Scalable Manufacturing of 3,4-Dichloro-2-Fluoroaniline via Optimized Nitration and Fluorination
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology intermediates, particularly those required for next-generation tyrosine kinase inhibitors. Patent CN114230471A introduces a highly efficient preparation method for 3,4-dichloro-2-fluoroaniline, a pivotal building block in the synthesis of Tesevatinib. This novel approach addresses significant challenges in regioselectivity and process safety by utilizing 1,2,3-trichlorobenzene as a cost-effective starting material. The disclosed methodology integrates classical organic transformations including nitration, ammonolysis, diazotization, and reduction into a cohesive, controllable sequence. For R&D directors and procurement specialists, this patent represents a strategic opportunity to secure a reliable supply chain for high-purity pharmaceutical intermediates while mitigating the risks associated with complex fluorination chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional routes to fluorinated anilines often suffer from poor regiocontrol and the use of hazardous reagents that complicate waste management and increase operational costs. Direct fluorination of chlorinated benzenes frequently results in mixtures of isomers that require energy-intensive purification steps, drastically reducing overall yield and economic viability. Furthermore, many existing processes rely on expensive transition metal catalysts or harsh conditions that pose significant safety risks during scale-up. The inability to precisely direct the substitution pattern on the aromatic ring leads to substantial material loss and inconsistent batch quality, which is unacceptable for GMP-grade API intermediate manufacturing. These inefficiencies create bottlenecks in the supply chain, leading to extended lead times and volatile pricing structures for downstream drug manufacturers.
The Novel Approach
The method described in CN114230471A overcomes these hurdles by employing a stepwise functionalization strategy that leverages the electronic properties of the substrate to guide reactivity. By initiating the synthesis with 1,2,3-trichlorobenzene, the process ensures that the nitro group is introduced at the most favorable position, setting the stage for a highly selective nucleophilic aromatic substitution. The subsequent conversion of the amine to a diazonium salt allows for the introduction of fluorine via the Balz-Schiemann reaction, a technique known for its reliability in forming carbon-fluorine bonds without requiring exotic reagents. This logical progression not only simplifies the operational workflow but also enhances the purity profile of the final product, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Nitration and Balz-Schiemann Fluorination
The core of this synthetic strategy lies in the precise manipulation of electron density on the benzene ring to achieve specific substitution patterns. In the initial nitration step, the presence of three chlorine atoms deactivates the ring, yet the reaction conditions utilizing mixed acid at 50-60°C are optimized to drive the formation of 2,3,4-trichloronitrobenzene with minimal byproduct formation. The subsequent ammonolysis is a classic bimolecular nucleophilic substitution where the strong electron-withdrawing nature of the nitro group activates the adjacent carbon-chlorine bond for attack by ammonia. This electronic activation is crucial, as it allows the reaction to proceed under relatively moderate temperatures compared to unactivated aryl chlorides, thereby preserving the integrity of the molecular scaffold.
Following the formation of the aniline derivative, the introduction of fluorine is achieved through a diazotization-decomposition sequence that exemplifies the utility of the Balz-Schiemann reaction. The amine is converted into a diazonium tetrafluoroborate salt using fluoroboric acid and sodium nitrite at low temperatures, ensuring the stability of the reactive intermediate. Upon heating in a high-boiling solvent like o-xylene, the diazonium group decomposes to release nitrogen gas and generate a highly reactive aryl cation, which is immediately trapped by the fluorine atom from the counterion. This mechanism avoids the use of dangerous elemental fluorine or corrosive hydrogen fluoride, providing a safer and more manageable pathway for introducing the fluoro substituent essential for the biological activity of the final drug molecule.
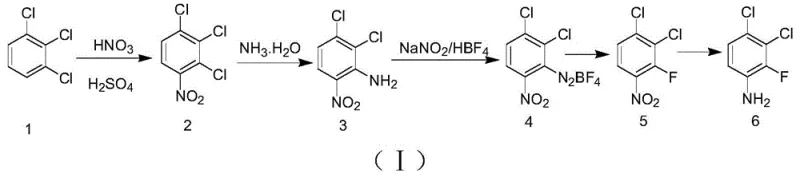
How to Synthesize 3,4-Dichloro-2-Fluoroaniline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this valuable intermediate with high consistency and yield. The process begins with the careful control of exothermic nitration, followed by a high-pressure ammonolysis step that requires robust reactor equipment to handle the ammonia atmosphere safely. The critical fluorination step demands strict temperature control during diazotization to prevent premature decomposition, followed by a controlled thermal breakdown in an inert solvent. Finally, the reduction of the nitro group to the amine is accomplished using economical metal powders, completing the transformation. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this optimized route.
