Advanced Grignard-Based Synthesis of C11 Aldehyde for Scalable Vitamin A Acetate Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways for producing critical vitamin intermediates, and patent CN111484510B introduces a transformative method for synthesizing (2, 6-trimethyl-1-cyclohexenyl) acetaldehyde, commonly known as C11 aldehyde. This specific aldehyde serves as a pivotal building block in the manufacture of C14 aldehyde and ultimately Vitamin A acetate, a compound essential for human health and widely used in medicine, food, and feed additives. The disclosed technology leverages a robust Grignard reaction strategy that fundamentally shifts the production paradigm away from hazardous and complex legacy processes. By utilizing halogenated ethanol acetals and magnesium powder, the invention establishes a direct, high-yield route that addresses long-standing issues regarding safety, cost, and environmental impact in the supply chain of fat-soluble vitamins.
Historically, the synthesis of C11 aldehyde and its downstream derivatives has been plagued by inefficient multi-step sequences and dangerous reaction conditions. Traditional approaches, such as the beta-ionone-chloroacetate method depicted in older synthetic routes, rely on Darzens condensation which generates substantial wastewater and suffers from poor atom economy. Furthermore, the trimethylcyclohexanone-acetylene method, while capable of high yields, necessitates the use of lithium amide and acetylene gas under ultra-low temperature conditions, creating severe safety hazards and operational complexities.
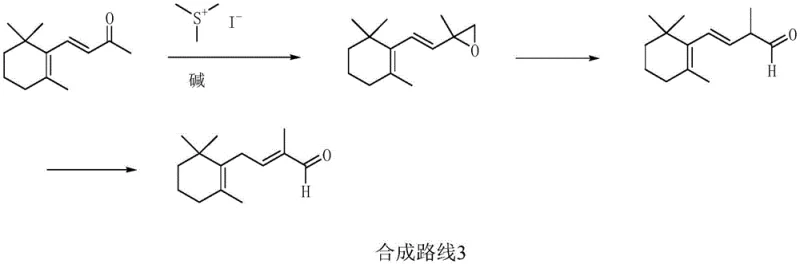
In stark contrast, the novel approach detailed in the patent simplifies the molecular construction by employing a Grignard reagent derived from readily available haloacetals. This method eliminates the need for cryogenic operations and explosive gases, replacing them with a controlled addition reaction between the Grignard species and 2,6-trimethylcyclohexanone. The process flow is significantly streamlined, reducing the number of unit operations and avoiding the formation of difficult-to-separate isomers that often plague rearrangement-based syntheses. This shift not only enhances the purity profile of the intermediate but also drastically reduces the capital investment required for specialized low-temperature reactors and safety containment systems.
Mechanistic Insights into Grignard Addition and Deprotection
The core of this technological advancement lies in the precise formation and utilization of the Grignard reagent from a protected haloacetal precursor. In the initial step, magnesium powder reacts with a compound of Formula II, such as bromoacetaldehyde dimethyl acetal, in a solvent system like tetrahydrofuran. The presence of activating agents like iodine ensures the rapid initiation of the organometallic species at moderate temperatures ranging from 20°C to 70°C. This controlled formation prevents the decomposition of the sensitive Grignard reagent, a common failure point in less optimized processes, ensuring a high concentration of the active nucleophile is available for the subsequent coupling step.
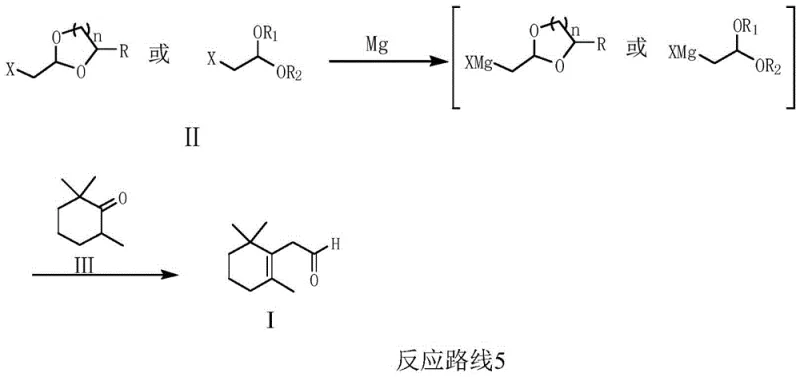
Following the generation of the Grignard reagent, the mechanism proceeds via a nucleophilic addition to the carbonyl group of 2,6-trimethylcyclohexanone (Formula III). This addition occurs efficiently at temperatures between 0°C and 40°C, forming an intermediate alkoxide. The final transformation involves a critical acidification and deprotection sequence where the acetal protecting group is hydrolyzed under acidic conditions (pH 1.0-4.0). This one-pot deprotection reveals the aldehyde functionality while simultaneously driving the dehydration or rearrangement necessary to form the conjugated double bond system of the C11 aldehyde. The high selectivity of this mechanism minimizes side reactions, resulting in a product with gas phase purity exceeding 99% and molar yields reaching up to 93.7%.
