Advanced Cobalt-Catalyzed Synthesis of Vonoprazan Fumarate for Industrial Scale Production
Advanced Cobalt-Catalyzed Synthesis of Vonoprazan Fumarate for Industrial Scale Production
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for the production of high-value active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in patent CN110452222B, which discloses a novel preparation method for Vonoprazan Fumarate, a potent potassium-competitive acid blocker (P-CAB) used in the treatment of erosive esophagitis and Helicobacter pylori infections. This patent introduces a streamlined synthetic strategy that utilizes 5-(2-fluorophenyl)-1-(3-pyridylsulfonyl)-3-cyano-1H-pyrrole as a key starting material. Unlike legacy processes that suffer from excessive step counts and hazardous operating conditions, this innovation employs a highly efficient cobalt-catalyzed reductive amination. By directly converting the cyano group into the requisite methylaminomethyl fragment in a single operational step, the technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for gastrointestinal therapeutics. The method not only simplifies the process flow but also addresses critical quality attributes regarding impurity profiles and metal residues, making it an ideal candidate for large-scale commercial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the methylaminomethyl fragment in Vonoprazan has been plagued by inefficiencies that hinder cost-effective mass production. The first generation of synthetic routes, as illustrated in prior art, typically relies on a linear progression starting from ester precursors. This involves a multi-step sequence where an ester is reduced to an alcohol, subsequently oxidized to an aldehyde, and finally subjected to reductive amination. 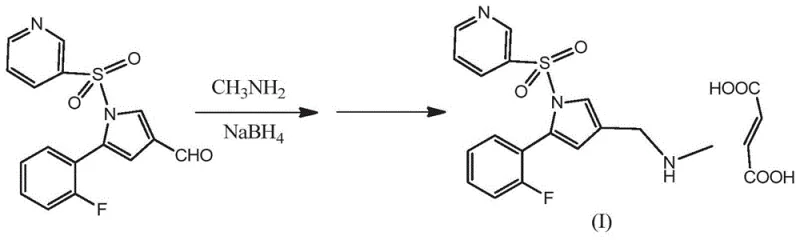 This elongated pathway inherently accumulates yield losses at every stage and generates substantial chemical waste. Furthermore, alternative strategies involving the bromination of methyl groups followed by N-alkylation present severe purification challenges. These alkylation reactions are notoriously prone to generating tertiary amine by-products due to the difficulty in controlling mono-alkylation selectivity. The presence of these structurally similar impurities necessitates complex and expensive chromatographic separations or repeated recrystallizations, drastically impacting the overall throughput and economic feasibility of the manufacturing process. Additionally, routes relying on selective hydrogenation of nitriles to aldehydes often suffer from instability, where the intermediate aldehyde is easily over-oxidized to carboxylic acids, further complicating the reaction control and lowering the final yield.
This elongated pathway inherently accumulates yield losses at every stage and generates substantial chemical waste. Furthermore, alternative strategies involving the bromination of methyl groups followed by N-alkylation present severe purification challenges. These alkylation reactions are notoriously prone to generating tertiary amine by-products due to the difficulty in controlling mono-alkylation selectivity. The presence of these structurally similar impurities necessitates complex and expensive chromatographic separations or repeated recrystallizations, drastically impacting the overall throughput and economic feasibility of the manufacturing process. Additionally, routes relying on selective hydrogenation of nitriles to aldehydes often suffer from instability, where the intermediate aldehyde is easily over-oxidized to carboxylic acids, further complicating the reaction control and lowering the final yield.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology disclosed in CN110452222B revolutionizes the synthesis by enabling a direct, one-step transformation. The core innovation lies in the direct reductive amination of the stable cyano precursor. By utilizing a specialized cobalt catalyst system in conjunction with a reducing agent and methylamine source, the process bypasses the need for unstable aldehyde intermediates or hazardous alkyl halides. 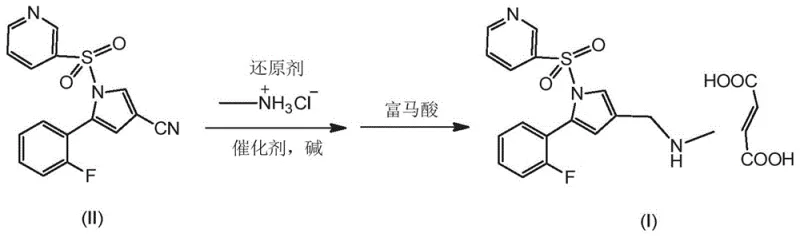 This approach effectively collapses multiple unit operations into a single reactor charge, significantly reducing the solvent footprint and energy consumption associated with isolation and purification of intermediates. The reaction conditions are remarkably mild, typically proceeding at temperatures around 70°C under atmospheric pressure, which eliminates the need for expensive high-pressure hydrogenation equipment. This simplification not only accelerates the production cycle time but also enhances the safety profile of the plant by removing high-risk operations. The result is a robust, high-yielding process that delivers the Vonoprazan base with superior purity, ready for immediate salt formation with fumaric acid to produce the final API.
