts the free dicarboxylic acids back into diesters using simple alcohols and acid catalysts like concentrated sulfuric acid. This final transformation is equilibrium-driven, necessitating the removal of water or the use of excess alcohol to push the reaction to completion. The entire sequence is designed to minimize side reactions; for example, the use of rectification as the final purification step ensures that high-purity glutaric acid derivatives are obtained by separating the target esters from unreacted alcohols and minor oligomeric impurities based on boiling point differences. This mechanistic clarity allows for precise tuning of reaction parameters to maximize yield and selectivity.
How to Synthesize 3-Substituted Glutaric Acid Diesters Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to ensure optimal conversion and minimize waste. The process begins with the generation of the nucleophilic enolate species, followed by the addition of the electrophile, and concludes with a workup that isolates the pure diester. The patent provides specific molar ratios and thermal profiles that have been validated to produce high-quality material suitable for downstream pharmaceutical applications. Operators must adhere to the specified heating and cooling cycles to manage the exothermic nature of the alkylation and the gas evolution during decarboxylation. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of the operational protocol.
- React malonic diester with alkali (e.g., sodium alkoxide) in a solvent like THF or ethanol at 70-110°C to form the enolate.
- Add mono-substituted dihalomethane or trihalomethane, then treat the intermediate with hydrochloric acid for hydrolysis and decarboxylation at 80-100°C.
- Perform esterification of the resulting dicarboxylic acid with alcohol under reflux with an acid catalyst, followed by rectification to purify the final diester.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented method offers substantial strategic advantages by fundamentally altering the risk profile and cost structure of producing these key intermediates. The shift from gaseous, hazardous reagents to stable, commodity-grade liquids and solids directly translates to lower logistical costs and reduced regulatory hurdles. Suppliers can source raw materials like dimethyl malonate and chloroform from a wide global market, ensuring continuity of supply even during regional disruptions. Furthermore, the elimination of high-pressure hydrogenation steps removes the need for expensive, specialized autoclaves, allowing production to occur in standard multipurpose reactors found in most fine chemical facilities. This flexibility significantly reduces the barrier to entry for manufacturing and allows for rapid capacity expansion to meet market demand without massive capital investment in new infrastructure.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the reaction setup and the use of inexpensive, widely available catalysts. By avoiding the need for specialized gas handling equipment and toxic Lewis acids like boron trifluoride, manufacturers can drastically reduce both capital expenditure (CAPEX) and operating expenditure (OPEX). The mild reaction conditions also imply lower energy consumption for heating and cooling compared to high-pressure or cryogenic processes. Additionally, the high yields reported in the examples suggest efficient atom economy, meaning less raw material is wasted, further driving down the cost per kilogram of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: Reliance on liquid and solid raw materials inherently stabilizes the supply chain against the volatility often seen with industrial gases. Malonic esters and halogenated methanes are produced at massive scales for various industries, ensuring a robust and competitive supplier base. This abundance prevents single-source bottlenecks and allows procurement managers to negotiate better terms. Moreover, the stability of these materials simplifies storage and transportation requirements, reducing the risk of delays caused by hazardous material shipping restrictions. This reliability is crucial for maintaining consistent production schedules for downstream drug manufacturers who depend on just-in-time delivery of critical intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution in 50L and 100L enamel reactors in the patent examples. Scaling to multi-ton production involves straightforward linear scaling of reagent quantities without the need for complex engineering redesigns. From an environmental standpoint, the avoidance of toxic fluoride byproducts and high-pressure hydrogen gas simplifies waste treatment and emissions control. The use of rectification for purification allows for the recovery and recycling of solvents like methanol and ethanol, aligning with green chemistry principles and reducing the overall environmental footprint of the manufacturing site. This compliance ease facilitates faster regulatory approvals and smoother audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technical disclosures and comparative advantages highlighted in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs. The answers reflect the practical realities of running this chemistry at an industrial scale, focusing on safety, purity, and operational simplicity.
Q: Why is the malonic ester method safer than traditional ketene routes?
A: Traditional methods often utilize ketene, a highly toxic and reactive gas that requires specialized high-pressure equipment and poses significant safety risks. The patented method uses liquid or solid raw materials like malonic diester and halogenated methane, eliminating the need for hazardous gas handling infrastructure.
Q: What are the typical purity levels achievable with this process?
A: The process utilizes rectification (distillation) as the final purification step, which is highly effective for removing solvent and byproduct impurities. Examples in the patent demonstrate contents reaching 98.8% to 99.5% (GC), suitable for high-grade pharmaceutical intermediate applications.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction conditions are mild (70-110°C) and utilize standard enamel reactors rather than specialized high-pressure vessels. The use of common solvents and catalysts facilitates easy scale-up from laboratory to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Substituted Glutaric Acid Diester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development pipelines. Our technical team has extensively analyzed the potential of the malonic ester alkylation route described in CN108358783B and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to full-scale market supply. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the 99%+ purity levels required for sensitive pharmaceutical applications, guaranteeing that every batch meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for glutaric acid derivatives. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our advanced manufacturing capabilities can enhance your project's efficiency and profitability. Let us be your trusted partner in navigating the complexities of fine chemical synthesis.
