Industrial Synthesis of Sodium Danshensu: Leveraging Dynamic Kinetic Resolution for Commercial Scale-Up
Industrial Synthesis of Sodium Danshensu: Leveraging Dynamic Kinetic Resolution for Commercial Scale-Up
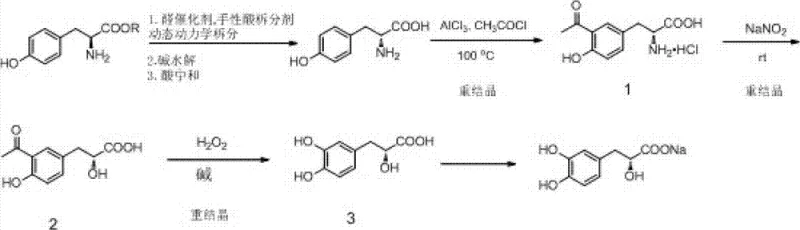
The pharmaceutical industry constantly seeks robust, scalable pathways for producing high-value chiral intermediates, particularly for cardiovascular therapeutics where stereochemical purity is non-negotiable. Patent CN103288630A presents a groundbreaking methodology for the synthesis of Sodium Danshensu (Salvianic acid A sodium), a water-soluble active component derived from Salvia miltiorrhiza. Unlike traditional extraction methods which suffer from low purity and complex isolation procedures, or previous synthetic routes reliant on expensive D-Tyrosine starting materials, this invention utilizes readily available L-Tyrosine ester. The core innovation lies in the application of Dynamic Kinetic Resolution (DKR) coupled with a streamlined sequence of Friedel-Crafts acylation, diazotization, and Baeyer-Villiger oxidation. This approach effectively bypasses the theoretical 50% yield limitation of classical resolution, offering a pathway that is both economically superior and technically feasible for multi-ton manufacturing.
For R&D directors evaluating process viability, the elimination of chromatographic purification stands out as a critical design feature. Conventional laboratory syntheses often rely on silica gel column chromatography to separate diastereomers or remove impurities, a technique that is notoriously difficult to translate to industrial scales due to solvent consumption and throughput bottlenecks. The patented process replaces these unit operations with crystallization-driven purifications. By leveraging the solubility differences of diastereomeric salts in anhydrous alcohol solvents during the resolution phase, and utilizing specific solvent systems like petroleum ether and ethyl acetate for subsequent intermediates, the protocol ensures high optical purity (ee > 99%) without the operational drag of column loading and elution. This structural simplicity in the workflow directly correlates to reduced capital expenditure on equipment and lower operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Salvianic acid A has been plagued by significant inefficiencies inherent to both natural extraction and early synthetic attempts. Extraction from plant sources, while conceptually direct, yields products with inconsistent chemical purity and requires extensive downstream processing to remove biological contaminants. On the synthetic front, earlier methodologies reported in literature, such as those by Bogucki and Charlton, relied on the conversion of L-Tyrosine to the target molecule but necessitated complex column chromatography separation steps to isolate the desired stereoisomer. Furthermore, alternative routes starting directly from D-Tyrosine face a severe economic barrier; D-Tyrosine is a non-natural amino acid that is significantly more expensive and less commercially available than its L-enantiomer counterpart. These factors combined create a supply chain vulnerability, where raw material costs fluctuate wildly and purification bottlenecks restrict the ability to meet large-scale demand for cardiovascular APIs.
The Novel Approach
The methodology outlined in CN103288630A fundamentally restructures the synthesis logic by inverting the chirality source. Instead of purchasing expensive D-Tyrosine, the process starts with abundant L-Tyrosine ester and employs a Dynamic Kinetic Resolution strategy. In this system, an aldehyde acts as a racemization agent while a chiral hydroxy acid serves as the resolving agent. This allows the unwanted L-isomer to continuously racemize back into the equilibrium mixture, where it can eventually be converted into the desired D-configured salt. Following hydrolysis to free D-Tyrosine, the route proceeds through a Friedel-Crafts acylation in nitrobenzene, followed by a diazotization reaction that converts the amino group into a hydroxyl group. The final transformation involves a Baeyer-Villiger oxidation using hydrogen peroxide under alkaline conditions. This sequence is not only chemically elegant but operationally robust, relying on standard industrial reagents like acetyl chloride, sodium nitrite, and hydrogen peroxide rather than exotic catalysts.
