Advanced Hydrogenation Strategy for L-Octahydroindoline-2-Formic Acid: Scaling High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for complex chiral intermediates that balance cost-efficiency with environmental sustainability. Patent CN101823993A introduces a groundbreaking preparation method for L-octahydroindoline-2-formic acid, a critical building block in the synthesis of various bioactive molecules and potential drug candidates. This technology departs from traditional, waste-intensive methodologies by leveraging a sophisticated sequence of condensation and catalytic hydrogenation reactions starting from readily available raw materials like diethyl oxalate and ortho-nitrotoluene. The disclosed process not only achieves a high yield of the final finished product but also addresses the growing regulatory pressure for cleaner manufacturing processes by minimizing wastewater generation and eliminating the need for hazardous reagents often associated with older synthetic routes.
For R&D directors and process chemists, the significance of this patent lies in its ability to construct the octahydroindoline scaffold with precise stereochemical control. The method integrates a clever resolution and racemization loop that ensures the efficient utilization of chiral precursors, thereby enhancing the overall atom economy of the synthesis. By shifting the paradigm from stoichiometric reductions to catalytic hydrogenation, the technology offers a scalable solution that maintains stringent purity specifications essential for pharmaceutical applications. This report analyzes the technical nuances of this innovation, providing a comprehensive evaluation of its feasibility for commercial adoption and its potential to redefine supply chain dynamics for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of indole and its derivatives has heavily relied on the Fischer indole synthesis, a method that, while classic, presents substantial drawbacks for modern large-scale manufacturing. The traditional Fischer approach typically involves the thermal rearrangement of phenylhydrazones under acidic catalysis, a process that necessitates the use of phenylhydrazine, a reagent known for its toxicity and high cost. Furthermore, this rearrangement reaction invariably produces ammonia as a byproduct, leading to significant challenges in waste management and requiring extensive downstream processing to neutralize and treat the effluent. The generation of large volumes of wastewater not only increases the environmental footprint of the manufacturing facility but also escalates operational costs related to waste disposal and regulatory compliance.
In addition to environmental concerns, the conventional methods often suffer from poor atom economy and limited scalability. The requirement for harsh acidic conditions and high temperatures can lead to the formation of complex impurity profiles, complicating the purification process and reducing the overall yield of the desired intermediate. For procurement managers and supply chain heads, these inefficiencies translate into higher raw material costs and unpredictable production timelines. The reliance on expensive starting materials like phenylhydrazine, coupled with the need for rigorous safety protocols due to the hazardous nature of the reagents, creates a bottleneck that hinders the cost-effective mass production of indoline-based pharmaceutical intermediates required by the global market.
The Novel Approach
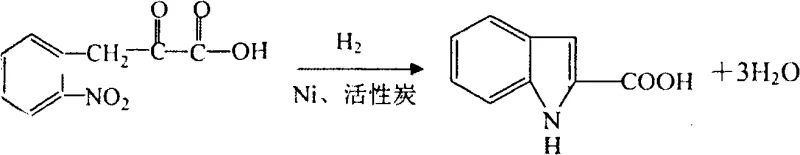
The methodology outlined in patent CN101823993A offers a transformative alternative by utilizing a condensation and hydrogenation strategy that circumvents the pitfalls of the Fischer synthesis. By employing ortho-nitrotoluene and diethyl oxalate as the primary feedstocks, the process accesses the indole core through a cleaner mechanistic pathway that avoids the generation of ammonia and significantly reduces wastewater output. The initial condensation step forms an o-nitrophenylpyruvic acid intermediate, which is then subjected to a catalytic hydrogenation using Raney Nickel and activated carbon. This step simultaneously reduces the nitro group and facilitates cyclization to form indole-2-carboxylic acid with impressive efficiency, reportedly achieving yields around 90% in the pilot examples provided.
Furthermore, the novel approach incorporates a strategic acylation and subsequent hydrogenation sequence to convert the indole derivative into the saturated indoline structure. This multi-stage hydrogenation protocol allows for precise control over the reduction levels, ensuring that the aromaticity of the benzene ring is preserved until the final stage where full saturation is desired. The integration of a chiral resolution step using methylphenethylamine, followed by a racemization loop for the unwanted isomer, ensures that the process maximizes the output of the specific L-enantiomer required for biological activity. This holistic design not only lowers the cost of goods sold (COGS) by recycling materials but also establishes a robust framework for commercial scale-up that aligns with the principles of green chemistry and sustainable manufacturing.
