Advanced Microwave-Assisted Synthesis of High-Purity Polyhydroxy Flavones for Commercial Scale-Up
Advanced Microwave-Assisted Synthesis of High-Purity Polyhydroxy Flavones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally sustainable pathways for synthesizing bioactive scaffolds. A pivotal advancement in this domain is detailed in Chinese Patent CN102558127B, which discloses a highly efficient method for synthesizing polyhydroxy flavone compounds. These compounds, characterized by their multiple hydroxyl groups on the flavone backbone, exhibit potent pharmacological properties including antiviral, antibacterial, antitumor, and antioxidant activities, making them invaluable candidates for drug development and nutraceutical applications. The patent outlines a strategic four-step synthesis that leverages microwave irradiation to overcome the kinetic barriers traditionally associated with flavone ring closure. By integrating modern process intensification techniques with classic organic transformations, this technology offers a compelling solution for producing high-purity intermediates with superior yield profiles.
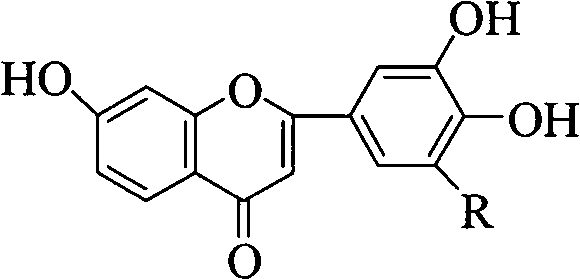
This technological breakthrough addresses the critical need for reliable pharmaceutical intermediate supplier capabilities that can deliver complex heterocyclic structures without the prohibitive costs often associated with multi-step natural product synthesis. The core innovation lies in the cyclization step, where microwave energy replaces conventional conductive heating, drastically reducing reaction times and energy consumption while maintaining exceptional product integrity. For R&D directors and process chemists, understanding the nuances of this pathway is essential for evaluating its potential integration into existing manufacturing pipelines for cardiovascular and oncology therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of flavonoids has relied on methods such as the Allan-Robinson condensation, Baker-Venkataraman rearrangement, or various cyclization protocols using iodine or strong organic bases like DBU. While effective on a laboratory scale, these conventional approaches often suffer from significant drawbacks when translated to commercial production. Traditional thermal cyclization frequently requires prolonged heating periods, sometimes extending over many hours, which not only consumes substantial energy but also increases the risk of thermal degradation of sensitive functional groups. Furthermore, many classical methods necessitate the use of harsh acidic or basic conditions that generate large volumes of hazardous waste, complicating downstream processing and environmental compliance. The reliance on stoichiometric amounts of expensive reagents or transition metals that are difficult to remove can also compromise the purity profile of the final API intermediate, necessitating costly and time-consuming purification steps such as column chromatography which are impractical at scale.
The Novel Approach
In stark contrast, the methodology presented in patent CN102558127B introduces a streamlined, microwave-assisted protocol that fundamentally reshapes the efficiency of flavone construction. The process begins with the esterification of substituted benzoyl chlorides with hydroxyacetophenones, followed by a Fries rearrangement to generate the critical 1,3-diketone precursor. The true game-changer, however, is the subsequent cyclization step. By subjecting the 1,3-diketone intermediate to microwave irradiation in the presence of a Lewis acid catalyst, the reaction proceeds to completion in merely 5 to 30 minutes at moderate temperatures of 50 to 80°C. This represents a paradigm shift in cost reduction in fine chemical manufacturing, as it eliminates the need for extended reflux times and significantly lowers the thermal load on the reactor system. The use of ethanol as a solvent for this key step further enhances the green chemistry profile, replacing more toxic alternatives and simplifying solvent recovery.