Mechanistic Insights into Dynamic Kinetic Resolution and Oxidation
The heart of this synthesis lies in the stereoselective generation of D-Tyrosine from L-Tyrosine ester. In a standard resolution, the maximum yield is capped at 50% because the unwanted enantiomer is discarded. However, the presence of an aldehyde racemization agent facilitates the reversible formation of a Schiff base with the amino acid ester. This imine intermediate allows for proton abstraction and reprotonation at the chiral center, effectively scrambling the stereochemistry. Simultaneously, the chiral resolving agent (such as D-tartaric acid or camphorsulfonic acid) selectively precipitates the D-Tyrosine ester salt due to lower solubility in the anhydrous alcohol solvent. As the D-salt crystallizes out, Le Chatelier's principle drives the equilibrium of the racemization reaction forward, theoretically allowing for nearly 100% conversion of the starting L-material into the D-product. This mechanism is crucial for cost reduction in pharmaceutical intermediate manufacturing, as it maximizes the utility of every kilogram of raw material input.
Following the acquisition of optically pure D-Tyrosine, the subsequent functionalization requires precise control to maintain stereochemical integrity. The Friedel-Crafts acylation introduces an acetyl group ortho to the phenolic hydroxyl, activated by aluminum chloride. Subsequently, the diazotization step converts the primary amine into a diazonium salt, which upon hydrolysis yields the alpha-hydroxy acid precursor. The final critical transformation is the Baeyer-Villiger oxidation. Here, the ketone moiety introduced in the acylation step is oxidized by hydrogen peroxide in a basic medium. The mechanism involves the nucleophilic attack of the hydroperoxide anion on the carbonyl carbon, followed by the migration of the aryl group to the oxygen atom. This rearrangement inserts an oxygen atom adjacent to the aromatic ring, establishing the characteristic 3,4-dihydroxyphenyl lactic acid skeleton of Salvianic acid. The use of 30% H2O2 is particularly advantageous from a safety and environmental perspective compared to peracids, generating water as the primary byproduct.
How to Synthesize Sodium Danshensu Efficiently
The synthesis of Sodium Danshensu via this patented route offers a clear blueprint for process chemists aiming to establish a GMP-compliant production line. The procedure begins with the DKR of L-Tyrosine ester in absolute ethanol at temperatures ranging from 40°C to 118°C, followed by hydrolysis to liberate D-Tyrosine. The subsequent acylation is performed in nitrobenzene at elevated temperatures (60°C to 150°C) to ensure complete conversion. The diazotization is conducted in aqueous media at controlled low temperatures to stabilize the diazonium intermediate before hydrolysis. Finally, the oxidation and salt formation steps are carried out under mild alkaline conditions. For a detailed breakdown of the specific molar ratios, solvent volumes, and crystallization parameters required to replicate this high-yield process, please refer to the standardized technical guide below.
- Perform Dynamic Kinetic Resolution on L-Tyrosine ester using an aldehyde racemization agent and chiral hydroxy acid resolving agent to obtain D-Tyrosine.
- Conduct Friedel-Crafts acylation on D-Tyrosine with acetyl chloride and aluminum chloride to form the acylated intermediate.
- Execute diazotization followed by Baeyer-Villiger oxidation using hydrogen peroxide under alkaline conditions to generate Salvianic Acid A.
