Optimizing 2,3-Dihydrothiochromen-4-one Production: A Strategic Shift for Global Pharmaceutical Supply Chains
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with economic viability, particularly for complex heterocyclic scaffolds like 2,3-dihydrothiochromen-4-one. Patent CN110698456A introduces a transformative methodology that addresses long-standing bottlenecks in the production of this critical pharmaceutical intermediate. By shifting away from regulated precursors and viscous catalysts, this innovation offers a streamlined pathway that enhances both operational safety and environmental compliance. For global supply chain leaders, understanding the nuances of this patent is essential for securing reliable sources of high-value anti-inflammatory and anti-tumor precursors. This report analyzes the technical merits of replacing polyphosphoric acid cyclization with a concentrated sulfuric acid protocol, demonstrating how such process intensification can drive substantial value across the entire manufacturing lifecycle.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
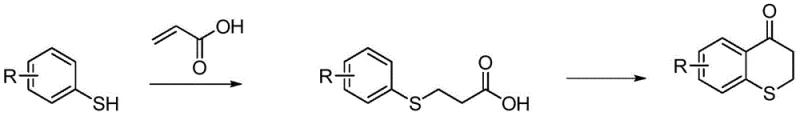
Historically, the synthesis of 2,3-dihydrothiochromen-4-one derivatives has relied heavily on the reaction between thiophenols and 3-chloropropionic acid, followed by cyclization using polyphosphoric acid (PPA). This legacy approach presents severe logistical and technical challenges that hinder efficient commercial scale-up. Firstly, 3-chloropropionic acid is frequently classified as a controlled precursor in many jurisdictions, creating significant procurement hurdles and regulatory burdens for purchasing departments. Secondly, polyphosphoric acid is notoriously viscous and difficult to handle on an industrial scale, complicating mixing and heat transfer operations which are critical for reaction homogeneity. Furthermore, the post-treatment of PPA reactions generates substantial quantities of phosphorus-containing wastewater, imposing heavy costs on waste management systems and complicating environmental compliance audits. These factors collectively result in lower overall yields and inflated production costs, making the conventional route increasingly unattractive for modern, sustainability-focused manufacturing facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN110698456A utilizes readily available acrylic acid and concentrated sulfuric acid to achieve the same structural transformation with superior efficiency. The process initiates with a Michael addition between aromatic thiophenols and acrylic acid to form an aromatic thiopropionic acid intermediate, followed by an intramolecular cyclization. This strategic substitution of raw materials eliminates the dependency on controlled chloropropionic acids, thereby stabilizing the supply chain against regulatory fluctuations. The use of concentrated sulfuric acid as the cyclization agent not only simplifies the feeding process due to its lower viscosity compared to PPA but also facilitates a much cleaner workup procedure involving simple acidification and extraction. This novel approach effectively decouples production capacity from complex waste treatment requirements, enabling manufacturers to achieve higher throughput with reduced operational friction.
Mechanistic Insights into Sulfuric Acid-Catalyzed Cyclization
The core chemical innovation lies in the efficient two-step sequence that leverages the nucleophilicity of the thiol group and the electrophilicity of the activated aromatic ring. In the first stage, the aromatic thiophenol undergoes a conjugate addition (Michael addition) to acrylic acid in the presence of an organic base such as triethylamine or pyridine. This step is typically conducted at low temperatures, ranging from -5°C to 5°C, to control exothermicity and prevent polymerization of the acrylic acid, ensuring the selective formation of the 3-aromatic thiopropionic acid intermediate. The precise control of stoichiometry, with a molar ratio of thiophenol to acrylic acid between 1:1 and 1:1.2, is critical to minimizing unreacted starting materials and side products. Following isolation, the intermediate is subjected to strong acidic conditions where the carboxylic acid moiety is activated by protonation, facilitating an intramolecular Friedel-Crafts acylation.
During the cyclization phase, the concentrated sulfuric acid serves a dual role as both a dehydrating agent and a strong Brønsted acid catalyst. Heating the reaction mixture to 90°C-110°C provides the necessary activation energy for the electrophilic aromatic substitution to occur, closing the six-membered ring to form the thiochroman-4-one core. The mechanism ensures high regioselectivity, particularly when dealing with substituted thiophenols, as the sulfur atom directs the incoming acyl group to the ortho position. Crucially, this acidic environment suppresses the formation of sulfonated byproducts that might occur under harsher conditions, thereby maintaining a clean impurity profile. The ability to quench the reaction simply by pouring into ice water allows for the rapid precipitation or extraction of the product, avoiding the emulsions often encountered with phosphate-based systems and ensuring consistent batch-to-batch quality.