- Nitration of 1,2,3-trichlorobenzene using mixed acid (HNO3/H2SO4) at 50-60°C to yield 2,3,4-trichloronitrobenzene.
- Ammonolysis of the nitro-compound with ammonia water under pressure (0.6-2.0 MPa) at 115-130°C to form 2,3-dichloro-6-nitroaniline.
- Diazotization with fluoroboric acid and sodium nitrite below 5°C, followed by thermal decomposition in o-xylene to introduce fluorine.
- Reduction of the fluoro-nitro intermediate using zinc or iron powder with a proton donor to obtain the final aniline product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain heads. The reliance on commodity chemicals such as 1,2,3-trichlorobenzene, nitric acid, and ammonia ensures that raw material availability is not a bottleneck, significantly enhancing supply chain reliability. The elimination of precious metal catalysts removes a major cost driver and simplifies the purification process, as there is no need for expensive metal scavenging steps. This streamlined approach translates to substantial cost savings in manufacturing, allowing for more competitive pricing in the global market for oncology intermediates. Additionally, the use of standard unit operations facilitates easier technology transfer and scale-up, reducing the time required to bring new batches to market.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and readily available starting materials, avoiding the need for costly specialized reagents or catalysts that often inflate production budgets. By optimizing reaction conditions to maximize yield at each step, the overall material consumption is minimized, leading to a more efficient use of resources. The simplified workup procedures, such as straightforward extractions and crystallizations, reduce solvent usage and energy consumption, further driving down operational expenses. This economic efficiency makes the production of 3,4-dichloro-2-fluoroaniline financially sustainable even in a fluctuating raw material market.
- Enhanced Supply Chain Reliability: Sourcing 1,2,3-trichlorobenzene and other bulk reagents is straightforward due to their widespread production in the fine chemical industry, mitigating the risk of supply disruptions. The robustness of the reaction steps means that minor variations in input quality can be accommodated without compromising the final product specifications, ensuring consistent output. This stability allows suppliers to maintain steady inventory levels and meet delivery schedules reliably, which is critical for pharmaceutical clients managing tight production timelines. The reduced dependency on niche or single-source suppliers strengthens the overall resilience of the supply network.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing reaction types that are well-understood and easily managed in large-scale reactors. The avoidance of highly toxic fluorinating agents reduces the environmental burden and simplifies waste treatment protocols, aligning with increasingly stringent regulatory standards. Efficient atom economy in the key transformation steps minimizes the generation of hazardous byproducts, contributing to a greener manufacturing footprint. These factors combined make the process highly attractive for commercial scale-up of complex pharmaceutical intermediates, ensuring long-term viability and compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 3,4-dichloro-2-fluoroaniline based on the patented technology. Understanding these aspects helps stakeholders evaluate the feasibility and benefits of adopting this specific synthetic route for their supply chains. The answers are derived directly from the technical disclosures and experimental data provided in the patent documentation.
Q: What is the key advantage of using 1,2,3-trichlorobenzene as the starting material?
A: Using 1,2,3-trichlorobenzene allows for precise regioselective nitration and subsequent nucleophilic substitution, avoiding complex separation of isomers often found in direct fluorination routes.
Q: How does the Balz-Schiemann reaction ensure high purity in this process?
A: The formation of the stable diazonium tetrafluoroborate salt minimizes side reactions during the fluorine introduction step, leading to a cleaner profile compared to halogen exchange methods.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process utilizes standard unit operations such as nitration, high-pressure ammonolysis, and metal reduction, which are well-established and easily scalable in industrial GMP facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dichloro-2-Fluoroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications like Tesevatinib. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,4-dichloro-2-fluoroaniline meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex chemistries efficiently, delivering products that support your R&D and manufacturing goals without compromise.
We invite you to collaborate with us to optimize your supply chain and reduce costs through our advanced manufacturing capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can add value to your operations. Partner with us to secure a stable, high-quality source of this essential pharmaceutical intermediate.
