Scalable Synthesis of 3,3'-Bismaleimido-4,4'-bis(4-nitrophenoxy)biphenyl for High-Performance Resins
Scalable Synthesis of 3,3'-Bismaleimido-4,4'-bis(4-nitrophenoxy)biphenyl for High-Performance Resins
The development of advanced thermosetting materials for aerospace and electronics relies heavily on the availability of high-performance monomers with precise structural integrity. Patent CN101250150A introduces a robust and industrially viable method for preparing 3,3'-bismaleimido-4,4'-bis(4-nitrophenoxy)biphenyl, a critical building block for next-generation polyimide resins. This specific bismaleimide derivative is engineered to provide exceptional thermal stability and reactivity, making it an ideal candidate for modifying epoxy systems and creating high-heat-resistant composite matrices. The patent outlines a streamlined two-step synthetic route that avoids the complexities often associated with traditional imidization processes, ensuring consistent quality for demanding applications such as aircraft structural components and printed circuit board laminates.
For R&D directors and procurement specialists in the fine chemical sector, understanding the nuances of this synthesis is paramount for securing a reliable polymer additive supplier. The methodology described leverages common organic solvents and straightforward dehydration techniques to achieve yields ranging from 90.1% to 97.2%, demonstrating significant potential for cost reduction in advanced material manufacturing. By optimizing the molar ratios of reactants and selecting appropriate dehydrating agents, the process minimizes impurity formation while maximizing throughput. This technical insight serves as a foundation for evaluating the feasibility of integrating this monomer into existing supply chains for high-value electronic and aerospace applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for complex bismaleimide monomers often suffer from inefficient cyclization steps that require harsh conditions or expensive catalysts, leading to inconsistent product quality and lower overall yields. Many conventional methods rely on high-temperature thermal imidization in the solid state or the use of toxic dehydrating agents that complicate downstream purification and waste management. Furthermore, the lack of standardized protocols for solvent recovery in older processes results in elevated production costs and environmental liabilities, which are increasingly scrutinized by global regulatory bodies. These inefficiencies create bottlenecks in the commercial scale-up of complex polymer additives, limiting the availability of high-purity intermediates needed for critical electronic insulation materials.
The Novel Approach
The innovative strategy presented in the patent addresses these challenges by employing a mild, two-stage liquid-phase reaction that ensures complete conversion and easy isolation of the final product. By initiating the reaction at room temperature to form the amic acid intermediate, the process controls exothermic risks and prevents premature polymerization or side reactions that could compromise molecular weight distribution. The subsequent cyclization step offers flexibility through two distinct dehydration pathways—azeotropic removal of water or chemical dehydration using anhydride-amine systems—allowing manufacturers to select the most cost-effective option based on their existing infrastructure. This adaptability not only simplifies the operation technique but also facilitates the recycling of organic solvents, thereby aligning with modern green chemistry principles and reducing the total cost of ownership for the manufacturing process.
Mechanistic Insights into Imidization Cyclization
The core of this synthesis lies in the precise control of the nucleophilic attack of the diamine on the maleic anhydride, followed by a dehydration-driven ring closure to form the stable imide structure. In the first stage, the amino groups of 3,3'-diamino-4,4'-bis(4-nitrophenoxy)biphenyl react with the carbonyl carbons of maleic anhydride in polar aprotic solvents such as N-methyl-2-pyrrolidone (NMP) or N,N-dimethylacetamide (DMAc). This step forms the 3,3'-dicarboxycrotonamido intermediate, a crucial precursor that must remain in solution to ensure homogeneity before cyclization. The presence of electron-withdrawing nitro groups on the phenoxy rings influences the nucleophilicity of the amines, necessitating careful control of the molar ratio (1.0:2.0-2.2) to drive the reaction to completion without excessive excess of the anhydride.
The second stage involves the dehydration of the amic acid intermediate to close the five-membered imide ring, a transformation that is thermodynamically driven by the removal of water. As illustrated in the reaction scheme below, this can be achieved either by azeotropic distillation using solvents like toluene or xylene, which physically remove water as it forms, or by chemical dehydration using acetic anhydride and a tertiary amine catalyst. The chemical pathway activates the carboxylic acid group, facilitating intramolecular nucleophilic attack by the amide nitrogen at moderate temperatures (50°C-100°C). This mechanistic understanding is vital for troubleshooting potential issues with residual moisture or incomplete cyclization, which could adversely affect the thermal properties of the final polyimide resin.
