Advanced Catalytic Hydrogenation Route for Commercial Scale Production of L-Octahydroindoline-2-Carboxylic Acid
Advanced Catalytic Hydrogenation Route for Commercial Scale Production of L-Octahydroindoline-2-Carboxylic Acid
The pharmaceutical industry constantly seeks more efficient pathways for complex chiral intermediates, and patent CN101823993A presents a groundbreaking methodology for the synthesis of L-octahydroindoline-2-carboxylic acid. This specific compound serves as a critical building block for various bioactive molecules, necessitating a production route that balances high stereochemical purity with economic viability. The disclosed technology departs from traditional, waste-intensive methods by leveraging a series of catalytic hydrogenation steps that transform simple, commercially available raw materials like ortho-nitrotoluene and diethyl oxalate into the highly saturated target structure. By integrating condensation, selective reduction, and chiral resolution with racemization recycling, this process offers a compelling solution for manufacturers aiming to optimize their supply chains for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole derivatives and their saturated counterparts has often relied on the Fischer indole synthesis, a method that, while chemically robust, presents significant environmental and operational challenges for modern green chemistry standards. The traditional Fischer route typically involves the thermal rearrangement of phenylhydrazones under strong acidic catalysis, a process that invariably generates substantial quantities of acidic wastewater containing ammonia and organic byproducts. Furthermore, the raw materials required for phenylhydrazone formation, such as phenylhydrazine and specific ketones or aldehydes, can be costly and pose handling hazards due to their toxicity. The need for extensive downstream purification to remove these acidic residues and colored impurities often results in lower overall yields and increased production costs, making it less attractive for large-scale commercial manufacturing where margin compression is a constant pressure.
The Novel Approach
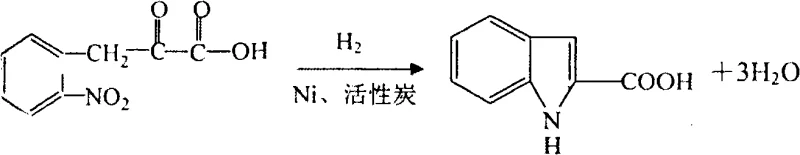
In stark contrast, the methodology outlined in patent CN101823993A introduces a streamlined, hydrogenation-centric strategy that fundamentally alters the economic and environmental profile of the synthesis. Instead of relying on harsh acid-catalyzed rearrangements, this novel approach utilizes a condensation reaction between diethyl oxalate and ortho-nitrotoluene to form an intermediate nitro-keto acid, which is then subjected to catalytic reduction. This shift allows for the construction of the indole core under much milder and more controllable conditions, significantly reducing the generation of hazardous waste streams. The subsequent steps involve precise acylation and further hydrogenation to saturate the pyrrole ring, followed by a sophisticated resolution and racemization cycle that ensures maximum material efficiency. This comprehensive route not only simplifies the purification workflow but also aligns perfectly with the increasing regulatory demands for cleaner manufacturing processes in the fine chemical sector.
Mechanistic Insights into Catalytic Hydrogenation and Chiral Resolution
The core of this synthetic innovation lies in its masterful application of heterogeneous catalysis to achieve selective bond reductions without compromising sensitive functional groups. The initial transformation of the nitro group and the concurrent cyclization to form the indole-2-carboxylic acid structure is achieved using a Nickel-based catalyst, likely Raney Nickel, under elevated hydrogen pressure. This step is critical as it must reduce the nitro functionality to an amine while simultaneously facilitating the intramolecular condensation with the adjacent keto-ester moiety. Following this, the process employs a second hydrogenation step, potentially using Palladium or Platinum on carbon, to selectively reduce the C2-C3 double bond of the indole ring to form the indoline scaffold. The precision required here is paramount to prevent over-reduction of the aromatic benzene ring at this stage, ensuring the intermediate retains the necessary structural integrity for subsequent chiral separation.
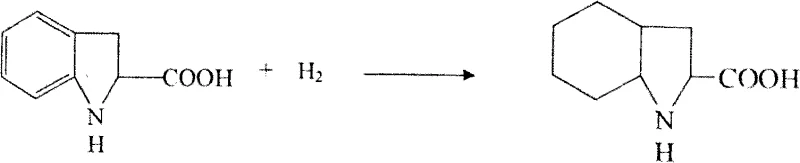
Perhaps the most economically significant aspect of the mechanism is the handling of chirality, which is managed through a dynamic kinetic resolution strategy involving resolution and racemization. The racemic indoline-2-carboxylic acid mixture is treated with a chiral resolving agent, such as methylbenzylamine, to precipitate the desired S-enantiomer salt. However, rather than discarding the mother liquor containing the unwanted R-enantiomer, the process subjects it to a high-temperature racemization treatment in the presence of a base like sodium carbonate. This converts the R-isomer back into a racemic mixture, which can be fed back into the resolution loop. This recycling mechanism theoretically allows for a yield approaching 100% regarding the chiral center utilization, a massive advantage over static resolution methods that cap yields at 50%. Finally, the S-indoline intermediate undergoes a rigorous hydrogenation using a Rhodium catalyst to fully saturate the benzene ring, yielding the target L-octahydroindoline structure with high stereochemical fidelity.
