Advanced Electrochemical Synthesis of Trifluoromethylated Aryl Amides for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking innovative methodologies to introduce trifluoromethyl groups into organic scaffolds, given the profound impact of the CF3 moiety on the lipophilicity, metabolic stability, and bioavailability of drug candidates. Patent CN111690947A presents a groundbreaking electrochemical synthesis method for trifluoromethylated aryl amide derivatives that addresses critical limitations in current manufacturing practices. This technology leverages constant current electrolysis to achieve direct C-H functionalization, bypassing the need for pre-functionalized substrates or harsh chemical oxidants. By utilizing electrons as a traceless reagent, this process aligns perfectly with the principles of green chemistry, offering a sustainable pathway for producing high-value intermediates used in the synthesis of blockbuster drugs such as Celebrex and Januvia. For R&D directors and procurement specialists, understanding this electrochemical paradigm shift is essential for optimizing supply chains and reducing the environmental footprint of complex molecule production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for the trifluoromethylation of aryl amines and amides have historically relied heavily on transition metal catalysis, often employing nickel, copper, or iron complexes alongside stoichiometric amounts of strong chemical oxidants like potassium persulfate. These conventional protocols present significant challenges for large-scale manufacturing, primarily due to the toxicity and high cost of the metal catalysts involved. The presence of residual heavy metals in the final product necessitates rigorous and expensive purification steps to meet stringent pharmaceutical quality standards, which drastically increases production costs and extends lead times. Furthermore, the use of large quantities of chemical oxidants generates substantial amounts of hazardous waste, creating environmental compliance burdens and disposal costs that erode profit margins. The requirement for pre-functionalized substrates in some older methods also adds extra synthetic steps, reducing overall atom economy and process efficiency.
The Novel Approach
In stark contrast, the electrochemical method disclosed in CN111690947A offers a transformative solution by replacing chemical oxidants and metal catalysts with electrical energy. This novel approach operates under mild conditions, typically at temperatures between 25°C and 75°C, using a simple undivided cell setup with a carbon anode and a platinum cathode. The reaction proceeds via anodic oxidation to generate reactive radical species in situ, which then selectively functionalize the ortho-position of the aryl amide directed by the picolinamide or quinoline group. This metal-free strategy not only eliminates the risk of heavy metal contamination but also simplifies the downstream processing workflow significantly. The absence of stoichiometric oxidants means less waste generation and a cleaner reaction profile, allowing for higher isolated yields ranging from 60% to 80% across a diverse array of substrates. This represents a substantial leap forward in process intensification and sustainability for the production of fluorinated intermediates.
Mechanistic Insights into Electrochemical C-H Functionalization
The core mechanism of this synthesis involves the anodic oxidation of the trifluoromethanesulfinate salt (Langlois reagent) to generate a trifluoromethyl radical species. Under the applied constant current of 5-20 mA, the electrolyte facilitates the transfer of electrons at the electrode surface, initiating the radical chain reaction without the need for external photo-catalysts or thermal initiators. The picolinamide or quinoline moiety on the substrate acts as a powerful directing group, coordinating with the transient species to ensure high regioselectivity for the ortho-C-H bond activation. This directed C-H functionalization is critical for maintaining structural integrity and avoiding the formation of difficult-to-separate isomers, which is a common pain point in aromatic substitution reactions. The mild electrochemical potential prevents the over-oxidation of sensitive functional groups, thereby preserving the chemical diversity required for late-stage functionalization in drug discovery campaigns.
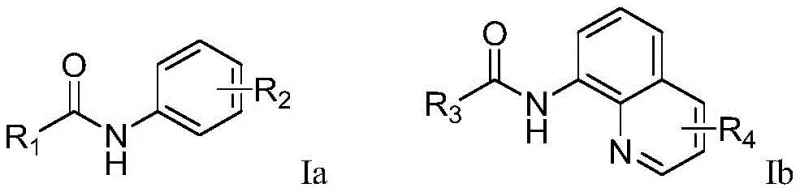
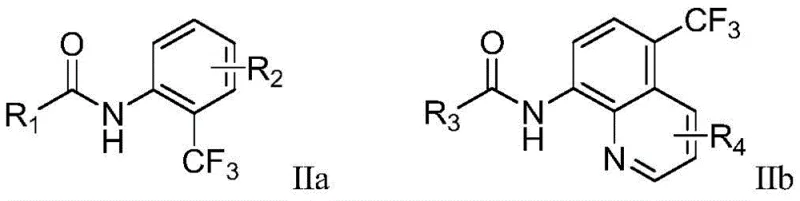
Furthermore, the electrochemical environment provides precise control over the reaction kinetics, allowing for the fine-tuning of radical concentration to minimize side reactions such as homocoupling or polymerization. The use of tetrabutylammonium salts as supporting electrolytes ensures high conductivity in organic solvents like acetonitrile, facilitating efficient electron transfer throughout the reaction medium. This mechanistic elegance translates directly into operational robustness, as the system is less sensitive to oxygen and moisture compared to many transition-metal-catalyzed alternatives. For process chemists, this means reduced requirements for inert atmosphere handling and specialized equipment, lowering the barrier to entry for implementing this technology in existing manufacturing facilities. The ability to achieve high conversion rates with minimal byproduct formation underscores the superiority of this electrochemical pathway for generating high-purity pharmaceutical intermediates.
