Scaling High-Heat Bismaleimide Resin Production For Advanced Aerospace Composites
Scaling High-Heat Bismaleimide Resin Production For Advanced Aerospace Composites
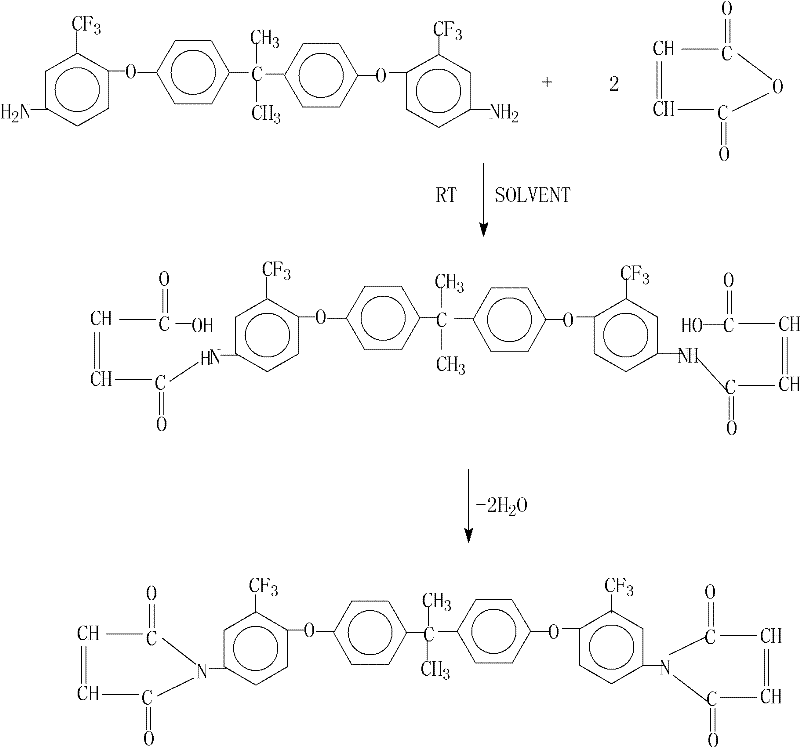
The relentless demand for materials capable of withstanding extreme thermal environments in the aerospace and electronics sectors has driven significant innovation in polymer chemistry. A pivotal development in this field is detailed in patent CN101955458B, which discloses a robust and highly efficient preparation method for 2,2-bis[4-(2-trifluoromethyl-4-maleimidophenoxy)phenyl]propane. This specific bismaleimide (BMI) resin represents a critical building block for next-generation advanced composite materials (ACM), offering superior thermal stability compared to traditional epoxy systems. For R&D directors and procurement strategists, understanding the nuances of this synthesis is vital, as it bridges the gap between laboratory-scale curiosity and viable industrial manufacturing. The patent outlines a pathway that not only achieves exceptional purity but also addresses the perennial challenges of yield optimization and waste reduction in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bismaleimide resins has been plagued by inefficiencies that hinder large-scale adoption. Traditional routes often suffer from incomplete cyclization of the bismaleamic acid intermediate, leading to products contaminated with unreacted precursors or isoimide byproducts. These impurities can severely compromise the thermal and mechanical performance of the final cured resin, a risk that aerospace manufacturers cannot afford. Furthermore, conventional dehydration methods frequently require harsh conditions or expensive catalysts that are difficult to remove, necessitating complex purification steps that drive up operational expenditures. Without effective water removal strategies, the equilibrium of the imidization reaction favors the reverse process, capping yields at suboptimal levels and generating significant volumes of aqueous waste that require treatment.
The Novel Approach
The methodology presented in the referenced patent introduces a sophisticated yet operationally simple solution to these bottlenecks. By employing a specific combination of anhydrous calcium chloride and N,N-dimethylaniline within an azeotropic distillation system, the process effectively drives the dehydration equilibrium toward the desired imide product. This approach allows the reaction to proceed under atmospheric pressure at moderate temperatures ranging from 100°C to 160°C, eliminating the need for costly high-pressure reactors. The result is a dramatic improvement in process reliability, where the reaction completes within a concise 2 to 3-hour window. This streamlined workflow not only simplifies the equipment requirements but also ensures a consistent product quality that meets the stringent specifications required for high-performance electronic and structural applications.
Mechanistic Insights into Calcium-Promoted Dehydrative Cyclization
The core chemical transformation in this process involves the conversion of the bismaleamic acid intermediate into the cyclic imide structure through a dehydration mechanism. The introduction of anhydrous calcium chloride serves a dual purpose: it acts as a Lewis acid to activate the carbonyl groups and simultaneously functions as a desiccant to sequester the water produced during cyclization. This synergistic effect lowers the activation energy for ring closure, facilitating a smoother transition state compared to thermal dehydration alone. The presence of N,N-dimethylaniline further enhances the reaction kinetics by acting as a basic catalyst, promoting the proton transfer steps necessary for the elimination of water. This precise orchestration of reagents ensures that the reaction proceeds to near-completion, minimizing the formation of open-chain acid impurities that could otherwise act as plasticizers or degradation points in the final polymer matrix.
