Advanced Iridium-Catalyzed Synthesis of Optically Active Alkane Chiral Allylic Thioethers for Pharmaceutical Applications
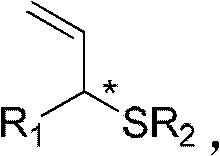
The pharmaceutical and fine chemical industries constantly seek robust methodologies for constructing chiral sulfur-containing motifs, which are prevalent in bioactive molecules ranging from vitamins to protein cross-linking reagents. Patent CN102675168A introduces a groundbreaking preparation method for alkane chiral allylic thioether compounds, addressing the longstanding challenge of synthesizing these structures with high optical purity. Unlike traditional approaches that often struggle with catalyst deactivation, this invention leverages a transition metal iridium complex to facilitate the reaction between allyl carbonates and sodium alkylthiolates. The result is a highly efficient pathway to optically pure compounds where the chiral center is precisely defined, offering a versatile scaffold for further drug development. This technology represents a significant leap forward in asymmetric synthesis, providing a reliable route to high-purity pharmaceutical intermediates that were previously difficult to access with such fidelity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral sulfur centers has relied heavily on asymmetric sulfur-Michael additions or traditional metal-catalyzed allylations, both of which present substantial drawbacks for large-scale manufacturing. The sulfur-Michael addition, while mature, often requires stoichiometric amounts of chiral auxiliaries or harsh reaction conditions that complicate downstream purification and increase waste generation. Furthermore, when attempting direct metal-catalyzed allylation, chemists frequently encounter the phenomenon of catalyst poisoning, where the sulfur nucleophile binds irreversibly to the metal center. This interaction precipitates the metal out of solution or forms catalytically inactive species, leading to poor conversion rates, inconsistent batch quality, and the need for excessive catalyst loading. These inefficiencies create significant bottlenecks in cost reduction in fine chemical manufacturing, as they necessitate complex workup procedures and result in lower overall yields.
The Novel Approach
The methodology described in CN102675168A circumvents these historical limitations by employing a specialized iridium catalyst system that exhibits remarkable tolerance to sulfur nucleophiles. By utilizing a chiral ligand in conjunction with an iridium precursor, the reaction proceeds with high regioselectivity and enantioselectivity under remarkably mild conditions, typically between -20°C and 30°C. This approach eliminates the need for stoichiometric chiral reagents, relying instead on a catalytic amount of the chiral inducer to drive the formation of the desired stereoisomer. The process accommodates a wide range of substrates, including various aryl and alkyl thiolates, demonstrating broad applicability across different chemical spaces. This robustness ensures consistent production of high-purity chiral thioethers, making it an ideal candidate for the commercial scale-up of complex organosulfur compounds required in modern drug synthesis.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Allylation
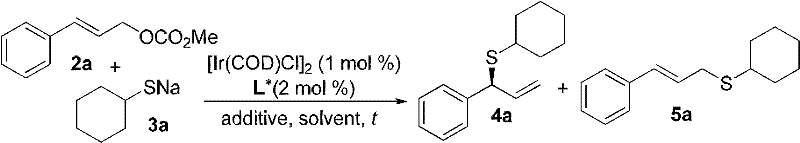
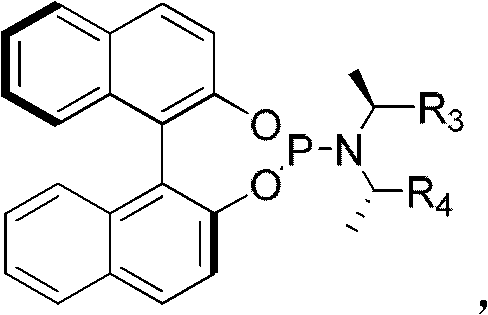
The core of this technological advancement lies in the unique structure of the chiral ligand and its interaction with the iridium metal center to form a highly active catalytic species. The ligand, characterized by a binaphthyl backbone modified with phosphoramidite groups, creates a sterically demanding chiral environment around the iridium atom. This specific geometry is crucial for differentiating between the enantiotopic faces of the allylic intermediate, thereby enforcing the formation of one specific enantiomer over the other with high precision. The presence of additives such as cesium fluoride or lithium chloride further modulates the reactivity of the nucleophile, enhancing the rate of the transmetallation step without compromising the integrity of the catalyst. This synergistic effect allows the reaction to proceed efficiently even at low catalyst loadings, typically around 1 mol%, which is economically advantageous for industrial applications.
