Scalable Palladium-Catalyzed Desulfurization Addition for High-Purity Aromatic Ketones
Scalable Palladium-Catalyzed Desulfurization Addition for High-Purity Aromatic Ketones
The synthesis of aromatic ketones represents a cornerstone capability in modern organic chemistry, serving as critical building blocks for pharmaceuticals, agrochemicals, and functional materials. A significant technological breakthrough in this domain is detailed in patent CN108383698B, which discloses a novel preparation method utilizing a palladium-catalyzed desulfurization addition reaction. This innovative approach leverages aryl sulfonyl hydrazides and nitrile compounds to construct the aryl-carbonyl motif under remarkably mild conditions. Unlike traditional methods that often rely on corrosive reagents or unstable intermediates, this protocol employs stable, solid aryl sulfonyl hydrazides as efficient aryl sources. The process operates effectively in polar aprotic solvents such as DMSO or DMF, facilitated by trace amounts of water and specific nitrogen-containing ligands. For global procurement teams and R&D directors seeking a reliable aromatic ketone supplier, this technology offers a robust pathway to high-value intermediates with exceptional functional group tolerance.
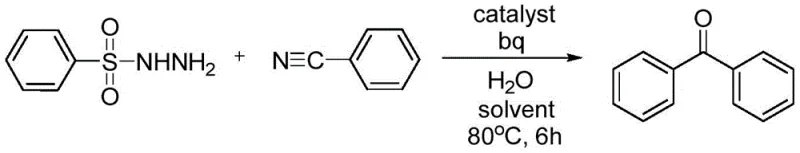
The strategic value of this methodology lies in its ability to bypass the limitations of classical synthetic routes. By activating the C-S bond rather than relying on traditional C-X or C-H activation, the process opens new avenues for cost reduction in pharmaceutical intermediate manufacturing. The reaction conditions are optimized to run at temperatures between 70°C and 100°C, typically completing within 5 to 7 hours, which aligns well with standard batch processing timelines in commercial plants. Furthermore, the compatibility with both aromatic and aliphatic nitriles ensures that a diverse library of ketone derivatives can be accessed from a single platform technology, enhancing supply chain flexibility for complex molecule production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of aromatic ketones has been dominated by the Friedel-Crafts acylation reaction, which involves the reaction of aromatic compounds with acyl chlorides in the presence of stoichiometric amounts of Lewis acids like aluminum trichloride. This classical approach suffers from severe drawbacks, including the generation of large quantities of corrosive waste, difficult product isolation, and poor atom economy. Additionally, the use of acyl chlorides introduces safety hazards due to their moisture sensitivity and lachrymatory nature, complicating logistics and storage. Alternative transition metal-catalyzed methods involving organoboron reagents or aryl halides often require expensive starting materials and generate stoichiometric salt byproducts. These conventional strategies frequently struggle with substrate scope, particularly when dealing with electron-deficient aromatics or sensitive functional groups, leading to inconsistent yields and purification challenges that inflate the overall cost of goods.
The Novel Approach
The patented method introduces a paradigm shift by utilizing aryl sulfonyl hydrazides as stable, non-toxic arylating agents. These reagents are easily prepared via hydrazinolysis of aryl sulfonyl chlorides and exist as solid compounds with excellent shelf stability, simplifying inventory management and handling. Under the catalytic influence of palladium complexes and bidentate nitrogen ligands, the aryl sulfonyl hydrazide undergoes denitrogenation and desulfurization to generate a reactive aryl-palladium species in situ. This species then inserts into the polar multiple bond of the nitrile substrate, followed by hydrolysis to yield the target aromatic ketone. This route eliminates the need for corrosive Lewis acids and avoids the use of unstable organometallic reagents. The inclusion of water as a necessary additive facilitates the hydrolysis step without requiring harsh acidic or basic workups, thereby streamlining the downstream processing and significantly improving the environmental profile of the synthesis.
Mechanistic Insights into Pd-Catalyzed C-S Bond Activation
The core of this transformation relies on the activation of the carbon-sulfur bond within the aryl sulfonyl hydrazide scaffold, a process that is thermodynamically challenging but kinetically accessible under palladium catalysis. The catalytic cycle initiates with the oxidative addition or coordination of the palladium center to the sulfonyl hydrazide, followed by the extrusion of nitrogen gas and sulfur dioxide (or sulfinate species) to form an aryl-palladium intermediate. This step is critical as it converts a stable precursor into a highly reactive nucleophilic species capable of attacking the electrophilic carbon of the nitrile group. The presence of bidentate ligands, such as biquinoline or phenanthroline, stabilizes the palladium center and modulates its electronic properties to favor this specific insertion pathway over competing side reactions. The subsequent insertion of the nitrile into the Pd-C bond forms an imino-palladium complex, which is then hydrolyzed by the added water to release the ketone product and regenerate the active catalyst species.