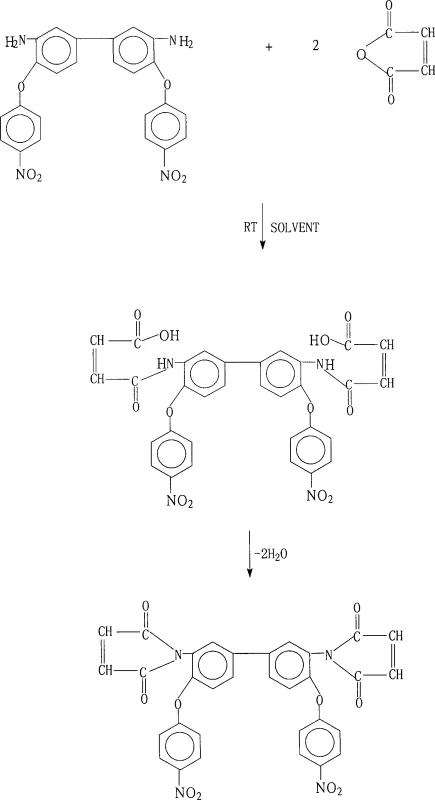
How to Synthesize 3,3'-Bismaleimido-4,4'-bis(4-nitrophenoxy)biphenyl Efficiently
Executing this synthesis requires strict adherence to the specified solvent systems and temperature profiles to ensure the high purity and yield reported in the patent data. The process begins with the dissolution of the diamine in a mixture of polar solvents, followed by the controlled addition of maleic anhydride to manage the exotherm and maintain a homogeneous reaction mixture. Detailed standard operating procedures for the dehydration step vary depending on whether an azeotropic or chemical method is chosen, but both require precise timing and filtration protocols to isolate the solid product effectively. For a comprehensive guide on the specific reagent quantities and workup procedures, please refer to the standardized synthesis steps outlined below.
- React 3,3'-diamino-4,4'-bis(4-nitrophenoxy)biphenyl with maleic anhydride (molar ratio 1: 2.0-2.2) in polar aprotic solvents like NMP or DMF at room temperature for 0.5-2 hours to form the amic acid intermediate.
- Add a dehydrating system: either an azeotropic agent like toluene/xylene with heating, or a chemical system using acetic anhydride and a tertiary amine catalyst at 50°C-100°C.
- Maintain the cyclization reaction for 2-18 hours, then cool, filter, wash the solid product with cold solvent or acetone, and dry to obtain the final bismaleimide monomer with yields exceeding 90%.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the operational simplicity of this patented method translates directly into enhanced reliability and reduced lead time for high-purity polymer additives. The ability to conduct the reaction under atmospheric pressure eliminates the need for specialized high-pressure vessels, lowering capital expenditure for new production lines and reducing maintenance costs for existing facilities. Moreover, the explicit inclusion of solvent recovery steps in the workflow allows manufacturers to recycle expensive polar aprotic solvents like NMP, which significantly lowers the variable cost per kilogram of the final product. This economic efficiency makes the material more competitive in price-sensitive markets such as consumer electronics and automotive composites, where margin pressures are constant.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the implementation of solvent recycling loops drastically reduce both CAPEX and OPEX associated with production. By recovering and reusing organic solvents, the process minimizes raw material waste and disposal fees, leading to substantial cost savings over the lifecycle of the manufacturing campaign. Additionally, the use of readily available starting materials like maleic anhydride and common industrial solvents ensures that supply chain disruptions are minimized, providing a stable cost base for long-term procurement contracts.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route, which tolerates slight variations in reaction time (2-18 hours) without compromising yield, ensures consistent output even in large-scale batch operations. This flexibility allows production planners to optimize reactor utilization and manage inventory levels more effectively, reducing the risk of stockouts for critical downstream customers. The high yields reported (consistently above 90%) mean that less raw material is required to produce the same amount of finished goods, further stabilizing the supply of this essential electronic chemical.
- Scalability and Environmental Compliance: The process is designed for industrial production, with straightforward workup steps involving filtration and washing that are easily automated in large reactors. The avoidance of heavy metal catalysts and the recovery of solvents align with increasingly strict environmental regulations, reducing the regulatory burden on the manufacturing site. This compliance advantage ensures uninterrupted production schedules and protects the brand reputation of companies sourcing these materials for sensitive applications like aerospace and medical devices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this bismaleimide monomer, based on the detailed specifications provided in the patent literature. Understanding these aspects helps stakeholders make informed decisions about integrating this material into their product formulations and supply chains. The answers reflect the practical realities of scaling this chemistry from the laboratory to commercial tonnage.
Q: What are the critical purity specifications for this bismaleimide monomer in electronic applications?
A: For high-performance electronic insulation and aerospace composites, the monomer requires stringent control over residual maleic anhydride and unreacted diamine. The patented process utilizes specific washing protocols with cold solvents and acetone soaking to ensure high purity suitable for FPC adhesives and PCB laminates.
Q: How does the solvent recovery process impact the overall production cost?
A: The process explicitly includes a step to concentrate the reaction solution and recover organic solvents like NMP and toluene for recycling. This significantly reduces raw material consumption and waste disposal costs, making the commercial scale-up economically viable compared to single-use solvent systems.
Q: Can this synthesis be performed without high-pressure equipment?
A: Yes, the entire reaction sequence operates under atmospheric pressure. The cyclization step utilizes reflux conditions or moderate heating (50°C-100°C), eliminating the need for expensive high-pressure autoclaves and enhancing operational safety for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Bismaleimido-4,4'-bis(4-nitrophenoxy)biphenyl Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate this patented laboratory method into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume demands of the global aerospace and electronics sectors. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,3'-bismaleimido-4,4'-bis(4-nitrophenoxy)biphenyl meets the exacting standards required for high-performance polyimide and epoxy resin applications.
We invite you to collaborate with us to optimize your material sourcing strategy and achieve significant efficiencies in your manufacturing operations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized production capabilities can support your next generation of advanced composite materials.
