Scalable Synthesis of 1,1-Dibromo-2,2-Bis(Chloromethyl)Cyclopropane for Pharma Applications
The pharmaceutical industry's relentless pursuit of novel bioisosteres has placed bicyclo[1.1.1]pentane derivatives at the forefront of medicinal chemistry, driving an urgent demand for reliable precursors. Patent CN110759840B introduces a groundbreaking methodology for the synthesis of 1,1-dibromo-2,2-bis(chloromethyl)cyclopropane, a pivotal intermediate in the construction of these strained cage systems. Unlike legacy processes that rely on hazardous thermal decomposition, this invention leverages a sophisticated five-step sequence originating from 2-amino-2-hydroxymethyl propane-1,3-diol. The technical breakthrough lies not merely in the chemical transformation but in the engineering of safety and scalability into the molecular architecture of the process itself. By replacing unstable diazo precursors with a stabilized aziridine pathway and implementing a novel thermal buffering system during the critical carbene generation step, this technology offers a viable solution for the commercial supply chain. For R&D directors and procurement strategists, this represents a shift from laboratory curiosity to industrial reality, ensuring a consistent supply of high-purity intermediates essential for next-generation drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
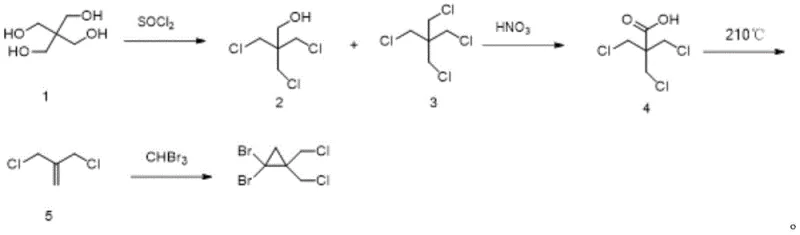
Historically, the synthesis of gem-dihalocyclopropanes required for bicyclic amine scaffolds has been plagued by severe operational hazards and poor scalability. The seminal work by Lynch and Daily, often cited as the standard reference, utilizes pentaerythritol as a starting material but necessitates a decarboxylation reaction at temperatures exceeding 210°C. Such extreme thermal conditions impose immense stress on reactor infrastructure and significantly increase energy consumption, rendering the process economically unviable for large-scale manufacturing. Furthermore, the final step involving the generation of dibromocarbene in sodium hydroxide solution is notoriously unstable; the reaction is characterized by violent, instantaneous heat release that frequently leads to material flushing and uncontrollable exotherms. These inherent safety risks create bottlenecks in production scheduling and pose unacceptable liabilities for contract development and manufacturing organizations (CDMOs) aiming for GMP compliance. The inability to effectively manage the thermal profile of the carbene addition step has traditionally resulted in low yields and inconsistent product quality, forcing supply chains to rely on small-batch, high-cost production models.
The Novel Approach
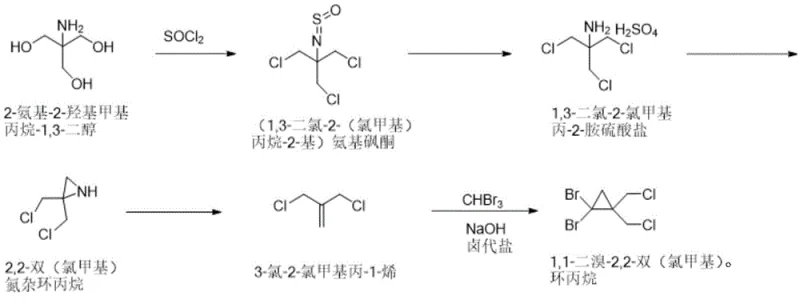
In stark contrast to the perilous legacy routes, the methodology disclosed in CN110759840B engineers safety directly into the reaction mechanism through a clever manipulation of nitrogen-containing intermediates. The process initiates with the chlorination of 2-amino-2-hydroxymethyl propane-1,3-diol using thionyl chloride, forming a stable aminosulfonone species that serves as a protected precursor. This intermediate undergoes controlled hydrolysis and subsequent alkaline ring closure to form 2,2-bis(chloromethyl)aziridine, a key structural pivot point that avoids the need for high-energy carboxylic acid derivatives. The subsequent conversion to the alkene via sodium nitrite reaction proceeds under mild conditions, setting the stage for the final cyclopropanation. Most critically, the invention introduces a halogenated salt buffer system, specifically utilizing potassium bromide, during the reaction with tribromomethane. This innovation acts as a thermal sink, moderating the exothermic nature of carbene generation and allowing the reaction to proceed within a narrow, safe temperature window of 18-25°C. This fundamental re-engineering of the reaction thermodynamics transforms a hazardous laboratory procedure into a robust, scalable industrial process capable of delivering consistent yields.
