Advanced Synthesis of 2-(Trifluoromethyl)benzaldehyde for Industrial Pharmaceutical Applications
Advanced Synthesis of 2-(Trifluoromethyl)benzaldehyde for Industrial Pharmaceutical Applications
The global demand for fluorinated aromatic compounds has surged in recent years, driven by their critical role in the development of next-generation pharmaceuticals and agrochemicals. Among these valuable building blocks, 2-(trifluoromethyl)benzaldehyde stands out as a pivotal intermediate for synthesizing complex bioactive molecules. As detailed in Chinese Patent CN102516047B, a groundbreaking preparation method has been developed that addresses the longstanding inefficiencies of traditional synthesis routes. This innovative technology leverages a sophisticated two-stage process involving selective fluorination followed by a unique fatty acid-catalyzed hydrolysis. By shifting away from harsh mineral acids and stoichiometric organometallic reagents, this methodology offers a pathway to high-purity products with significantly reduced environmental impact. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a reliable 2-(trifluoromethyl)benzaldehyde supplier capable of meeting stringent quality standards while optimizing manufacturing costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-(trifluoromethyl)benzaldehyde has been plagued by severe operational and economic drawbacks that hinder efficient cost reduction in pharmaceutical intermediates manufacturing. One prominent legacy method, disclosed in US2180772, relies on the hydrolysis of o-trifluoromethyl benzal fluoride using 95% concentrated sulfuric acid at elevated temperatures. While this route can achieve yields around 95%, it necessitates a massive excess of sulfuric acid—approximately three times the weight of the substrate—resulting in the generation of substantial quantities of hazardous spent acid waste. Furthermore, the evolution of corrosive hydrogen fluoride gas during the reaction poses significant safety risks and requires specialized, expensive corrosion-resistant equipment. Alternative pathways, such as the Grignard reaction described in US4564619, introduce different challenges, including the high cost of magnesium chips and the extreme sensitivity of Grignard reagents to moisture, which complicates process control and increases the risk of batch failures in a commercial setting.
The Novel Approach
In stark contrast to these archaic techniques, the novel approach outlined in the patent introduces a remarkably cleaner and more controllable hydrolysis strategy. This method utilizes a buffer system composed of C1-C4 straight-chain saturated monobasic fatty acids, such as acetic acid, and their corresponding alkali metal salts, like sodium acetate. This specific chemical environment facilitates the hydrolysis of o-trifluoromethyl benzal chloride (Formula II) under moderate pressure and temperature conditions, effectively bypassing the need for aggressive mineral acids. The reaction proceeds smoothly at temperatures between 150°C and 190°C and pressures ranging from 0.3MPa to 0.78MPa. By employing this organic acid salt system, the process not only mitigates equipment corrosion but also simplifies the downstream purification steps, as the reaction mixture can be easily separated and distilled. This represents a paradigm shift towards greener chemistry, offering a robust solution for the commercial scale-up of complex fluorinated intermediates without the burden of excessive waste treatment.
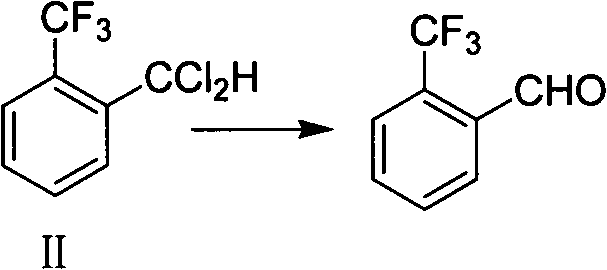
Mechanistic Insights into Selective Fluorination and Acid-Catalyzed Hydrolysis
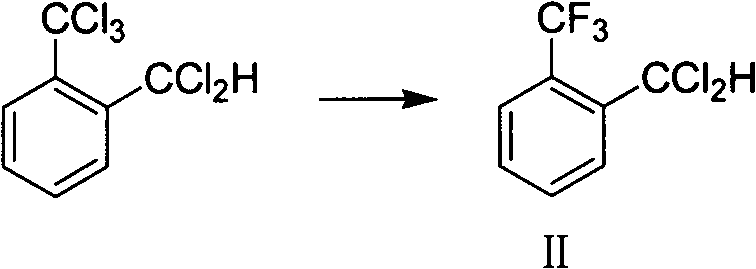
The success of this synthesis hinges on the precise execution of the precursor synthesis via selective fluorination, a critical step that determines the overall yield and purity profile of the final aldehyde. The process begins with the conversion of o-trichloromethyl benzal chloride into the key intermediate, o-trifluoromethyl benzal chloride (Formula II), using hydrogen fluoride in the presence of an antimony halide catalyst. Antimony trifluoride is particularly effective, operating at a catalytic loading of merely 0.05% to 5% by weight. The reaction is conducted at temperatures between 60°C and 110°C, where the antimony catalyst facilitates the exchange of chlorine atoms for fluorine atoms with high selectivity. This step is crucial because incomplete fluorination leads to chlorinated impurities that are difficult to separate later, while over-reaction can degrade the aromatic ring. The use of a 2-to-6-fold molar excess of hydrogen fluoride ensures complete conversion, driving the equilibrium towards the desired trifluoromethyl species while maintaining a manageable reaction exotherm.