This approach effectively collapses multiple unit operations into a single reactor charge, significantly reducing the solvent footprint and energy consumption associated with isolation and purification of intermediates. The reaction conditions are remarkably mild, typically proceeding at temperatures around 70°C under atmospheric pressure, which eliminates the need for expensive high-pressure hydrogenation equipment. This simplification not only accelerates the production cycle time but also enhances the safety profile of the plant by removing high-risk operations. The result is a robust, high-yielding process that delivers the Vonoprazan base with superior purity, ready for immediate salt formation with fumaric acid to produce the final API.
Mechanistic Insights into Cobalt-Catalyzed Reductive Amination
The success of this novel pathway hinges on the sophisticated design of the catalytic system, which utilizes earth-abundant cobalt rather than precious metals like palladium or platinum. The catalyst is formed in situ by combining a cobalt salt, such as cobalt chloride (CoCl2), with a specifically tailored phosphine-amine ligand. 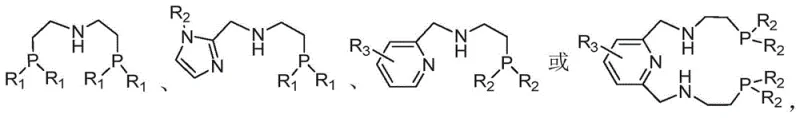 These ligands, characterized by their bis-phosphino amine architecture, play a critical role in stabilizing the cobalt center and modulating its electronic properties to facilitate the selective reduction of the nitrile group. The mechanism likely involves the coordination of the nitrile nitrogen to the cobalt center, followed by hydride transfer from the reducing agent (such as sodium borohydride or ammonia borane) to form an imine intermediate. This imine is subsequently reduced to the secondary amine in the same pot. The choice of ligand is paramount; substituents on the phosphorus atoms, such as tert-butyl or cyclohexyl groups, provide the necessary steric bulk to prevent catalyst deactivation while maintaining high turnover frequencies. This homogeneous catalytic system ensures that the reaction proceeds efficiently in organic solvents like tetrahydrofuran or alcohols, allowing for excellent mass transfer and reaction kinetics.
These ligands, characterized by their bis-phosphino amine architecture, play a critical role in stabilizing the cobalt center and modulating its electronic properties to facilitate the selective reduction of the nitrile group. The mechanism likely involves the coordination of the nitrile nitrogen to the cobalt center, followed by hydride transfer from the reducing agent (such as sodium borohydride or ammonia borane) to form an imine intermediate. This imine is subsequently reduced to the secondary amine in the same pot. The choice of ligand is paramount; substituents on the phosphorus atoms, such as tert-butyl or cyclohexyl groups, provide the necessary steric bulk to prevent catalyst deactivation while maintaining high turnover frequencies. This homogeneous catalytic system ensures that the reaction proceeds efficiently in organic solvents like tetrahydrofuran or alcohols, allowing for excellent mass transfer and reaction kinetics.
From a quality control perspective, the mechanistic pathway offers distinct advantages in impurity management. Because the reaction proceeds through a controlled catalytic cycle rather than a stochastic alkylation event, the formation of over-alkylated tertiary amines is virtually eliminated. This inherent selectivity is a major benefit for R&D teams focused on regulatory compliance, as it simplifies the impurity profile of the drug substance. Furthermore, the solubility characteristics of the cobalt-ligand complex are engineered to facilitate removal. Post-reaction, the addition of water quenches the reducing agent and alters the solubility of the metal species, allowing them to be separated from the organic phase containing the product. Subsequent recrystallization steps, particularly during the salt formation with fumaric acid, act as a final polishing operation to ensure that residual metal levels are well below the strict thresholds mandated by ICH Q3D guidelines for elemental impurities in pharmaceuticals.
How to Synthesize Vonoprazan Fumarate Efficiently
The implementation of this synthesis route is straightforward and amenable to standard chemical processing equipment found in most multipurpose API manufacturing facilities. The process begins with the preparation of the catalytic solution under an inert nitrogen atmosphere to prevent oxidation of the sensitive cobalt species. Following the activation of the catalyst, the cyano-pyrrole substrate is introduced along with the amine source and base. The reaction is then driven to completion by heating, after which a simple aqueous workup isolates the crude base. The final step involves the reaction of the isolated base with fumaric acid in a suitable solvent to precipitate the high-purity fumarate salt. This operational simplicity reduces the training burden on plant personnel and minimizes the risk of operator error during scale-up.