Furthermore, the mechanism effectively mitigates the issue of sulfur poisoning that plagues other transition metal systems. The electronic properties of the iridium complex are tuned such that it can coordinate with the allylic carbonate and facilitate the nucleophilic attack by the thiolate without forming stable, inactive sulfur-metal bonds that would terminate the catalytic cycle. This stability is evidenced by the high turnover numbers and the ability to maintain high enantiomeric excess (up to 98% ee) across a diverse substrate scope. The reaction predominantly yields the branched allylic sulfide product with excellent regioselectivity, often exceeding a 90:10 ratio in favor of the chiral branched isomer over the linear byproduct. Such control is essential for R&D directors focused on impurity profiles, as it minimizes the formation of difficult-to-separate regioisomers.
How to Synthesize Chiral Allylic Thioethers Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent quality to maximize yield and stereochemical purity. The process begins with the in situ generation of the active iridium catalyst by mixing the iridium dimer precursor with the chiral ligand in a dry, oxygen-free environment, typically using tetrahydrofuran (THF) as the solvent. Once the catalyst is activated, the reaction is initiated by adding the allyl carbonate substrate and the sodium alkylthiolate salt along with a fluoride-based additive in a solvent like dichloromethane. The detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during laboratory or pilot-scale operations.
- Prepare the catalyst by reacting [Ir(COD)Cl]2 with the chiral phosphoramidite ligand in THF at 50°C for 30 minutes under argon protection.
- In a separate vessel, mix sodium alkylthiolate, cesium fluoride additive, and allyl carbonate in dry dichloromethane (DCM).
- Combine the catalyst solution with the reaction mixture and stir at temperatures between -20°C and 30°C for 2 to 48 hours until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iridium-catalyzed protocol offers distinct strategic advantages over legacy synthetic routes. The primary benefit stems from the drastic simplification of the process workflow, which directly translates to operational efficiency and reduced resource consumption. By avoiding the use of stoichiometric chiral auxiliaries and harsh reagents, the overall material cost per kilogram of product is significantly lowered, while the simplified purification process reduces solvent usage and waste disposal costs. Additionally, the mild reaction temperatures and ambient pressure requirements mean that existing standard reactor infrastructure can be utilized without the need for specialized cryogenic or high-pressure equipment, facilitating a smoother technology transfer from lab to plant.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high catalytic efficiency, which allows for the use of minimal amounts of the expensive iridium complex and chiral ligand. Since the catalyst loading is as low as 1 mol%, the direct cost associated with precious metals is kept to a minimum compared to processes requiring stoichiometric chiral reagents. Moreover, the high regioselectivity reduces the burden on downstream purification, meaning less chromatography or recrystallization is needed to isolate the target API intermediate. This streamlined workflow not only saves on labor and materials but also shortens the overall production cycle time, contributing to substantial cost savings in the long run.
- Enhanced Supply Chain Reliability: The robustness of this chemical transformation ensures a stable and predictable supply of critical chiral building blocks. The starting materials, such as allyl carbonates and sodium thiolates, are commercially available and inexpensive, reducing the risk of raw material shortages that can plague more exotic synthetic routes. The tolerance of the catalyst system to various functional groups means that a single standardized protocol can be adapted for multiple derivatives, simplifying inventory management and production planning. This flexibility allows manufacturers to respond quickly to changing market demands without the need for extensive process re-validation for each new analog.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method aligns well with green chemistry principles by operating under mild conditions and generating less hazardous waste. The absence of heavy metal precipitation issues simplifies effluent treatment, as the iridium remains in solution and can potentially be recovered or managed more easily than solid metal sludge. The scalability is further supported by the use of common organic solvents like DCM and THF, which are well-understood in industrial settings. This ease of scale-up ensures that production can be ramped from pilot batches to multi-ton commercial runs with minimal technical risk, securing the supply continuity for downstream pharmaceutical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this method overcome sulfur poisoning of metal catalysts?
A: The patented method utilizes a specialized chiral iridium complex that maintains high catalytic activity even in the presence of sulfur nucleophiles, which typically deactivate traditional transition metal catalysts by forming inactive precipitates.
Q: What is the enantiomeric excess (ee) achievable with this process?
A: The process demonstrates exceptional stereocontrol, achieving enantiomeric excess values up to 98% ee, with typical results ranging between 94% and 98% ee depending on the specific substrate and ligand combination used.
Q: Is this synthesis scalable for industrial production?
A: Yes, the reaction operates under mild conditions (-20°C to 30°C) using common organic solvents like DCM and THF, and employs readily available starting materials, making it highly suitable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Allylic Thioether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify enantiomeric excess and chemical identity. By leveraging the advanced iridium-catalyzed technology described in CN102675168A, we can offer you a competitive edge through superior product quality and consistent supply.
We invite you to collaborate with us to optimize your supply chain for these valuable sulfur-containing compounds. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your R&D and commercial manufacturing goals effectively.