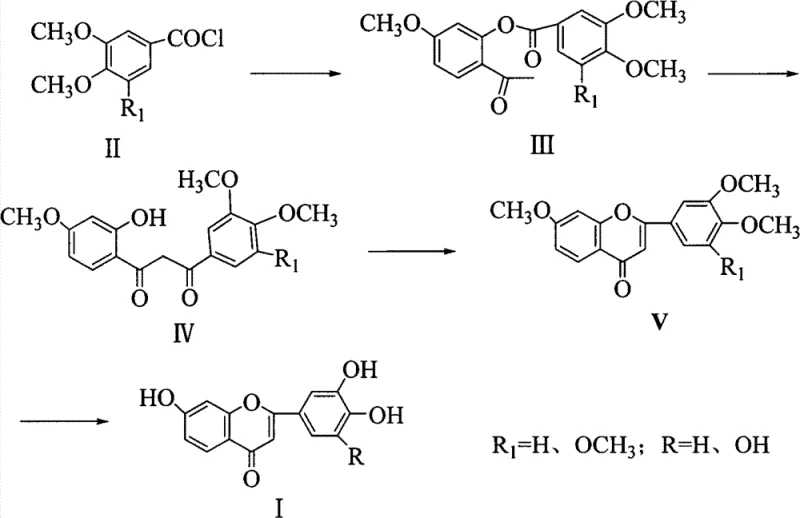
The visual representation of this synthetic pathway highlights the logical progression from simple aromatic starting materials to the complex polyhydroxy flavone core. The ability to toggle the substitution pattern (R=H or OH) allows for the versatile production of various analogues, such as 7,3',4'-trihydroxyflavone or 7,3',4',5'-tetrahydroxyflavone, catering to diverse structure-activity relationship (SAR) studies. This modularity ensures that the process remains relevant for the commercial scale-up of complex polymer additives or pharmaceutical intermediates requiring specific substitution patterns for optimal biological activity.
Mechanistic Insights into Lewis Acid-Catalyzed Microwave Cyclization
The efficacy of this synthesis hinges on the synergistic interaction between the Lewis acid catalyst and microwave dielectric heating during the cyclization of the 1,3-diketone intermediate. In this mechanism, metal chlorides such as CuCl2, ZnCl2, or FeCl2 act as Lewis acids to coordinate with the carbonyl oxygen atoms of the diketone. This coordination increases the electrophilicity of the carbonyl carbon, thereby facilitating the intramolecular nucleophilic attack by the phenolic hydroxyl group. Under conventional heating, this activation energy barrier often requires high temperatures and long durations to overcome. However, microwave irradiation interacts directly with the polar molecules and ions in the reaction mixture, generating heat internally and instantaneously. This "molecular friction" effect ensures that the activation energy is reached uniformly throughout the reaction volume, preventing the formation of hot spots that could lead to byproduct formation.
Furthermore, the specific choice of catalyst concentration, typically ranging from 0.05 to 0.1 mol/L, is critical for balancing reaction rate and impurity control. The patent data indicates that this optimized catalytic loading, combined with the rapid energy transfer of microwaves, suppresses side reactions such as polymerization or decomposition that are common in prolonged thermal treatments. The result is a crude product with significantly higher purity, reducing the burden on the final recrystallization steps. For quality assurance teams, this mechanistic robustness translates to a more consistent impurity profile, which is a prerequisite for regulatory approval in pharmaceutical applications. The final demethylation step using hydrobromic acid in acetic acid then cleanly reveals the free hydroxyl groups, completing the transformation to the bioactive polyhydroxy species with high fidelity.
How to Synthesize Polyhydroxy Flavones Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of microwave assistance. The process is divided into four distinct operational stages: ester formation, rearrangement to the diketone, microwave-assisted cyclization, and final hydrolysis. Each stage has been optimized in the patent examples to demonstrate reproducibility and high yield, providing a clear roadmap for process engineers. The following guide summarizes the critical operational windows derived from the intellectual property, ensuring that the transition from benchtop to pilot plant maintains the integrity of the chemical transformations.
- React 3,4-dimethoxy-5-substituted benzoyl chloride with 2-hydroxy-4-methoxyacetophenone in anhydrous pyridine at 60-80°C to form the diaryl ester intermediate.
- Perform a rearrangement reaction using potassium hydroxide or sodium hydroxide in pyridine at 80-115°C to generate the 1,3-disubstituted propanedione compound.