- Prepare the catalytic system by mixing a cobalt salt (e.g., CoCl2) with a specialized phosphine-amine ligand in a solvent like THF under inert atmosphere.
- Add the cyano-pyrrole substrate, methylamine hydrochloride, a base (e.g., NaOH), and a reducing agent (e.g., NaBH4) to the catalyst mixture.
- Heat the reaction mixture to reflux (approx. 70°C) for 10 hours, then quench with water, extract the product, and react with fumaric acid to crystallize the final salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed methodology translates into tangible strategic benefits that extend beyond mere technical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized intermediates like unstable aldehydes or reactive alkyl halides, manufacturers can source more stable and commoditized starting materials, thereby reducing exposure to price volatility and supply disruptions. The reduction in process steps also means a significant decrease in the consumption of solvents and reagents, which directly correlates to lower variable costs per kilogram of produced API. Moreover, the avoidance of high-pressure hydrogenation removes a major bottleneck in many contract manufacturing organizations (CMOs), as it frees up specialized equipment and reduces the safety audit burden associated with handling hydrogen gas. This flexibility allows for faster technology transfer and quicker time-to-market for generic or new formulations of Vonoprazan.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of multiple isolation and purification stages. In traditional multi-step syntheses, each intermediate isolation incurs costs related to filtration, drying, and quality control testing, alongside yield losses. By consolidating the synthesis into a fewer number of steps, the overall material throughput is maximized. Additionally, the use of a cobalt-based catalyst represents a substantial cost saving compared to noble metal catalysts, which are subject to fluctuating market prices and require rigorous recovery protocols. The ability to operate at atmospheric pressure further reduces capital depreciation costs associated with high-pressure reactors, leading to a lower fixed cost allocation for the production line.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The process tolerates a wider range of operating parameters without compromising product quality, making it less susceptible to batch failures caused by minor deviations in temperature or pressure. The starting material, the cyano-pyrrole derivative, is chemically stable and can be stored for extended periods without degradation, allowing manufacturers to maintain strategic stockpiles without the risk of spoilage. This stability ensures a continuous flow of production even in the face of logistical delays, providing a reliable buffer against market fluctuations and ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is significantly greener. The reduction in solvent usage and the elimination of hazardous reagents like brominating agents lower the facility's environmental footprint and waste disposal costs. The process generates less hazardous waste, simplifying compliance with increasingly stringent environmental regulations. Scalability is inherently supported by the homogeneous nature of the reaction, which ensures uniform heat and mass transfer even in large-scale reactors. This predictability allows for seamless scale-up from pilot plant to commercial tonnage without the need for extensive re-optimization, ensuring that the quality established during development is maintained throughout the product lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for potential licensing or manufacturing partnerships. Understanding these nuances is critical for making informed decisions about process adoption and integration into existing manufacturing portfolios.
Q: Why is the cobalt-catalyzed route superior to traditional hydrogenation methods?
A: Traditional methods often require high-pressure hydrogenation equipment and precise control to avoid over-reduction. The cobalt-catalyzed chemical reduction operates at atmospheric pressure with milder conditions, significantly reducing capital expenditure on specialized high-pressure reactors and enhancing operational safety.
Q: How does this method control impurities compared to alkylation routes?
A: Conventional N-alkylation routes using alkyl halides frequently generate difficult-to-remove tertiary amine by-products due to over-alkylation. This reductive amination pathway selectively targets the nitrile group, inherently minimizing the formation of higher-order amine impurities and simplifying downstream purification.
Q: Is the cobalt catalyst easily removed from the final API?
A: Yes, the catalyst system is designed to be homogeneous yet removable. The specific ligand architecture allows the metal complex to remain soluble during reaction but facilitates removal during the aqueous workup and recrystallization steps, ensuring the final product meets stringent heavy metal residue specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vonoprazan Fumarate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110452222B for the global pharmaceutical market. As a leading CDMO and supplier, we possess the technical expertise and infrastructure to leverage such innovative routes for the commercial production of high-purity Vonoprazan Fumarate. Our facilities are equipped to handle complex homogeneous catalytic reactions with precision, ensuring that every batch meets the rigorous quality standards required by international regulatory bodies. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, demonstrating our capability to support both clinical trial material needs and full-scale commercial launch volumes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles.
We invite pharmaceutical companies and procurement leaders to collaborate with us to optimize their supply chains for gastrointestinal medications. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific cobalt-catalyzed route can reduce your total cost of ownership. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. Together, we can accelerate the delivery of affordable and high-quality medicines to patients worldwide, leveraging cutting-edge chemistry to drive efficiency and reliability in the pharmaceutical supply chain.
