Revolutionizing Battery Safety: Scalable Synthesis of High-Purity Unsaturated Propyl Phosphates
Introduction to Next-Generation Electrolyte Additives
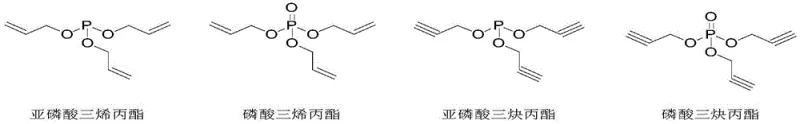
The rapid evolution of the electric vehicle (EV) sector has placed unprecedented demand on the safety and performance metrics of lithium-ion batteries. As thermal runaway incidents remain a critical concern, the industry is aggressively seeking advanced electrolyte additives that can form stable passivation films on electrode surfaces without compromising ionic conductivity. Patent CN110066292B introduces a transformative methodology for synthesizing unsaturated propyl phosphite and phosphate esters, specifically triallyl phosphite, triallyl phosphate, tripropargyl phosphite, and tripropargyl phosphate. These compounds are not merely chemical intermediates; they are functional materials capable of significantly enhancing the high-temperature storage and cycle life of power batteries. By addressing the longstanding synthesis challenges associated with these molecules, this technology offers a pathway to safer, more reliable energy storage solutions.
The strategic importance of these additives lies in their dual functionality: they act as flame retardants to mitigate fire hazards during charging and discharging, and they improve the overall electrochemical stability of the cell. However, the commercial viability of such high-performance materials has historically been hindered by complex synthesis routes that yield impure products. The disclosed invention overcomes these barriers by utilizing a direct reaction between phosphorus halides and specific unsaturated esters. This approach eliminates the need for auxiliary catalysts and leverages the volatility of byproducts to drive reaction completion, resulting in yields and purities that meet the rigorous standards of the electronic chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of triallyl phosphate has relied on the reaction of phosphorus oxychloride with allyl alcohol in the presence of acid binders like triethylamine. While conceptually straightforward, this legacy process suffers from severe inefficiencies that render it suboptimal for modern large-scale manufacturing. The primary drawback is the generation of substantial amounts of amine salt waste, which complicates the workup procedure and increases the environmental footprint of the operation. Furthermore, the reactivity of phosphorus oxychloride towards alcohols can be difficult to control, often leading to incomplete conversion and a complex mixture of partially substituted intermediates.
Another significant bottleneck in traditional synthesis is the difficulty in separating the final product from unreacted starting materials and side products. The boiling points of the intermediates and the target phosphate ester are often dangerously close, requiring high-efficiency fractional distillation columns and multiple purification passes. This not only escalates energy consumption but also leads to product degradation due to prolonged exposure to heat. Consequently, the final purity often falls short of the >99.5% threshold required for premium battery electrolyte applications, limiting the utility of the material in high-end electronics and forcing manufacturers to accept lower performance margins.
The Novel Approach
The methodology outlined in the patent data represents a paradigm shift by replacing the alcohol-amine system with an ester-exchange mechanism. By reacting phosphorus trichloride (PCl3) or phosphorus oxychloride (POCl3) directly with allyl or propargyl esters, the process bypasses the formation of non-volatile salt byproducts entirely. Instead, the reaction generates acyl chlorides, which are volatile compounds that can be continuously distilled off as they form. This continuous removal of the byproduct effectively shifts the chemical equilibrium towards the product side, ensuring near-quantitative conversion without the need for excessive reagent loading or harsh driving forces.
This catalyst-free protocol simplifies the reactor setup and drastically reduces the complexity of the downstream purification train. Since the byproduct is removed during the reaction phase, the crude reaction mixture is already significantly enriched in the target phosphate or phosphite ester. This allows for a single, efficient vacuum rectification step to achieve pharmaceutical-grade purity. The elimination of amine catalysts also removes a major source of potential metal ion contamination, which is critical for preventing micro-short circuits in battery cells. This streamlined approach not only improves the quality of the final additive but also aligns perfectly with green chemistry principles by minimizing waste generation.
Mechanistic Insights into Catalyst-Free Transesterification
The core of this innovation lies in the nucleophilic substitution mechanism at the phosphorus center. In the absence of a base catalyst, the oxygen atom of the allyl or propargyl ester acts as the nucleophile, attacking the electrophilic phosphorus atom in PCl3 or POCl3. This attack displaces a chloride ion, forming a transient intermediate that rapidly collapses to release an acyl chloride molecule. The key to the success of this reaction is the thermodynamic favorability provided by the formation of the strong P-O bond coupled with the entropic drive of releasing a volatile gas or low-boiling liquid (the acyl chloride). Unlike traditional alcoholysis where the leaving group is HCl (which requires a base to trap), here the leaving group is a neutral organic molecule that naturally exits the reaction phase.