Mechanistic Insights into Aziridine-Mediated Cyclopropanation
The core mechanistic advantage of this synthesis lies in the strategic use of the aziridine ring as a latent source of the alkene functionality, bypassing the instability associated with direct diazo compound handling. The transformation of the amine sulfate salt into the aziridine involves a precise intramolecular nucleophilic substitution where the nitrogen lone pair displaces a chloride ion under carefully controlled pH conditions of 9-10. This ring-closing step is critical, as it establishes the carbon skeleton required for the subsequent elimination reaction. Upon treatment with sodium nitrite and hydrochloric acid, the aziridine undergoes a ring-opening elimination to yield 3-chloro-2-chloromethylprop-1-ene. This alkene is perfectly poised for the Simmons-Smith type cyclopropanation, yet the true genius of the process is observed in the final step. The generation of dibromocarbene from bromoform and base is typically a runaway reaction; however, the presence of the halogenated salt buffer modulates the activity of the hydroxide ions. This buffering effect prevents the localized accumulation of reactive carbene species, thereby minimizing side reactions such as polymerization or over-halogenation. Additionally, the inclusion of phase transfer catalysts like dibenzo-18-crown-6 facilitates the transport of the anionic carbene precursor into the organic phase, ensuring efficient contact with the alkene substrate and maximizing the atom economy of the transformation.
From an impurity control perspective, the multi-step nature of this synthesis allows for the purification of intermediates before the final critical bond-forming event. The initial chlorination and hydrolysis steps effectively remove oxygenated impurities, while the distillation of the alkene intermediate ensures that only the correct geometric isomer enters the cyclopropanation reactor. The use of pinacol as a co-catalyst further refines the reaction environment, likely stabilizing the transition state of the carbene addition and suppressing the formation of poly-halogenated byproducts. This rigorous control over the reaction trajectory results in a final product with exceptional purity profiles, often exceeding 98% as determined by HPLC analysis. For quality assurance teams, this means a significantly reduced burden on downstream purification processes, as the crude product emerging from the reactor is already of high pharmaceutical grade. The ability to crystallize the final product from n-heptane at low temperatures further enhances the rejection of trace impurities, ensuring that the material meets the stringent specifications required for clinical trial applications.
How to Synthesize 1,1-Dibromo-2,2-Bis(Chloromethyl)Cyclopropane Efficiently
The execution of this synthesis requires precise adherence to thermal gradients and stoichiometric ratios to maximize yield and safety. The process begins with the dissolution of the amino-diol in toluene, followed by the controlled addition of thionyl chloride at temperatures between 20-40°C to manage gas evolution. Subsequent gradient heating to 105-110°C ensures complete conversion to the aminosulfonone. The hydrolysis step demands careful temperature management below 25°C during acid addition to prevent degradation, followed by prolonged heating at 85-95°C to drive the reaction to completion. The ring closure to the aziridine is performed under alkaline conditions with strict pH monitoring, while the final carbene reaction utilizes a biphasic system with phase transfer catalysis. Detailed standardized operating procedures for each unit operation, including specific workup and crystallization protocols, are essential for reproducible manufacturing.
- Perform chlorine substitution on 2-amino-2-hydroxymethyl propane-1,3-diol with thionyl chloride to form the aminosulfonone intermediate.
- Hydrolyze the aminosulfonone with concentrated sulfuric acid to obtain the amine sulfate salt, followed by alkaline ring closure to form the aziridine.