How to Synthesize 2,3-Dihydrothiochromen-4-one Efficiently
Implementing this synthesis route requires careful attention to temperature gradients and reagent addition rates to maximize yield and safety. The process begins with the dissolution of the thiophenol substrate in a polar aprotic solvent like tetrahydrofuran (THF) or dioxane, followed by the addition of an organic base to scavenge protons generated during the thiol-acrylate coupling. Detailed standardized operating procedures for temperature control and workup are essential for reproducibility. For a comprehensive breakdown of the specific experimental parameters and stoichiometric ratios required for successful execution, please refer to the step-by-step guide below.
- Dissolve substituted aromatic thiophenol in an organic solvent like THF, add an organic base such as triethylamine, and control temperature between -5°C to 5°C while dropwise adding acrylic acid.
- After the initial low-temperature reaction, allow the mixture to stir at room temperature until completion, then acidify to pH 1-2 and extract to isolate the aromatic thiopropionic acid intermediate.
- Add the isolated intermediate into concentrated sulfuric acid (80-98%) and heat to 90°C-110°C to effect ring closure, followed by quenching in ice water and standard extraction workup.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for procurement managers and supply chain directors aiming to optimize cost structures and mitigate risk. The transition from specialized, regulated precursors to commodity chemicals fundamentally alters the cost basis of the final intermediate. By utilizing acrylic acid, a bulk chemical produced on a massive global scale, manufacturers can secure raw materials at significantly lower price points with guaranteed availability. This shift reduces the vulnerability of the supply chain to geopolitical restrictions or sudden regulatory changes that often affect halogenated carboxylic acids. Furthermore, the simplified downstream processing reduces the consumption of solvents and utilities, contributing to a leaner manufacturing model that is more resilient to market volatility.
- Cost Reduction in Manufacturing: The elimination of polyphosphoric acid removes the need for expensive and energy-intensive cleanup procedures associated with phosphorus waste. Since PPA requires extensive washing and neutralization to meet discharge standards, replacing it with sulfuric acid drastically reduces the operational expenditure related to effluent treatment. Additionally, the higher yields reported in the patent examples imply that less raw material is wasted per kilogram of product, directly improving the gross margin. The use of common organic bases and solvents further ensures that the input costs remain stable and predictable, allowing for more accurate financial forecasting and competitive pricing strategies in the global marketplace.
- Enhanced Supply Chain Reliability: Sourcing 3-chloropropionic acid can be a bottleneck due to its dual-use nature and varying international transport regulations. By switching to acrylic acid, which is a standard feedstock for the polymer and coating industries, the supply chain becomes far more robust and less prone to disruption. The ease of handling liquid reagents compared to viscous pastes like PPA also accelerates the turnover time of reactor vessels, increasing the overall asset utilization rate of the production facility. This fluidity in operations ensures that delivery schedules can be met consistently, reinforcing the manufacturer's reputation as a dependable partner for downstream pharmaceutical clients who operate on tight just-in-time inventory models.
- Scalability and Environmental Compliance: The environmental benefits of this process are substantial, particularly regarding waste minimization and hazard reduction. The absence of phosphorus in the waste stream simplifies the permitting process for new production lines and reduces the liability associated with long-term environmental monitoring. The reaction conditions are amenable to continuous flow chemistry or large batch processing, offering flexibility in scaling from pilot plant quantities to multi-ton commercial campaigns. This scalability ensures that the technology can grow alongside market demand without requiring fundamental re-engineering of the process infrastructure, making it a future-proof investment for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process robustness and product quality. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer.
Q: Why is the acrylic acid route preferred over the traditional 3-chloropropionic acid method?
A: The traditional method relies on 3-chloropropionic acid, which is often a controlled substance with procurement difficulties, and uses polyphosphoric acid (PPA) which is viscous and generates hazardous phosphorus wastewater. The new route uses commodity acrylic acid and concentrated sulfuric acid, simplifying handling and waste treatment.
Q: What are the critical temperature controls for the cyclization step?
A: The cyclization step requires heating the reaction mixture in concentrated sulfuric acid to a range of 90°C to 110°C. Maintaining this temperature window is crucial for ensuring complete ring closure while minimizing thermal degradation of the sensitive thiochromanone core.
Q: How does this process impact environmental compliance in large-scale manufacturing?
A: By eliminating polyphosphoric acid, the process avoids the generation of large volumes of phosphorus-containing wastewater, which is difficult and costly to treat. The use of sulfuric acid allows for simpler neutralization and extraction protocols, significantly reducing the environmental footprint.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydrothiochromen-4-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to more efficient synthetic routes is critical for maintaining competitiveness in the pharmaceutical intermediate sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN110698456A are fully realized in practical manufacturing settings. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2,3-dihydrothiochromen-4-one meets the exacting standards required for drug substance synthesis. Our commitment to process excellence means we can deliver high-quality intermediates with consistent impurity profiles, supporting your R&D and commercialization goals with unwavering reliability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this sulfuric acid-mediated protocol. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume needs. Let us collaborate to build a more efficient, cost-effective, and sustainable supply chain for your next-generation therapeutic programs.
