Advanced Ruthenium-Catalyzed Synthesis of Bioactive Triazine Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for constructing complex heterocyclic scaffolds, particularly those serving as core structures for antiviral and antitumor agents. Patent CN112159360B, published in early 2022, introduces a groundbreaking preparation method for 2-dimethylamino-6-benzylamino substituted triazine compounds, which are pivotal intermediates in the synthesis of bioactive molecules targeting HIV and leukemia. This technology represents a paradigm shift from classical nucleophilic substitution strategies to a sophisticated ruthenium-catalyzed cascade reaction. By leveraging a borrowing hydrogen methodology, the process simultaneously achieves the reduction of a carbon-carbon double bond and the N-alkylation of an amine functionality in a single operational step. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: it eliminates the generation of hazardous hydrogen chloride gas, drastically reduces waste streams, and utilizes inexpensive, stable raw materials like benzyl alcohol instead of reactive halides. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented technologies is crucial for securing a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino-substituted triazine derivatives has relied heavily on the stepwise substitution of cyanuric chloride (2,4,6-trichloro-1,3,5-triazine) with various amines. While chemically straightforward, this traditional approach suffers from severe drawbacks that hinder its viability for modern, large-scale green manufacturing. The primary issue is the stoichiometric generation of hydrogen chloride (HCl) gas as a byproduct during each substitution step, which necessitates the use of excess base scavengers and creates significant corrosion risks for reactor equipment. Furthermore, controlling the regioselectivity of substitution on the triazine ring often requires cryogenic conditions and precise temperature management to prevent the formation of difficult-to-remove polysubstituted impurities. The purification of the final product often involves extensive chromatography or recrystallization to remove inorganic salts and unreacted starting materials, leading to poor overall mass balance and increased environmental burden. Additionally, introducing the benzylamino moiety typically requires benzyl halides, which are lachrymators and pose safety hazards, further complicating the regulatory compliance and safety profile of the manufacturing process.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a transition metal-catalyzed transfer hydrogenation strategy that elegantly bypasses these limitations. The core transformation involves reacting a styryl-substituted triazine precursor (Formula I) directly with benzyl alcohol in the presence of a ruthenium catalyst and a base. This single-pot operation achieves two distinct chemical transformations: the hydrogenation of the exocyclic vinyl group to an ethyl linkage and the N-alkylation of the triazine amine to form the benzylamino derivative (Formula II). 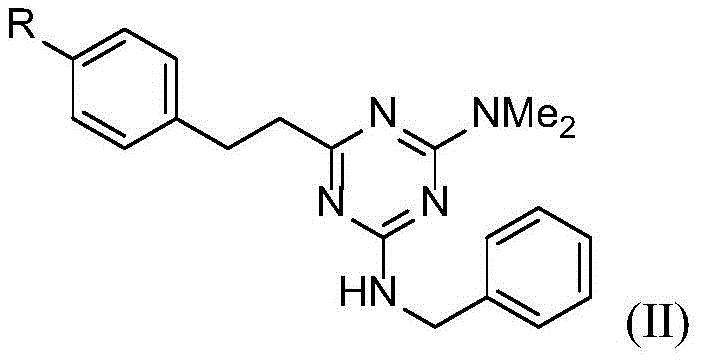 This methodology is inherently greener, as the only byproduct of the N-alkylation step is water, and the hydrogen required for the alkene reduction is borrowed from the alcohol reactant itself. The process operates under relatively mild thermal conditions (120-150°C) in a sealed system, ensuring high atom economy and minimizing volatile organic compound (VOC) emissions. For a reliable pharmaceutical intermediate supplier, adopting such a route translates directly to cost reduction in API manufacturing by simplifying the workflow and reducing the need for hazardous reagent handling.
This methodology is inherently greener, as the only byproduct of the N-alkylation step is water, and the hydrogen required for the alkene reduction is borrowed from the alcohol reactant itself. The process operates under relatively mild thermal conditions (120-150°C) in a sealed system, ensuring high atom economy and minimizing volatile organic compound (VOC) emissions. For a reliable pharmaceutical intermediate supplier, adopting such a route translates directly to cost reduction in API manufacturing by simplifying the workflow and reducing the need for hazardous reagent handling.