- React the aziridine with sodium nitrite to generate the alkene, then perform the final carbene reaction with bromoform under buffered alkaline conditions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this synthesis route offers profound strategic benefits that extend far beyond simple unit cost calculations. The primary value driver is the substantial reduction in operational risk associated with the elimination of high-temperature processing and unstable intermediates. By removing the requirement for reactors capable of sustaining 210°C and managing violent exotherms, manufacturers can utilize standard glass-lined or stainless steel equipment that is more readily available and easier to maintain. This compatibility with existing infrastructure drastically reduces capital expenditure requirements for new production lines and accelerates the time-to-market for critical intermediates. Furthermore, the use of 2-amino-2-hydroxymethyl propane-1,3-diol as a starting material leverages a commodity chemical supply chain that is robust and price-stable, insulating the production cost from the volatility often seen with specialized fine chemical precursors. The overall simplification of the process flow, combined with the high selectivity of the reaction steps, leads to a significant reduction in waste generation and solvent consumption, aligning perfectly with modern sustainability mandates and reducing the environmental compliance burden.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of expensive and hazardous reagents typically associated with diazo chemistry. By substituting high-energy decarboxylation steps with mild acid-base transformations, the energy intensity of the manufacturing process is drastically lowered, resulting in direct utility cost savings. The high selectivity of the phase-transfer catalyzed carbene reaction minimizes the formation of difficult-to-remove byproducts, which in turn reduces the volume of solvents and adsorbents required for purification. This efficiency gain translates into a lower cost of goods sold (COGS) per kilogram of active intermediate, providing a competitive margin advantage in the global marketplace. Additionally, the ability to recycle toluene and other organic solvents used in the extraction phases further enhances the overall process economics, making it an attractive option for cost-sensitive generic drug programs.
- Enhanced Supply Chain Reliability: Supply continuity is often compromised by the safety incidents and regulatory shutdowns associated with hazardous chemical processes. The inherent safety features of this new route, particularly the thermal buffering system that prevents runaway reactions, ensure a predictable and uninterrupted production schedule. This reliability is crucial for pharmaceutical companies managing just-in-time inventory strategies for clinical trials and commercial launches. The use of widely available raw materials reduces the risk of supply disruptions caused by single-source vendor dependencies, creating a more resilient supply network. Moreover, the scalability of the process means that production volumes can be rapidly ramped up to meet surging demand without the need for complex process re-validation or extensive equipment modifications, providing agility in a dynamic market environment.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden complexities, but this methodology is explicitly designed for amplification. The controlled exotherm and moderate operating pressures allow for safe operation in large-volume reactors, mitigating the heat transfer limitations that often plague batch processes. From an environmental standpoint, the avoidance of heavy metal catalysts and the reduction in hazardous waste streams simplify the wastewater treatment requirements. The process generates fewer toxic byproducts, facilitating easier disposal and lowering the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only satisfies regulatory bodies but also enhances the corporate social responsibility profile of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a transparent view of the method's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own development pipelines. The answers reflect the consensus on safety, purity, and scalability established by the disclosed examples.
Q: What are the safety advantages of this new synthesis route compared to traditional methods?
A: The novel process eliminates the hazardous high-temperature decarboxylation step (210°C) required in prior art. Furthermore, it introduces a halogenated salt buffer system during the exothermic carbene generation, preventing violent thermal runaways and ensuring operational safety during scale-up.
Q: What is the purity profile of the final intermediate?
A: The optimized crystallization protocol yields 1,1-dibromo-2,2-bis(chloromethyl)cyclopropane with high purity specifications, typically exceeding 98% HPLC purity, which is critical for downstream pharmaceutical applications.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the method utilizes inexpensive, commercially available starting materials and avoids extreme reaction conditions. The implementation of phase transfer catalysis and thermal buffering makes the process robust and highly amenable to industrial amplification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Dibromo-2,2-Bis(Chloromethyl)Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant is seamless and efficient. We have rigorously validated the safety protocols associated with carbene chemistry and phase transfer catalysis, implementing stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. Our facility is equipped to handle the specific thermal and containment requirements of this synthesis, providing a secure environment for the manufacture of high-value intermediates.
We invite you to collaborate with us to optimize your supply chain for bicyclo[1.1.1]pentane derivatives. By leveraging our technical capabilities, you can secure a stable source of 1,1-dibromo-2,2-bis(chloromethyl)cyclopropane that supports your long-term development goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