Following the formation of the intermediate, the hydrolysis mechanism relies on the nucleophilic attack of water on the dichloromethyl group, activated by the acidic environment provided by the fatty acid. The presence of the alkali metal salt, such as sodium acetate, acts as a buffer to maintain a stable pH, preventing the rapid decomposition of the aldehyde product which can occur in strongly acidic or basic media. Impurity control is rigorously managed through gas chromatography monitoring, with the reaction endpoint defined by the residual content of the starting material dropping below 0.5%. This level of analytical control ensures that the subsequent vacuum distillation yields a product with a GC area purity exceeding 98%. The phase-transfer catalyst, often tetrabutylammonium bromide, further enhances the reaction kinetics by improving the miscibility of the organic and aqueous phases, thereby reducing reaction times from potentially days to just 3 to 4 hours. This mechanistic elegance translates directly into a more predictable and reproducible manufacturing process.
How to Synthesize 2-(Trifluoromethyl)benzaldehyde Efficiently
To implement this high-efficiency synthesis route, manufacturers must adhere to a strict protocol that balances reaction kinetics with safety parameters. The process is divided into two distinct operational phases: the initial fluorination to generate the intermediate, followed by the hydrolytic cleavage to form the aldehyde. Each stage requires specific attention to temperature ramping, pressure containment, and quenching procedures to maximize yield and minimize byproduct formation. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate these results in a pilot or production plant. Detailed standardized synthesis steps are provided in the guide below.
- Perform selective fluorination of o-trichloromethyl benzal chloride with hydrogen fluoride using antimony halide catalysts at 60°C-110°C.
- Isolate the intermediate o-trifluoromethyl benzal chloride (Formula II) via vacuum distillation after neutralizing residual acid.
- Hydrolyze the intermediate using a C1-C4 fatty acid and alkali metal salt system at 150°C-190°C under pressure to yield the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers compelling strategic benefits that extend far beyond simple yield improvements. The elimination of stoichiometric Grignard reagents and concentrated sulfuric acid fundamentally alters the cost structure of production, removing the need for expensive raw materials and the costly disposal of hazardous acidic waste streams. This shift results in substantial cost savings in raw material procurement and waste management, allowing for more competitive pricing in the global market. Furthermore, the reliance on commodity chemicals like acetic acid and sodium hydroxide ensures a stable supply chain, reducing the risk of production stoppages due to the scarcity of specialized reagents. The robustness of the reaction conditions also means that the process can be scaled from laboratory benchtop to multi-ton production with minimal re-engineering, ensuring consistent supply continuity for long-term contracts.
- Cost Reduction in Manufacturing: The economic viability of this process is largely driven by the replacement of high-cost, high-hazard reagents with inexpensive, bulk-available organic acids and salts. By avoiding the use of magnesium metal and anhydrous ether required for Grignard reactions, the process eliminates significant safety infrastructure costs and raw material expenses. Additionally, the mild hydrolysis conditions reduce energy consumption compared to high-temperature pyrolysis methods, while the simplified workup procedure lowers labor and solvent usage costs. These cumulative efficiencies lead to a drastically simplified cost model that enhances profit margins without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available feedstocks that are not subject to the same geopolitical or logistical constraints as specialty organometallics. The intermediate, o-trifluoromethyl benzal chloride, can be produced in large batches and stored, decoupling the fluorination step from the hydrolysis step and providing flexibility in production scheduling. This modularity allows manufacturers to respond quickly to fluctuations in demand, ensuring that lead times for high-purity pharmaceutical intermediates remain short and predictable even during periods of market volatility.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior to legacy methods. The absence of large volumes of spent sulfuric acid simplifies wastewater treatment and reduces the facility's environmental footprint. The closed-system nature of the autoclave reactions minimizes operator exposure to toxic vapors, aligning with modern occupational safety standards. These factors make the process highly scalable, as regulatory hurdles for expansion are lower, facilitating the rapid deployment of additional production capacity to meet growing global demand for fluorinated building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific supply chain needs. Please refer to the specific technical sections above for deeper mechanistic explanations.
Q: What are the primary advantages of this hydrolysis method over traditional sulfuric acid routes?
A: Unlike traditional methods requiring 95% sulfuric acid which generate massive amounts of spent acid and corrosive HF gas, this novel process utilizes a fatty acid and alkali metal salt buffer system. This significantly reduces equipment corrosion, minimizes hazardous waste disposal costs, and simplifies the post-reaction workup procedure.
Q: How is the purity of the intermediate controlled during the fluorination step?
A: The process employs gas chromatography (GC) monitoring to ensure the content of the starting material, o-trichloromethyl benzal chloride, remains below 0.5% before proceeding. The use of specific antimony halide catalysts, particularly antimony trifluoride, ensures high selectivity, preventing over-fluorination or side reactions that could compromise the final aldehyde purity.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the reaction conditions are highly amenable to scale-up. The hydrolysis step operates at moderate pressures (0.3MPa-0.78MPa) and temperatures (150°C-190°C) achievable in standard industrial autoclaves. Furthermore, the raw materials, such as acetic acid and sodium acetate, are commodity chemicals, ensuring supply chain stability for metric ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(Trifluoromethyl)benzaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We understand the critical nature of stringent purity specifications in the life sciences sector and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Whether you require custom synthesis or established catalog products, our infrastructure is designed to support your most demanding projects with reliability and precision.
We invite you to collaborate with us to explore how this advanced hydrolysis technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us be your partner in delivering high-quality fluorinated intermediates that drive innovation in your drug discovery and development pipelines.