How to Synthesize (2, 6-trimethyl-1-cyclohexenyl) acetaldehyde Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing strict temperature control and reagent stoichiometry to maximize efficiency. The process begins with the activation of magnesium and the slow addition of the haloacetal to maintain thermal stability, followed by the controlled dosing of the ketone substrate. Detailed standard operating procedures regarding solvent recovery and aqueous workup are essential to maintain the high purity specifications required for pharmaceutical intermediates. For the complete standardized synthesis steps and specific reaction parameters, please refer to the guide below.
- Prepare the Grignard reagent by reacting a halogenated ethanol acetal with magnesium powder in a solvent like THF at 20-70°C.
- Add 2,6-trimethylcyclohexanone to the Grignard reagent at 0-40°C to facilitate the nucleophilic addition reaction.
- Perform acidification and deprotection using an acid reagent to obtain the final (2, 6-trimethyl-1-cyclohexenyl) acetaldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Grignard-based synthesis route offers profound strategic benefits beyond mere technical feasibility. The transition from hazardous acetylene chemistry to a stable Grignard process fundamentally alters the risk profile of the manufacturing site, reducing insurance liabilities and safety compliance costs. Moreover, the reliance on commodity chemicals like magnesium and simple haloacetals insulates the production cost from the volatility associated with specialized reagents like lithium amide. This stability in raw material sourcing ensures a more predictable cost structure and mitigates the risk of supply disruptions caused by the limited availability of high-purity specialty gases.
- Cost Reduction in Manufacturing: The elimination of ultra-low temperature requirements significantly lowers energy consumption, as the process operates effectively at near-ambient or mildly elevated temperatures. By removing the need for expensive cryogenic cooling infrastructure and the associated maintenance, the overall capital expenditure for setting up production lines is substantially reduced. Additionally, the high atom economy and yield minimize raw material waste, directly translating to lower variable costs per kilogram of the final active pharmaceutical ingredient intermediate.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as haloacetals and magnesium are produced at a massive global scale for various chemical applications. This abundance ensures that procurement teams can secure long-term contracts with multiple suppliers, preventing bottlenecks that often occur with niche reagents. The robustness of the reaction conditions also means that production can be maintained consistently across different seasons and geographic locations without the sensitivity to ambient temperature fluctuations that plagues cryogenic processes.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional routes, particularly by avoiding the heavy metal catalysts or sulfur-containing byproducts found in alternative methods. The aqueous waste streams contain manageable salts that can be treated using standard effluent protocols, facilitating easier regulatory approval for plant expansions. This environmental friendliness aligns perfectly with modern green chemistry initiatives, allowing manufacturers to meet stringent sustainability goals while scaling up production volumes to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis pathway. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety profiles, and scalability potential. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary safety advantages of this new C11 aldehyde synthesis route?
A: Unlike traditional acetylene-based routes that require hazardous lithium amide and ultra-low temperatures, this Grignard-based method operates at mild temperatures (20-70°C) and avoids explosive gases, significantly enhancing operational safety.
Q: How does this method improve yield compared to the Darzens condensation route?
A: The patented Grignard route achieves yields up to 93.7% with high purity, whereas the older Darzens condensation method suffers from low atom economy, significant wastewater generation, and lower overall yields due to complex rearrangement steps.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes cheap, commercially available raw materials and classical reaction types that are easy to control, making it highly suitable for green industrial production and commercial scale-up without specialized cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2, 6-trimethyl-1-cyclohexenyl) acetaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of essential vitamins and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory success to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of C11 aldehyde meets the exacting standards required for downstream Vitamin A synthesis, providing our partners with absolute confidence in material consistency.
We invite you to collaborate with us to leverage this advanced Grignard-based technology for your production needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative process can optimize your supply chain and enhance your competitive position in the market.