- Execute the critical cyclization step using a Lewis acid catalyst (e.g., CuCl2, ZnCl2) under microwave irradiation at 50-80°C for 5-30 minutes to form the methoxy flavone core.
- Conduct final demethylation using hydrobromic acid in glacial acetic acid at 100-110°C to yield the target polyhydroxy flavone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microwave-assisted technology offers tangible strategic advantages beyond mere technical elegance. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply security. By shortening the cycle time of the most critical step—the ring closure—the overall throughput of the production facility is significantly increased without the need for additional capital investment in reactor volume. This efficiency gain allows for more flexible production scheduling and faster response times to market demand fluctuations, a critical factor in the volatile landscape of active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of energy-intensive long-duration heating and the reduction of solvent usage. Traditional methods often require liters of solvent per kilogram of product and hours of reflux, whereas the microwave method operates with reduced solvent volumes and minute-scale reaction times. Additionally, the use of inexpensive and readily available Lewis acid catalysts like zinc chloride or copper chloride avoids the high costs associated with precious metal catalysts such as palladium or platinum. This substitution results in substantial cost savings on raw materials and simplifies the waste treatment process, as the removal of base metal residues is generally less complex and costly than removing noble metals, thereby lowering the overall cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as starting materials. The precursors, such as substituted benzoyl chlorides and hydroxyacetophenones, are widely available from multiple global suppliers, mitigating the risk of single-source bottlenecks. Furthermore, the robustness of the microwave cyclization step means that the process is less susceptible to variations in raw material quality compared to more sensitive catalytic systems. This reliability ensures consistent delivery schedules for downstream customers, reducing the risk of production stoppages due to intermediate shortages. The simplified workup procedures also mean that the lead time from reaction completion to finished goods is shortened, enabling a more agile inventory management strategy.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this process offers a distinct advantage through its green chemistry credentials. The replacement of hazardous solvents with ethanol in the cyclization step and the minimization of waste acid generation align with modern environmental, social, and governance (ESG) goals. Industrial microwave reactors have advanced significantly, allowing for the seamless scale-up of batch processes to continuous flow or large-batch modes suitable for multi-ton production. The high atom economy and reduced waste stream lower the costs associated with effluent treatment and disposal, ensuring long-term operational viability in regions with strict environmental regulations. This makes the technology not just a laboratory curiosity but a viable solution for the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microwave-assisted synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the technology's capabilities and limitations for potential partners and licensees.
Q: How does microwave assistance improve the cyclization step compared to traditional heating?
A: Microwave irradiation provides rapid, uniform internal heating which significantly accelerates the cyclization of 1,3-diketones. This method reduces reaction time from several hours to mere minutes while improving selectivity and yield, avoiding the prolonged thermal stress associated with conventional oil-bath heating.
Q: What catalysts are suitable for this microwave-assisted flavone synthesis?
A: The patent specifies the use of transition metal chlorides such as Copper(II) chloride (CuCl2), Zinc chloride (ZnCl2), Iron(II) chloride (FeCl2), or Manganese(II) chloride (MnCl2). These Lewis acids effectively promote the ring-closure reaction under microwave conditions with high efficiency.
Q: Is this synthesis route scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It utilizes readily available raw materials and avoids complex purification steps. The use of ethanol as a solvent for the cyclization step and the elimination of large amounts of waste acid compared to traditional methods make it environmentally compliant and suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyhydroxy Flavone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the microwave-assisted synthesis described in CN102558127B for the production of high-value flavonoid intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies demonstrated at the gram scale are faithfully reproduced in multi-ton campaigns. Our state-of-the-art facilities are equipped with advanced microwave reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We understand that the transition to a new synthetic route requires confidence in both the chemistry and the manufacturer, and our track record in delivering complex organic molecules underscores our commitment to quality and reliability.
We invite pharmaceutical and agrochemical companies to explore how this innovative technology can optimize their supply chains and reduce manufacturing costs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and samples for your evaluation. By partnering with us, you gain access to a secure, scalable, and cost-effective source of high-purity polyhydroxy flavones, positioning your organization at the forefront of therapeutic innovation.