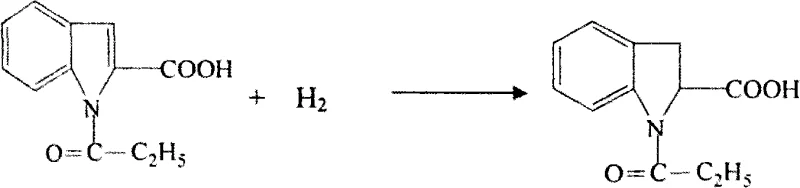
Mechanistic Insights into Catalytic Hydrogenation and Chiral Resolution
The core of this synthetic innovation lies in its sophisticated application of catalytic hydrogenation to achieve structural transformations that are difficult to accomplish via stoichiometric reagents. The initial conversion of o-nitrophenylpyruvic acid to indole-2-carboxylic acid is a tandem reaction where the nitro group is reduced to an amine, which then spontaneously condenses with the adjacent ketone functionality to close the five-membered pyrrole ring. The use of Raney Nickel in conjunction with activated carbon provides a highly active surface for hydrogen adsorption, facilitating the rapid transfer of hydrogen atoms to the substrate. This catalytic system is particularly effective in suppressing side reactions such as over-reduction of the newly formed double bonds or the cleavage of sensitive functional groups, thereby maintaining the integrity of the indole scaffold which is crucial for subsequent derivatization.
Following the formation of the indole core, the process employs acetic anhydride to protect the nitrogen atom via acylation, forming an N-acyl indole intermediate. This protection step is critical as it modulates the electron density of the pyrrole ring, making it more susceptible to the subsequent catalytic hydrogenation that reduces the C2-C3 double bond to form the indoline structure. The hydrogenation of the acylated indole is conducted under controlled pressure and temperature conditions, typically using glacial acetic acid as a solvent to ensure solubility and stability of the intermediates. The precipitation of the product upon completion of the reaction simplifies the isolation process, allowing for the recovery of a faint yellow solid that serves as the precursor for the chiral resolution stage.
The stereochemical integrity of the final product is secured through a dynamic kinetic resolution strategy. The racemic (R,S) indoline-2-carboxylic acid mixture is treated with a chiral resolving agent, specifically methylphenethylamine, in a dehydrated alcohol solvent. This forms diastereomeric salts that exhibit different solubilities, allowing for the selective crystallization of the desired isomer. A key feature of this mechanism is the treatment of the mother liquor containing the unwanted isomer; by subjecting it to alkaline conditions at elevated temperatures (170-185°C), the chiral center is epimerized, effectively racemizing the material. This racemized material is then fed back into the resolution loop, theoretically allowing for nearly 100% conversion of the racemic starting material into the single desired enantiomer, a feat that significantly enhances the economic viability of the process.
How to Synthesize L-Octahydroindoline-2-Formic Acid Efficiently
The execution of this synthesis requires precise control over reaction parameters to ensure safety and reproducibility, particularly during the high-pressure hydrogenation steps. The process begins with the careful preparation of the o-nitrophenylpyruvic acid intermediate, followed by the critical hydrogenation steps that build the core heterocyclic structure. Operators must adhere to strict temperature and pressure protocols, especially during the final saturation of the benzene ring which utilizes a precious metal catalyst. The detailed standardized synthesis steps below outline the specific operational procedures derived from the patent embodiments, providing a clear roadmap for laboratory validation and pilot plant trials.
- Condense ortho-nitrotoluene with diethyl oxalate using sodium ethylate, followed by hydrolysis to obtain o-nitrophenylpyruvic acid.
- Perform catalytic hydrogenation using Raney Nickel and activated carbon to cyclize and reduce the nitro group, forming indole-2-carboxylic acid.
- Execute acylation with acetic anhydride followed by a second hydrogenation step to generate the racemic indoline-2-carboxylic acid mixture.