How to Synthesize L-Octahydroindoline-2-Carboxylic Acid Efficiently
The execution of this synthesis requires careful control of reaction parameters, particularly during the hydrogenation and resolution phases, to ensure consistent quality and yield. The process begins with the preparation of ortho-nitrophenylpyruvic acid via condensation, followed by a series of catalytic reductions that build the core heterocyclic structure. Operators must maintain strict control over hydrogen pressure and temperature during the reduction steps to prevent side reactions, while the resolution phase demands precise pH adjustments and temperature management to maximize the recovery of the desired enantiomer. For a detailed breakdown of the specific operating conditions, reagent ratios, and workup procedures required to implement this technology in a pilot or production plant, please refer to the standardized synthesis guide below.
- Condense diethyl oxalate with ortho-nitrotoluene using sodium ethylate to form ortho-nitrophenylpyruvic acid, followed by catalytic hydrogenation to generate indole-2-carboxylic acid.
- Perform acylation with acetic anhydride and subsequent hydrogenation to obtain a racemic mixture of N-acetyl indoline-2-carboxylic acid.
- Resolve the racemic mixture to isolate the S-enantiomer, recycle the R-enantiomer via racemization, and finally hydrogenate the aromatic ring using a rhodium catalyst to yield the target L-octahydroindoline derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend far beyond simple chemical yield improvements. By shifting away from the traditional Fischer synthesis, manufacturers can eliminate the need for expensive and hazardous phenylhydrazine derivatives, replacing them with commodity chemicals like ortho-nitrotoluene and diethyl oxalate which are widely available and cost-effective. This substitution drastically reduces the raw material cost basis and mitigates supply risks associated with specialty reagents. Furthermore, the reduction in wastewater generation and the elimination of harsh acidic waste streams translate directly into lower environmental compliance costs and reduced fees for waste disposal, contributing to a leaner and more sustainable cost structure for the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The implementation of a racemization recycling loop for the unwanted chiral isomer represents a profound efficiency gain that directly impacts the cost of goods sold. In traditional resolution processes, fifty percent of the material is often lost or requires complex reprocessing, but this method allows for the continuous conversion of the R-enantiomer back into the usable pool. Additionally, the use of robust heterogeneous catalysts like Raney Nickel and Rhodium on carbon, which can often be recovered and reused, minimizes the consumption of expensive precious metals. These factors combined result in a significantly optimized production cost profile without compromising the high purity required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals as starting materials ensures a stable and resilient supply chain that is less susceptible to market volatility. Ortho-nitrotoluene and diethyl oxalate are produced on a massive global scale for various industrial applications, guaranteeing consistent availability and competitive pricing. This contrasts sharply with routes dependent on niche intermediates that may have limited suppliers or long lead times. By securing a synthesis path based on foundational chemical feedstocks, procurement teams can negotiate better long-term contracts and ensure uninterrupted production schedules for their downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The unit operations described in this patent, primarily hydrogenation and crystallization, are inherently scalable and well-understood within the chemical industry. Hydrogenation reactors are standard equipment in most fine chemical plants, allowing for easy technology transfer from laboratory to commercial scale without the need for specialized or exotic infrastructure. Moreover, the cleaner nature of the process, characterized by reduced solvent usage and minimal aqueous waste, simplifies the permitting process and aligns with increasingly stringent global environmental regulations. This makes the facility more future-proof against tightening ecological standards and reduces the operational burden on environmental health and safety teams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of L-octahydroindoline-2-carboxylic acid using this advanced catalytic method. These insights are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity on process capabilities and quality control measures. Understanding these details is essential for partners evaluating the feasibility of integrating this intermediate into their own drug development pipelines.
Q: What are the primary advantages of this hydrogenation route over traditional Fischer synthesis?
A: Unlike the Fischer synthesis which generates substantial wastewater and requires harsh acidic conditions for rearrangement, this patented method utilizes a clean catalytic hydrogenation pathway. This significantly reduces environmental waste treatment costs and improves the overall atom economy of the process.
Q: How is chirality controlled in the production of L-octahydroindoline-2-carboxylic acid?
A: Chirality is established through a resolution step using methylbenzylamine to separate the R and S enantiomers of the indoline intermediate. Crucially, the unwanted R-enantiomer is not discarded but is subjected to a racemization process under alkaline conditions, allowing it to be recycled back into the S-enantiomer stream, thereby maximizing yield.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process relies heavily on standard unit operations such as catalytic hydrogenation and crystallization, which are highly scalable in industrial settings. The use of robust catalysts like Raney Nickel and Rhodium on carbon ensures consistent performance across large batch sizes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Octahydroindoline-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of L-octahydroindoline-2-carboxylic acid meets the highest international standards. Our capability to implement complex catalytic hydrogenation and chiral resolution technologies positions us as a strategic partner capable of delivering both technical excellence and supply security.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient process can optimize your overall budget. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can accelerate your time to market.
