Revolutionizing Topramezone Intermediate Production via Copper-Catalyzed Mesylation
Revolutionizing Topramezone Intermediate Production via Copper-Catalyzed Mesylation
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing. Topramezone, a potent HPPD inhibitor developed for corn crop protection, relies heavily on the availability of its key building block, 2,3-dimethyl-4-methanesulfonyl bromobenzene. Recent intellectual property developments, specifically Chinese Patent CN112321466A, have unveiled a transformative green synthesis pathway that addresses long-standing environmental and operational challenges in producing this critical molecule. This technical insight report analyzes the breakthrough methodology disclosed in the patent, highlighting its potential to redefine supply chain standards for reliable agrochemical intermediate suppliers. By shifting away from traditional, odor-intensive sulfide chemistry toward a controlled copper-catalyzed system, manufacturers can achieve superior purity profiles while mitigating significant EHS risks associated with volatile sulfur compounds.
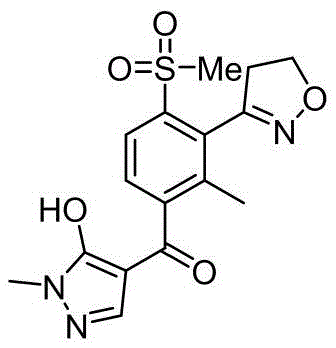
The structural complexity of Topramezone necessitates precise functionalization of the aromatic core, particularly the installation of the methylsulfonyl group at the para-position relative to the amino precursor. Conventional literature, such as the foundational work by BASF (US20030216580), has historically relied on Sandmeyer reactions involving dimethyl disulfide. While chemically effective, these legacy processes generate methyl mercaptan, a compound notorious for its pervasive and offensive odor, posing severe containment challenges in large-scale facilities. The novel approach detailed in CN112321466A circumvents these issues entirely by utilizing sodium methanesulfinate as the sulfur source. This strategic substitution not only eliminates the generation of noxious gases but also streamlines the reaction workflow, offering a compelling value proposition for cost reduction in herbicide manufacturing through simplified waste treatment and improved worker safety protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dimethyl-4-methanesulfonyl bromobenzene has been bottlenecked by the inherent hazards of thiol-based chemistry. Traditional routes typically involve the diazotization of 2,3-dimethylaniline followed by reaction with dimethyl disulfide to install the methylthio group, which is subsequently oxidized to the sulfone. This multi-step sequence is fraught with operational difficulties; the handling of low-valence sulfides requires specialized scrubbing systems and negative pressure environments to prevent fugitive emissions. Furthermore, the oxidation step often necessitates strong oxidants that can lead to over-oxidation or side reactions, complicating the impurity profile and reducing overall yield. For procurement managers, these factors translate into higher capital expenditure for safety infrastructure and increased operational costs for environmental compliance, making the conventional route less attractive for modern, sustainability-focused production facilities.
The Novel Approach
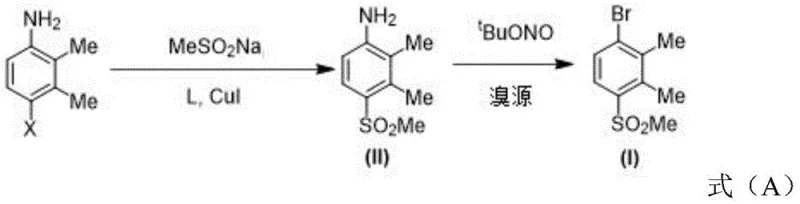
The innovative strategy presented in the patent fundamentally reimagines the construction of the carbon-sulfur bond. Instead of relying on foul-smelling thiols, the process employs a direct copper-catalyzed coupling between a halo-aniline derivative and sodium methanesulfinate. This reaction proceeds efficiently in polar aprotic solvents like DMSO, leveraging specific ligand-accelerated catalysis to achieve high conversion rates. Following the mesylation, the aniline moiety is converted to the bromide via a streamlined Sandmeyer reaction using tert-butyl nitrite and a bromine source. This two-step sequence is remarkably concise and robust. By avoiding the isolation of unstable thiol intermediates and eliminating the need for harsh oxidation steps post-coupling, the new method significantly enhances process safety and throughput, positioning it as a preferred route for the commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into CuI-Catalyzed C-S Bond Formation
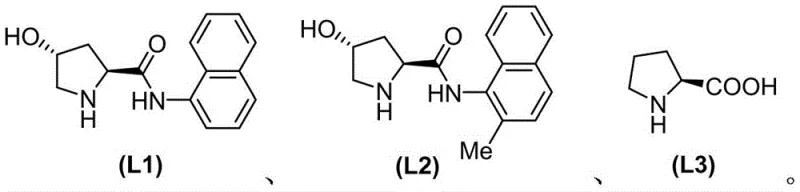
The core of this technological advancement lies in the efficiency of the copper-catalyzed mesylation step. The reaction utilizes copper(I) iodide (CuI) as the catalyst precursor, activated by bidentate nitrogen-containing ligands. The patent explicitly identifies three proline-derived ligands (L1, L2, and L3) as effective promoters for this transformation. Mechanistically, the copper center facilitates the oxidative addition of the aryl halide bond, followed by transmetallation with the sulfinate anion and subsequent reductive elimination to forge the C-S bond. The choice of ligand is critical; experimental data within the patent demonstrates that Ligand L1, featuring a naphthyl-amide backbone, provides superior steric and electronic stabilization of the copper species compared to L2 or L3. This results in markedly higher yields, with optimization studies showing yields jumping from modest levels to over 80% simply by adjusting the ligand and temperature parameters.