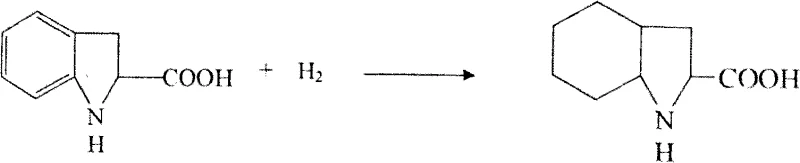
- Resolve the racemic mixture using chiral amines, recycle the unwanted isomer via racemization, and perform final rhodium-catalyzed hydrogenation to saturate the benzene ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of the technology described in CN101823993A presents a compelling value proposition centered on cost optimization and supply security. The shift away from the Fischer synthesis eliminates the dependency on phenylhydrazine, a volatile and costly reagent subject to fluctuating market prices and strict regulatory controls. By substituting this with ortho-nitrotoluene and diethyl oxalate, both of which are commodity chemicals produced on a massive global scale, manufacturers can secure a more stable and predictable raw material supply base. This fundamental change in the bill of materials (BOM) drastically reduces the exposure to supply chain disruptions and allows for more accurate long-term cost forecasting.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the high atom economy and the efficient recycling of chiral intermediates. The implementation of the racemization loop means that the theoretical yield of the valuable L-enantiomer is no longer capped at 50%, effectively doubling the output from the resolution step without additional raw material input. Furthermore, the use of heterogeneous catalysts like Raney Nickel and Rhodium on carbon allows for the potential recovery and reuse of the catalytic metals, further driving down the variable costs per kilogram. The simplified workup procedures, characterized by filtration and crystallization rather than complex extractions or chromatography, also contribute to significant savings in labor and solvent consumption.
- Enhanced Supply Chain Reliability: The robustness of the hydrogenation route ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply to downstream pharmaceutical customers. The process is less sensitive to minor variations in reaction conditions compared to the thermal rearrangements of the Fischer method, resulting in a more reliable production schedule. Additionally, the reduced generation of hazardous waste simplifies the logistics of waste disposal and minimizes the risk of production stoppages due to environmental compliance issues. This reliability translates directly into shorter lead times and the ability to respond swiftly to surges in market demand for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From a facility perspective, the cleaner nature of this synthesis reduces the burden on wastewater treatment plants, allowing existing infrastructure to support higher production volumes without costly upgrades. The elimination of ammonia emissions and the reduction in acidic waste streams align perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against regulatory tightening. The scalability of catalytic hydrogenation is well-established in the fine chemical industry, meaning that transferring this process from pilot scale to multi-ton commercial production involves manageable engineering risks, ensuring a smooth ramp-up to meet commercial requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, aiming to clarify the operational advantages and chemical principles for potential partners and stakeholders.
Q: What are the primary environmental advantages of this synthesis route compared to traditional Fischer indole synthesis?
A: Unlike the Fischer synthesis which generates significant ammonia waste and requires harsh acidic rearrangement conditions, this patented method utilizes a cleaner catalytic hydrogenation pathway. The process significantly reduces wastewater generation and avoids the use of expensive phenylhydrazine reagents, resulting in a more environmentally compliant manufacturing profile suitable for modern green chemistry standards.
Q: How does the process ensure high optical purity for the L-enantiomer?
A: The process employs a robust chiral resolution strategy using methylphenethylamine to separate the (R,S) indoline-2-carboxylic acid mixture. Crucially, the unwanted isomer is not discarded but is subjected to a racemization loop under alkaline conditions, allowing it to be recycled back into the resolution step. This iterative approach maximizes the theoretical yield of the desired S-isomer before the final hydrogenation step.
Q: What catalysts are critical for the successful scale-up of this reaction sequence?
A: The synthesis relies on a dual-catalyst system tailored for specific reduction stages. Raney Nickel combined with activated carbon is utilized for the initial nitro reduction and cyclization, while a specialized 5% Rhodium catalyst is employed in the final high-pressure hydrogenation step to fully saturate the aromatic benzene ring without compromising the stereochemistry of the adjacent chiral center.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Octahydroindoline-2-Formic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development programs. Our team of expert process chemists has extensively evaluated the pathway described in CN101823993A and possesses the technical capability to optimize and scale this route for industrial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced hydrogenation reactors capable of handling high-pressure reactions safely, guaranteeing that every batch of L-octahydroindoline-2-formic acid meets stringent purity specifications required by global regulatory bodies.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced synthetic technology for their pipeline projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing expertise can accelerate your development timelines and reduce your overall cost of goods. Let us be your strategic partner in bringing next-generation therapeutics to market efficiently and sustainably.