From an impurity control perspective, this mechanism is exceptionally clean. Traditional methods often suffer from etherification side reactions where the alcohol reacts with itself or the solvent, creating hard-to-remove organic impurities. In the ester-based route, the specificity of the acyl group as a leaving group minimizes these competing pathways. Furthermore, because the reaction can be conducted in a variety of solvents—or even solvent-free depending on the viscosity requirements—the risk of solvent participation in side reactions is negligible. The ability to tune the reaction temperature between 70°C and 140°C allows operators to optimize the rate of acyl chloride evolution, ensuring that the reaction proceeds smoothly without thermal degradation of the sensitive unsaturated double or triple bonds in the allyl and propargyl groups.
How to Synthesize Triallyl Phosphate Efficiently
Implementing this synthesis route requires precise control over stoichiometry and temperature to maximize the efficiency of the byproduct removal. The process begins with the preparation of a homogeneous mixture of the phosphorus halide and a selected solvent, such as 1,4-dioxane or toluene, although the patent notes that solvent-free conditions are also viable. The allyl ester, such as diallyl carbonate or allyl acetate, is then added dropwise to the heated phosphorus mixture. Critical to the operation is the maintenance of a temperature window between 70°C and 140°C, which ensures that the acyl chloride byproduct boils off immediately upon formation, driving the reaction to completion without the need for post-reaction stripping.
- Mix phosphorus trichloride (PCl3) or phosphorus oxychloride (POCl3) with a suitable solvent in a reaction vessel.
- Heat the mixture to 70-140°C and dropwise add allyl or propargyl esters while continuously separating the low-boiling acyl chloride byproduct.
- Maintain temperature for 2-4 hours post-addition, then purify the final product via vacuum rectification at specific temperatures and pressures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this catalyst-free esterification process offers tangible economic and operational benefits that extend beyond simple yield improvements. The most immediate impact is the drastic reduction in raw material costs associated with acid binders and the subsequent waste disposal fees. By eliminating the need for triethylamine or pyridine, manufacturers remove a significant line item from the bill of materials. Additionally, the simplified purification process reduces the energy load on distillation units, allowing for faster batch cycles and higher throughput in existing production facilities without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The elimination of expensive amine catalysts and the reduction in solvent usage directly lower the variable cost per kilogram of the final product. Since the byproduct is a valuable acyl chloride that can potentially be recovered and sold or reused, the process moves closer to a zero-waste model. This inherent efficiency allows suppliers to offer more competitive pricing for high-purity battery additives, making advanced safety features more accessible to cell manufacturers who are under constant pressure to reduce the cost per kilowatt-hour.
- Enhanced Supply Chain Reliability: Traditional synthesis routes are prone to bottlenecks caused by the handling and disposal of large volumes of amine salts, which can delay batch release and shipment. The new method's continuous removal of volatile byproducts streamlines the production timeline, significantly reducing the lead time from raw material intake to finished goods. This agility is crucial for meeting the fluctuating demands of the EV market, where supply chains must be responsive to rapid scaling requirements. The robustness of the reaction conditions also ensures consistent batch-to-batch quality, reducing the risk of supply disruptions due to out-of-specification material.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies waste management challenges, but this technology inherently mitigates that risk. The absence of solid salt waste simplifies compliance with increasingly stringent environmental regulations regarding hazardous waste disposal. The process is highly scalable from pilot plants to multi-ton reactors because the heat management and byproduct removal mechanisms rely on fundamental physical properties (boiling points) rather than complex mixing dynamics. This makes it an ideal candidate for rapid capacity expansion to support the growing global demand for safe, high-performance lithium-ion batteries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these unsaturated phosphorus esters. The answers are derived directly from the mechanistic advantages and process parameters detailed in the underlying patent literature, providing a clear understanding of why this method represents a superior choice for industrial adoption.
Q: Why is the catalyst-free method superior for battery additive production?
A: Traditional methods often require amine catalysts like triethylamine, which generate salt waste and complicate purification. The novel catalyst-free approach eliminates these impurities, ensuring the ultra-high purity required for sensitive lithium-ion battery electrolytes.
Q: How does the new process handle byproduct separation?
A: The reaction generates acyl chloride compounds as byproducts, which have significantly lower boiling points than the target phosphate esters. This allows for continuous removal during the reaction, driving the equilibrium forward and simplifying downstream processing.
Q: What are the primary applications of triallyl phosphate esters?
A: These compounds serve as critical flame-retardant additives in lithium-ion battery electrolytes, enhancing thermal stability and preventing thermal runaway. They are also valuable in polymer synthesis for creating wear-resistant and transparent materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triallyl Phosphate Supplier
As the global demand for safer and more efficient energy storage solutions accelerates, the need for high-quality electrolyte additives has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the catalyst-free esterification process to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major battery manufacturers. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of triallyl phosphate or tripropargyl phosphite meets the exacting standards required for next-generation lithium-ion cells.
We invite R&D directors and procurement specialists to collaborate with us to optimize their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of these critical materials, ensuring your products remain competitive in the rapidly evolving landscape of electronic chemicals and energy storage.