Furthermore, the subsequent Sandmeyer bromination is executed under mild conditions that preserve the integrity of the newly formed sulfone group. The use of tert-butyl nitrite as the diazotizing agent allows for the generation of the diazonium salt in situ, which is immediately trapped by the bromine source (either CuBr2 or tetrabutylammonium bromide). This tandem-like efficiency minimizes the accumulation of potentially explosive diazonium salts, enhancing process safety. From an impurity control perspective, the specificity of the CuI/ligand system ensures that dehalogenation or homocoupling side products are kept to a minimum. The rigorous control of reaction atmosphere, specifically the use of nitrogen protection to prevent CuI oxidation, further guarantees consistent batch-to-bquality, a critical metric for high-purity agrochemical intermediates destined for final API synthesis.
How to Synthesize 2,3-Dimethyl-4-methanesulfonyl Bromobenzene Efficiently
Implementing this synthesis requires careful attention to the catalytic loading and thermal profiles described in the patent embodiments. The process begins with the preparation of the halo-aniline starting material, followed by the pivotal mesylation step in DMSO. Operators must maintain strict anaerobic conditions to preserve catalyst activity. Following the coupling, the workup involves standard extraction and purification techniques, yielding the sulfone-aniline intermediate which is then subjected to bromination. The detailed standardized synthesis steps, including precise stoichiometric ratios and temperature ramps for optimal yield, are outlined below to guide process development teams in replicating this efficient pathway.
- Perform mesylation of 4-halo-2,3-dimethylaniline with sodium methanesulfinate using CuI and a specific ligand (L1, L2, or L3) in DMSO under nitrogen protection.
- Conduct a one-step Sandmeyer bromination on the resulting aniline intermediate using tert-butyl nitrite and a bromine source (CuBr2 or TBAB) to yield the target bromobenzene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the adoption of this green synthesis route offers tangible strategic benefits beyond mere chemical elegance. The most immediate impact is the drastic simplification of EHS (Environment, Health, and Safety) management. By eliminating the use of malodorous dimethyl disulfide and the generation of methyl mercaptan, facilities can operate with reduced ventilation loads and lower risk of community odor complaints. This directly correlates to substantial cost savings in terms of regulatory compliance and waste treatment infrastructure. Additionally, the reliance on sodium methanesulfinate, a stable and commercially available solid salt, improves raw material security compared to volatile liquid sulfides, thereby reducing lead time for high-purity intermediates and ensuring continuous production schedules even during supply chain disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous scrubbing systems required for thiol handling significantly lowers capital and operational expenditures. Furthermore, the high yields achieved with Ligand L1 reduce raw material consumption per kilogram of product, driving down the overall cost of goods sold without compromising quality standards.
- Enhanced Supply Chain Reliability: The starting materials, such as 4-halo-2,3-dimethylanilines, are readily accessible from established chemical suppliers. The robustness of the copper-catalyzed step tolerates minor variations in feedstock quality better than sensitive thiol-based chemistries, ensuring a steady flow of intermediates to downstream formulation plants.
- Scalability and Environmental Compliance: The process operates in common solvents like DMSO and acetonitrile, which are easily recovered and recycled on a large scale. The absence of toxic sulfur byproducts simplifies wastewater treatment, aligning perfectly with increasingly stringent global environmental regulations and supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims within CN112321466A, providing clarity on reaction conditions, catalyst selection, and scalability potential for industrial partners evaluating this technology for their production portfolios.
Q: What is the primary advantage of this new synthesis method over conventional routes?
A: The primary advantage is the elimination of malodorous low-valence sulfides like dimethyl disulfide. By using sodium methanesulfinate in a copper-catalyzed system, the process becomes odor-free and significantly more suitable for industrial scale-up and environmental compliance.
Q: Which ligands are effective for the copper-catalyzed mesylation step?
A: The patent specifies three proline-derived ligands (L1, L2, and L3). Experimental data indicates that Ligand L1 generally provides superior yields (up to 87%) compared to L2 (13%) under optimized temperature conditions.
Q: Is this process scalable for commercial herbicide production?
A: Yes, the method is designed for industrial production. It utilizes common solvents like DMSO and acetonitrile, avoids hazardous gaseous reagents, and operates under relatively mild conditions (80-120°C), facilitating safe commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dimethyl-4-methanesulfonyl Bromobenzene Supplier
As the agrochemical sector evolves towards greener and more efficient manufacturing paradigms, NINGBO INNO PHARMCHEM stands at the forefront of translating laboratory innovations into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and compliant. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the exacting standards required for herbicide synthesis.
We invite global partners to leverage our technical expertise to optimize their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can enhance your competitive advantage in the global agrochemical market.