How to Synthesize Trifluoromethylated Aryl Amides Efficiently
The practical implementation of this electrochemical synthesis is straightforward and highly reproducible, making it an attractive option for both laboratory-scale optimization and pilot plant operations. The standard protocol involves charging a reaction vessel equipped with a carbon anode and a platinum cathode with the aryl amide substrate, sodium trifluoromethanesulfinate, and a quaternary ammonium electrolyte in acetonitrile. The mixture is then subjected to constant current electrolysis at 50°C for approximately 120 minutes, after which the product is isolated via standard workup procedures including concentration and silica gel chromatography. This streamlined workflow eliminates the need for complex catalyst preparation or ligand screening, significantly reducing the technical expertise required to execute the transformation. Detailed standardized synthesis steps for specific derivatives are outlined in the guide below to assist technical teams in replicating these results.
- Prepare the reaction mixture by combining the aryl amide substrate, sodium trifluoromethanesulfinate (1.5 equivalents), and tetrabutylammonium tetrafluoroborate electrolyte in acetonitrile solvent within an undivided cell equipped with carbon anode and platinum cathode.
- Apply a constant current of 15 mA to the reaction system while maintaining the temperature at 50°C with continuous stirring for a duration of 120 minutes to facilitate the electrochemical C-H functionalization.
- Upon completion, concentrate the reaction mixture under reduced pressure and purify the crude residue via silica gel column chromatography using a petroleum ether and ethyl acetate eluent system to isolate the high-purity trifluoromethylated product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical technology offers compelling advantages for procurement managers and supply chain heads focused on cost reduction in pharmaceutical manufacturing. By eliminating the reliance on precious or toxic metal catalysts, manufacturers can achieve significant cost savings associated with raw material procurement and waste disposal. The removal of heavy metals from the process flow also mitigates the risk of supply chain disruptions caused by the volatility of metal markets and regulatory restrictions on metal residues in APIs. Additionally, the simplified purification process reduces the consumption of silica gel and solvents during chromatography, further driving down the cost of goods sold. The mild reaction conditions and use of commercially available reagents ensure a stable and reliable supply of key starting materials, enhancing overall supply chain resilience against geopolitical or logistical shocks.
- Cost Reduction in Manufacturing: The exclusion of expensive transition metal catalysts and stoichiometric oxidants fundamentally alters the cost structure of producing trifluoromethylated intermediates. Without the need for costly metal scavengers or extensive purification to remove metal traces, the downstream processing becomes significantly more economical. This leaner manufacturing approach allows for substantial cost savings that can be passed down the supply chain, improving margin profiles for both intermediate suppliers and API manufacturers. The efficiency of the electrochemical cell also means lower energy consumption per unit of product compared to high-temperature thermal processes, contributing to a more sustainable and cost-effective operation.
- Enhanced Supply Chain Reliability: The reagents required for this electrochemical method, such as sodium trifluoromethanesulfinate and tetrabutylammonium salts, are commodity chemicals with robust global supply chains. This contrasts sharply with specialized ligands or custom-synthesized catalysts that may have single-source suppliers and long lead times. By relying on widely available materials, manufacturers can secure long-term contracts and maintain consistent inventory levels, ensuring uninterrupted production schedules. The simplicity of the equipment setup, requiring only standard power supplies and electrode materials, further reduces the risk of technical downtime and maintenance issues, guaranteeing a steady flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable, particularly when adapted to flow chemistry systems, allowing for seamless transition from gram-scale discovery to multi-ton commercial production. The green nature of this synthesis, characterized by the absence of hazardous oxidants and heavy metals, aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. This compliance reduces the regulatory burden and potential fines associated with waste management, while also enhancing the brand reputation of companies adopting green chemistry principles. The ability to scale up complex fluorination reactions without compromising safety or purity makes this technology a strategic asset for expanding production capacity in a responsible manner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of trifluoromethylated aryl amides. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity on the feasibility and advantages of this method for industrial application. Understanding these details is crucial for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of this electrochemical method over traditional metal-catalyzed trifluoromethylation?
A: Unlike conventional methods that rely on toxic heavy metal catalysts like nickel or copper and stoichiometric chemical oxidants, this electrochemical approach utilizes electrons as the reagent. This eliminates the need for expensive metal removal steps, reduces hazardous waste generation, and simplifies the purification process, resulting in a greener and more cost-effective manufacturing route for pharmaceutical intermediates.
Q: What are the optimal reaction conditions described in the patent for this synthesis?
A: The patent specifies optimal conditions involving a constant current of 15 mA applied at a moderate temperature of 50°C for approximately 120 minutes. The reaction employs a carbon anode and a platinum plate cathode in an acetonitrile solvent system with tetrabutylammonium salts as the supporting electrolyte, ensuring high conversion rates and excellent regioselectivity for the ortho-trifluoromethylated products.
Q: Is this synthetic route applicable to a wide range of substrate structures?
A: Yes, the methodology demonstrates broad substrate scope, successfully accommodating various N-aryl picolinamides and N-quinolinyl benzamides. It tolerates diverse functional groups including halogens, alkyls, cyano, and alkoxy groups on the aromatic rings, making it a versatile platform for synthesizing complex trifluoromethylated building blocks required in modern drug discovery and agrochemical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylated Aryl Amide Supplier
As the demand for fluorinated pharmaceutical intermediates continues to surge, partnering with a technically proficient CDMO like NINGBO INNO PHARMCHEM is essential for securing a competitive edge. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped to handle electrochemical synthesis and other advanced transformations, adhering to stringent purity specifications and rigorous QC labs to deliver materials that meet the highest global standards. We understand the critical nature of supply continuity and are committed to providing reliable support for your drug development programs.
We invite you to contact our technical procurement team to discuss how we can tailor this electrochemical technology to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free process for your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-purity trifluoromethylated aryl amides efficiently. Let us collaborate to optimize your supply chain and accelerate your path to market with superior chemical solutions.