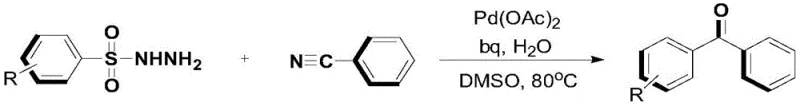
Impurity control in this system is inherently managed by the specificity of the C-S bond activation mechanism. Since the aryl source is pre-functionalized as a sulfonyl hydrazide, there is minimal risk of homocoupling side reactions that often plague cross-coupling chemistries involving aryl halides. The reaction tolerates a wide range of substituents on both the hydrazide and the nitrile components, including halogens, nitro groups, alkoxy groups, and trifluoromethyl groups. This broad tolerance suggests that the catalytic cycle is robust against electronic variations, preventing the accumulation of unreacted starting materials or partially reacted intermediates. Furthermore, the use of mild temperatures (80°C) minimizes thermal decomposition pathways, ensuring that the final crude product profile is clean and amenable to standard purification techniques like silica gel chromatography or crystallization, which is essential for meeting the stringent purity specifications required in API synthesis.
How to Synthesize Aromatic Ketones Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and atmospheric controls to maximize yield and reproducibility. The process begins by dispersing the aryl sulfonyl hydrazide and nitrile compound in a dry polar aprotic solvent under an inert atmosphere to prevent catalyst oxidation. The addition of the palladium catalyst and ligand must be followed by the careful introduction of a controlled amount of water, which acts as the oxygen source for the carbonyl group. Detailed standardized operating procedures regarding temperature ramping, reaction monitoring, and workup protocols are essential for scaling this chemistry from gram to kilogram scales. The following guide outlines the critical operational parameters derived from the patent data to ensure successful execution.
- Disperse phenylsulfonyl hydrazide, nitrile compound, palladium catalyst (e.g., Pd(OAc)2), and nitrogen-containing ligand in an organic solvent like DMSO or DMF under inert gas.
- Add a trace amount of water (molar ratio 1.5-2.5: 1 to nitrile) to facilitate the reaction cycle.
- Heat the mixture to 70-100°C for 5-7 hours, then extract with ethyl acetate and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed desulfurization technology offers tangible benefits in terms of raw material security and process economics. The primary advantage stems from the use of aryl sulfonyl hydrazides, which are commercially available or easily synthesized from abundant sulfonyl chlorides, ensuring a stable and continuous supply of key starting materials. Unlike methods relying on exotic organometallic reagents, these solid precursors are easy to transport and store, reducing logistical risks and warehousing costs. The reaction's high functional group tolerance means that fewer protection and deprotection steps are required during multi-step syntheses, leading to a significant reduction in overall process time and solvent consumption. This streamlined workflow directly translates to lower manufacturing costs and a smaller environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of stoichiometric Lewis acids and corrosive reagents drastically reduces waste disposal costs and equipment maintenance expenses associated with corrosion. Furthermore, the low catalyst loading (typically around 2 mol%) combined with the use of relatively inexpensive ligands like biquinoline ensures that the cost of the catalytic system remains manageable even at large scales. The high yields reported across a broad substrate scope minimize the loss of valuable intermediates, optimizing the overall material balance and reducing the cost per kilogram of the final aromatic ketone product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for flexibility in sourcing raw materials, as the process tolerates various substituents on the aromatic rings. This versatility means that supply chain disruptions for specific substituted nitriles or hydrazides can be mitigated by adjusting the synthetic route without compromising the final product quality. The use of common solvents like DMSO and DMF further simplifies procurement, as these are standard commodities in the chemical industry with reliable global availability, ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of hazardous gas evolution (other than nitrogen) make this process highly scalable from laboratory to commercial production volumes. The simplified workup procedure, involving standard extraction and chromatography, reduces the complexity of downstream processing units. Additionally, the reduced generation of acidic or saline waste streams simplifies effluent treatment, facilitating compliance with increasingly stringent environmental regulations and supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and mechanistic understanding presented in the patent documentation, providing clarity for technical teams evaluating this route for potential adoption. Understanding these nuances is crucial for assessing the feasibility of integrating this technology into existing production lines or new product development pipelines.
Q: What are the advantages of using aryl sulfonyl hydrazides over traditional acyl chlorides?
A: Aryl sulfonyl hydrazides are solid compounds with good stability, low toxicity, and ease of preparation compared to corrosive acyl chlorides. They serve as effective aryl sources via C-S bond activation, avoiding the harsh conditions of Friedel-Crafts acylation.
Q: Does this method support aliphatic nitriles?
A: Yes, the patented process demonstrates broad substrate applicability, successfully reacting with both aromatic and aliphatic nitrile compounds to yield corresponding ketones with high efficiency.
Q: What catalyst system is required for this transformation?
A: The reaction utilizes a palladium catalyst such as Pd(OAc)2 or Pd(PhCN)2Cl2 combined with a bidentate nitrogen-containing ligand like biquinoline (bq) or phenanthroline (phen) in solvents like DMSO or DMF.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the Pd-catalyzed desulfurization addition described in CN108383698B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot scale to full manufacturing is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of aromatic ketone meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for similar compounds and discuss route feasibility assessments for your upcoming campaigns. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