- Finalize the process by reacting Salvianic Acid A with a base in alcoholic solvent to crystallize pure Sodium Danshensu.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the shift from extraction or D-Tyrosine-based synthesis to this L-Tyrosine DKR route represents a substantial strategic advantage. The primary driver of value is the raw material cost differential; L-Tyrosine is a commodity chemical produced in massive quantities for the food and feed industries, whereas D-Tyrosine is a niche fine chemical with limited suppliers and premium pricing. By inverting the chirality in-house, manufacturers decouple their production costs from the volatile market dynamics of non-natural amino acids. Furthermore, the reliance on crystallization rather than chromatography drastically reduces solvent consumption and waste generation. This simplification of the purification train means that batch cycles are shorter, equipment turnover is faster, and the overall facility throughput is significantly enhanced without requiring additional capital investment in separation columns.
- Cost Reduction in Manufacturing: The elimination of expensive D-Tyrosine starting materials creates an immediate and significant reduction in the Bill of Materials (BOM) cost. Additionally, the recycling of the resolving agent and racemization agent, which can be recovered from the mother liquor after crystallization, further drives down variable costs. The process avoids the use of precious metal catalysts or specialized enzymes that often require strict temperature controls and have limited shelf-lives, replacing them with robust, shelf-stable reagents like aluminum chloride and sodium nitrite. This transition from biotech-dependent or extraction-dependent models to a purely chemical synthesis model stabilizes the cost structure and improves margin predictability for long-term contracts.
- Enhanced Supply Chain Reliability: Relying on agricultural extraction for key intermediates introduces risks related to crop yields, seasonal variations, and geographical concentration of raw materials. This synthetic route mitigates those risks by utilizing petrochemical-derived or fermentation-derived L-Tyrosine, which has a diversified and resilient global supply base. The robustness of the chemical steps, which tolerate a wider range of operating conditions compared to enzymatic processes, ensures consistent production schedules even in varying environmental conditions. This reliability is critical for pharmaceutical customers who require Just-In-Time delivery to maintain their own API production schedules without interruption.
- Scalability and Environmental Compliance: The absence of column chromatography is a major enabler for scalability. Chromatography is often the bottleneck in scaling organic synthesis due to the large volumes of silica and solvents required. By designing the process around crystallization and liquid-liquid extraction, the technology is inherently ready for transfer from pilot plant to commercial scale reactors. Moreover, the use of hydrogen peroxide as the oxidant aligns with green chemistry principles, as it decomposes into water and oxygen, reducing the burden on wastewater treatment facilities. The simplified waste stream, devoid of silica gel and complex organic eluents, facilitates easier compliance with increasingly stringent environmental regulations regarding hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific operational parameters and benefits detailed in the patent documentation, providing clarity on purity profiles, reaction mechanisms, and scalability factors. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: Why is Dynamic Kinetic Resolution preferred over traditional resolution for D-Tyrosine production?
A: Traditional resolution is limited to a theoretical maximum yield of 50% for the desired enantiomer. Dynamic Kinetic Resolution (DKR) continuously racemizes the unwanted isomer in situ, allowing for theoretical yields approaching 100% and significantly improving atom economy and cost efficiency.
Q: Does this synthesis method require column chromatography for purification?
A: No. A key advantage of the patented process (CN103288630A) is that all purification steps are achieved through simple recrystallization and extraction. This eliminates the need for column chromatography, making the process highly suitable for industrial mass production.
Q: What are the critical reaction conditions for the Baeyer-Villiger oxidation step?
A: The oxidation utilizes 30% hydrogen peroxide in an alkaline aqueous solution. Temperature control is critical, typically ranging from -20°C to 60°C, with preferred embodiments operating between 30°C and 50°C to ensure high conversion while minimizing side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium Danshensu Supplier
The technological advancements detailed in patent CN103288630A underscore the potential for high-efficiency manufacturing of cardiovascular intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such complex laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to reactor is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped to verify optical purity and impurity profiles, guaranteeing that every batch of Sodium Danshensu meets the exacting standards required for pharmaceutical applications.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis capabilities, we can help you secure a stable supply of high-purity intermediates while reducing overall project costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming projects.