Mechanistic Insights into Ruthenium-Catalyzed Cascade Transformation
The success of this synthesis hinges on the unique ability of the ruthenium catalyst, specifically complexes like RuCl2(PPh3)3, to mediate a "borrowing hydrogen" or "hydrogen autotransfer" cycle. Mechanistically, the ruthenium center first dehydrogenates the benzyl alcohol to generate benzaldehyde and a ruthenium-hydride species in situ. The generated benzaldehyde then undergoes a condensation reaction with the primary amine group on the triazine ring to form an imine intermediate. Subsequently, the ruthenium-hydride species reduces this imine back to the secondary amine (the benzylamino group), regenerating the active catalyst. Concurrently, the same catalytic system facilitates the reduction of the styryl double bond present on the triazine scaffold. This dual functionality is critical; without the simultaneous reduction, the resulting styryl-benzylamino product might exhibit different biological activity or stability profiles compared to the saturated phenethyl analog. The choice of base, preferably potassium tert-butoxide (t-BuOK), is essential to drive the deprotonation steps and maintain the catalytic cycle efficiency. 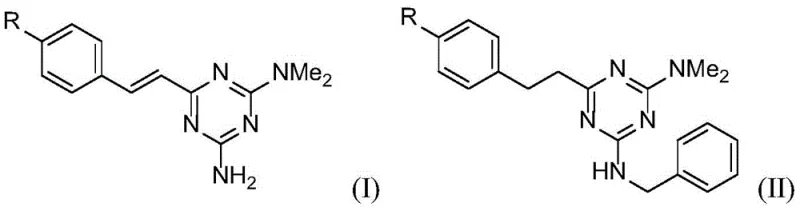 The versatility of this mechanism is demonstrated by its tolerance to various electronic environments on the aromatic ring, as evidenced by the successful synthesis of derivatives where R is hydrogen, chlorine, or methoxy. This robustness ensures that the process can be adapted for a wide library of analogs without requiring extensive re-optimization of reaction parameters.
The versatility of this mechanism is demonstrated by its tolerance to various electronic environments on the aromatic ring, as evidenced by the successful synthesis of derivatives where R is hydrogen, chlorine, or methoxy. This robustness ensures that the process can be adapted for a wide library of analogs without requiring extensive re-optimization of reaction parameters.
From an impurity control perspective, this mechanism offers superior selectivity compared to direct alkylation with halides. Traditional alkylation often leads to over-alkylation (forming tertiary amines) or N-alkylation at unintended sites. However, the imine intermediate formed in this borrowing hydrogen pathway is sterically and electronically constrained, favoring the formation of the desired secondary amine. Furthermore, the reduction of the alkene is highly chemoselective, leaving the triazine ring intact while saturating the side chain. This high level of control minimizes the formation of structural isomers and homologs, resulting in a crude reaction mixture that is significantly cleaner than those obtained from conventional routes. For quality control teams, this means simpler analytical profiles and higher confidence in batch-to-batch consistency, which is a prerequisite for supplying high-purity triazine derivatives to regulated markets.
How to Synthesize 2-Dimethylamino-6-Benzylamino Substituted Triazine Efficiently
Implementing this synthesis requires careful attention to the stoichiometry and reaction environment to maximize yield and minimize catalyst loading. The patent outlines a robust protocol where the molar ratio of the triazine precursor to benzyl alcohol is maintained between 1:3.1 and 1:4.2, ensuring a slight excess of the alcohol to drive the equilibrium forward. The ruthenium catalyst loading is remarkably low, ranging from 0.032 to 0.055 equivalents, which is economically advantageous for large-scale operations. The reaction is conducted in 1,4-dioxane, a solvent that provides excellent solubility for both the polar triazine species and the organic alcohol while withstanding the elevated temperatures required.
- Combine the styryl-substituted triazine precursor (Formula I) with benzyl alcohol and a ruthenium catalyst such as RuCl2(PPh3)3 in a sealed reactor.
- Add an alkaline substance like potassium tert-butoxide (t-BuOK) and a solvent such as 1,4-dioxane to the mixture.