From an impurity control perspective, the choice of solvent system and precipitation method is equally critical. The use of strong polar aprotic solvents like N,N-dimethylacetamide (DMAc) or N-methyl-2-pyrrolidone (NMP) in the initial amidation step ensures complete solubility of the fluorinated diamine and maleic anhydride, preventing localized concentration gradients that could lead to oligomerization. Subsequently, the addition of ethanol as an anti-solvent induces rapid crystallization of the target bismaleimide. This precipitation step is highly selective; the specific solubility profile of the trifluoromethyl-substituted product ensures that it crashes out of the solution while soluble byproducts and residual catalysts remain in the mother liquor. This inherent purification mechanism reduces the reliance on energy-intensive recrystallization or chromatography, directly contributing to a cleaner impurity profile and higher overall process efficiency.
How to Synthesize 2,2-bis[4-(2-trifluoromethyl-4-maleimidophenoxy)phenyl]propane Efficiently
The synthesis protocol described in the patent offers a clear roadmap for replicating this high-yield process in a pilot or production setting. The procedure begins with the formation of the bismaleamic acid precursor at room temperature, followed by a controlled heating phase for cyclization. Critical parameters such as the molar ratio of maleic anhydride to diamine (optimized at 2.0 to 2.1) and the specific weight ratios of the dehydration agents must be strictly adhered to in order to replicate the reported 99.6% yield. The following guide summarizes the standardized operational steps derived from the patent data, providing a foundation for process engineers to establish robust manufacturing protocols.
- React 2,2-bis[4-(2-trifluoromethyl-4-aminophenoxy)phenyl]propane with maleic anhydride in a polar aprotic solvent at room temperature to form the bismaleamic acid intermediate.
- Add anhydrous calcium chloride, an azeotropic dehydrating agent like toluene, and N,N-dimethylaniline to the reaction mixture.
- Heat the mixture to reflux for water separation, distill off the solvent, and precipitate the final product using ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the technical merits of this synthesis translate directly into tangible business value. The ability to produce high-purity bismaleimide resins with minimal waste generation addresses two of the most pressing concerns in modern chemical sourcing: cost volatility and environmental compliance. By optimizing the reaction stoichiometry and implementing solvent recovery loops, manufacturers can significantly reduce the cost of goods sold (COGS) without compromising on quality. This economic efficiency is further bolstered by the use of readily available raw materials, such as maleic anhydride and commercially sourced fluorinated diamines, which mitigates the risk of supply disruptions associated with exotic or proprietary reagents.
- Cost Reduction in Manufacturing: The implementation of this process eliminates the need for expensive high-pressure equipment and reduces energy consumption by operating at atmospheric pressure. Furthermore, the patent highlights the recovery and recycling of organic solvents and mother liquors, which drastically cuts down on raw material expenditure. By avoiding the use of corrosive substances and minimizing the generation of hazardous waste, facilities can also lower their operational overhead related to waste disposal and safety compliance, leading to substantial long-term savings.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions and the use of common industrial solvents like toluene and ethanol ensure that the supply chain remains resilient against market fluctuations. Since the process does not rely on sensitive transition metal catalysts that might be subject to geopolitical supply constraints, production continuity is greatly enhanced. The high yield reported in the patent examples indicates a robust process window, meaning that minor variations in feedstock quality are less likely to result in batch failures, thereby ensuring a steady and reliable flow of material to downstream customers.
- Scalability and Environmental Compliance: The described method is inherently scalable, having been designed with industrial production in mind. The absence of corrosive byproducts simplifies the selection of construction materials for reactors, allowing for easier scale-up from pilot plants to multi-ton production lines. Additionally, the closed-loop solvent recovery system aligns with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. This proactive approach to environmental stewardship not only future-proofs the manufacturing site against regulatory changes but also enhances the brand reputation of suppliers who prioritize sustainable chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized bismaleimide resin. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains.
Q: What is the primary advantage of using calcium chloride in this bismaleimide synthesis?
A: According to patent CN101955458B, the addition of anhydrous calcium chloride acts as a crucial dehydration promoter. Comparative examples show that omitting this specific salt combination significantly drops the yield from nearly quantitative levels (99.6%) to roughly 79%, proving its essential role in driving the cyclization equilibrium forward efficiently.
Q: Can the solvents used in this process be recycled for cost efficiency?
A: Yes, the process is designed for industrial sustainability. The patent explicitly details the recovery and recycling of the organic solvents and the mother liquor after filtration. This closed-loop capability drastically reduces raw material consumption and waste disposal costs, aligning with modern green chemistry standards.
Q: What are the thermal properties of the resulting resin?
A: The synthesized 2,2-bis[4-(2-trifluoromethyl-4-maleimidophenoxy)phenyl]propane exhibits a sharp melting point around 90.6°C and undergoes exothermic curing reactions above 260°C. These properties make it highly suitable for creating high-heat resistant thermosetting polyimides and epoxy modifiers for aerospace applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-bis[4-(2-trifluoromethyl-4-maleimidophenoxy)phenyl]propane Supplier
As the global demand for high-performance polymers continues to surge, partnering with a manufacturer that possesses deep technical expertise is essential for success. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch of 2,2-bis[4-(2-trifluoromethyl-4-maleimidophenoxy)phenyl]propane meets the exacting standards required for aerospace and electronic applications. We understand that consistency is key, and our state-of-the-art facilities are equipped to handle the complexities of fluorinated chemistry with precision and safety.
We invite you to collaborate with us to optimize your material sourcing strategy. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project goals. Let us help you secure a competitive advantage through superior material performance and supply chain reliability.