- Heat the reaction mixture to 120-150°C for 5-15 hours to facilitate simultaneous alkene reduction and N-alkylation, followed by standard aqueous workup and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond simple chemistry. The shift from hazardous benzyl halides to commodity-grade benzyl alcohol represents a significant optimization in raw material sourcing. Benzyl alcohol is widely available, stable, and non-regulated, unlike many alkyl halides which face increasing scrutiny due to their toxicity and environmental persistence. This change simplifies the logistics of raw material storage and handling, reducing the need for specialized containment systems and lowering insurance costs associated with hazardous chemical inventory. Moreover, the elimination of HCl gas generation removes the requirement for expensive scrubbing systems and corrosion-resistant reactors (such as glass-lined steel), allowing the process to be run in standard stainless steel equipment with appropriate linings. This flexibility enhances the agility of the supply chain, enabling faster turnaround times and the ability to utilize a broader range of manufacturing partners.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven primarily by the simplification of the downstream processing. Since the reaction generates water as the only byproduct of the alkylation step and avoids inorganic salt formation, the workup procedure is drastically simplified. There is no need for extensive aqueous washing to remove salts or neutralization steps to quench acid, which reduces water consumption and wastewater treatment costs. Additionally, the high yields reported (up to 89% in optimized examples) mean that less raw material is wasted per kilogram of product produced. The low catalyst loading further contributes to cost efficiency, as precious metal recovery or disposal costs are minimized. These factors combine to deliver substantial cost savings in the overall cost of goods sold (COGS), making the final intermediate more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-sensitive reagents. Benzyl alcohol and the triazine precursors are stable solids or liquids that do not require cold chain logistics or inert atmosphere storage, unlike some sensitive organometallic reagents used in alternative cross-coupling approaches. The reaction conditions (120-150°C) are easily achievable in standard industrial reactors, reducing the risk of batch failures due to equipment limitations. This reliability ensures consistent delivery schedules, which is critical for downstream API manufacturers who operate on tight timelines. By reducing the complexity of the synthesis, the risk of supply disruption due to regulatory changes on specific reagents is also mitigated, providing a more secure long-term supply source for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is straightforward due to the homogeneous nature of the catalytic system and the absence of exothermic hazards associated with acid-base neutralizations. The process aligns perfectly with modern green chemistry principles, significantly reducing the E-factor (mass of waste per mass of product). For companies aiming to meet stringent ESG (Environmental, Social, and Governance) goals, this route offers a clear advantage. The reduction in solvent usage (due to higher concentrations being feasible) and the elimination of halogenated waste streams simplify environmental permitting and compliance reporting. This makes the commercial scale-up of complex heterocycles not just technically feasible, but also environmentally responsible, appealing to eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their existing development pipelines.
Q: What are the primary advantages of this ruthenium-catalyzed method over traditional trichlorotriazine substitution?
A: Unlike traditional methods that generate corrosive HCl gas and multiple byproducts, this patented process utilizes benign benzyl alcohol and achieves high atom economy through a cascade reduction-alkylation sequence, significantly simplifying purification and waste treatment.
Q: Can this synthesis be scaled for industrial production of antiviral intermediates?
A: Yes, the process uses robust conditions (120-150°C) and commercially available catalysts (RuCl2(PPh3)3) with high yields (up to 89%), making it highly suitable for the commercial scale-up of complex heterocycles required in pharmaceutical manufacturing.
Q: What represents the critical quality attributes for the final triazine product?
A: The key quality attributes include the complete reduction of the styryl double bond to a phenethyl group and the selective mono-benzylation of the amine, ensuring high purity specifications essential for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Dimethylamino-6-Benzylamino Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the potential of the ruthenium-catalyzed route described in CN112159360B and is fully prepared to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including high-temperature pressure reactions and rigorous QC labs capable of verifying stringent purity specifications for every batch. We understand that in the pharmaceutical sector, quality is non-negotiable, and our commitment to excellence ensures that every gram of material we supply meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of specific analogs or bulk supply of the standard intermediates, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are eager to provide you with specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your supply chain and reduce your overall development costs.
