Revolutionizing Palladium Catalyst Precursors via One-Step Acetate Synthesis Technology
Revolutionizing Palladium Catalyst Precursors via One-Step Acetate Synthesis Technology
The landscape of supported palladium catalyst manufacturing is undergoing a significant transformation driven by the urgent need for halogen-free precursors that ensure longevity and efficiency in automotive and fine chemical applications. A pivotal advancement in this domain is detailed in patent CN103450280A, which discloses a novel, one-step synthetic methodology for producing tetraamminepalladium(II) acetate dihydrate. This water-soluble complex, characterized by the formula [Pd(NH3)4](CH3COO)2·2H2O, addresses critical limitations associated with traditional chloride and nitrate-based palladium sources. By leveraging a direct coordination reaction between trinuclear palladium acetate and ammonia, this technology enables the production of ultra-high purity precursors (>99.0%) with exceptional yields (>98.0%), positioning it as a cornerstone for next-generation catalytic systems that demand rigorous environmental compliance and operational stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of supported palladium catalysts has relied heavily on impregnation techniques using precursors such as palladium dichloride or palladium nitrate. These conventional pathways are fraught with inherent chemical liabilities that compromise the final performance of the catalyst. Specifically, the presence of chloride ions in the precursor structure is known to severely inhibit catalytic activity in automotive exhaust purification systems, while nitrate residues can react with moisture to form corrosive nitric acid, damaging the casing of purification units. Furthermore, alternative halogen-free routes often involve multi-step anion exchange processes utilizing expensive reagents like silver acetate or complex ion-exchange resin columns. These legacy methods suffer from difficult endpoint control, potential contamination with silver or chloride ions, and prohibitively high operational costs due to the consumption of precious metal salts and the generation of substantial chemical waste streams.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the innovative strategy outlined in the patent utilizes a direct, one-step coordination reaction that bypasses the need for halogenated starting materials entirely. By reacting commercially available trinuclear palladium acetate [Pd3(CH3COO)6] directly with concentrated ammonia water, the synthesis achieves immediate solubilization and ligand exchange to form the target tetraammine complex. This approach eliminates the introduction of chlorine or nitrate anions at the source, thereby obviating the need for downstream purification steps to remove these harmful impurities. The process is further optimized by using acetone for reverse precipitation, which ensures the isolation of the product as a stable, light-yellow crystalline solid. This streamlined workflow not only simplifies the operational procedure but also drastically enhances the economic viability of producing high-performance catalytic precursors on a commercial scale.
Mechanistic Insights into One-Step Coordination and Thermal Decomposition
The core chemical innovation lies in the unexpected reactivity of trinuclear palladium acetate, a cluster compound previously regarded as chemically stable and insoluble in aqueous alkaline media. Under the specific conditions of 40-50°C in concentrated ammonia, the carboxylate bridges of the trinuclear cluster are cleaved, allowing ammonia molecules to coordinate directly with the palladium centers. This ligand substitution transforms the neutral, hydrophobic cluster into a cationic tetraammine complex paired with acetate counter-ions, resulting in a species with remarkable water solubility (up to 4g/mL). The structural integrity of this complex is crucial for its function as a precursor, as it ensures uniform distribution of the active metal species across the catalyst support during the impregnation phase, leading to superior dispersion of the final metallic palladium particles.
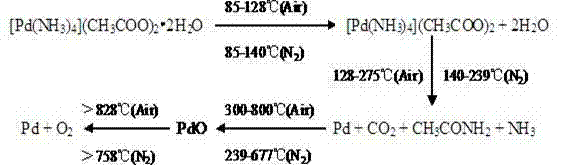
Furthermore, the thermal behavior of this complex offers distinct advantages for catalyst activation. As illustrated in the thermal decomposition pathway, the complex undergoes a clean, multi-stage degradation process upon heating. Initial endothermic events correspond to the loss of crystal water, followed by the decomposition of the ammine and acetate ligands at temperatures between 200°C and 275°C. Crucially, this decomposition leaves behind pure metallic palladium without residual carbonaceous or halogenated contaminants. In oxidative atmospheres, the transient formation of palladium oxide is observed, which subsequently reduces back to the active metal state at higher temperatures. This predictable and clean thermal profile allows manufacturers to precisely tune the calcination parameters to maximize the exposure of active sites, ensuring that the final catalyst exhibits optimal activity for hydrogenation or exhaust gas treatment applications.
How to Synthesize Tetraamminepalladium(II) Acetate Efficiently
The practical implementation of this synthesis route is designed for scalability and ease of operation within standard chemical processing facilities. The protocol avoids the need for specialized high-pressure equipment or cryogenic conditions, relying instead on moderate thermal inputs and common solvent systems. Operators can achieve consistent batch-to-batch reproducibility by strictly adhering to the temperature window of 40-50°C during the dissolution phase and utilizing acetone for the final isolation step. This simplicity reduces the training burden on technical staff and minimizes the risk of operational errors that often plague more complex multi-step syntheses. For a comprehensive breakdown of the standardized operating procedures, including precise stoichiometric ratios and safety handling guidelines, please refer to the technical guide below.
- Dissolve palladium acetate [Pd3(CH3COO)6] in concentrated ammonia water at 40-50°C with stirring until complete solubilization occurs.
- Filter the resulting light yellow solution to remove any unreacted palladium powder or insoluble impurities.
- Concentrate the filtrate under reduced pressure and induce precipitation by adding acetone, followed by drying at 40-50°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthesis technology offers profound implications for cost structures and supply chain resilience in the catalyst manufacturing sector. By shifting away from chloride-based precursors, companies can mitigate the risks associated with corrosion-related equipment failure and the regulatory penalties linked to halogen emissions. The elimination of silver acetate from the process flow removes a significant variable cost driver, as silver salts are notoriously expensive and subject to volatile market pricing. Additionally, the high yield reported in the patent data suggests a maximization of palladium atom economy, ensuring that valuable precious metal inventory is converted into saleable product with minimal loss to mother liquors or filtration cakes. This efficiency translates directly into improved margin protection for manufacturers operating in highly competitive markets.
- Cost Reduction in Manufacturing: The removal of expensive anion exchange reagents such as silver acetate fundamentally alters the cost basis of production. Traditional methods require stoichiometric amounts of silver to precipitate chloride, generating silver chloride waste that must be recovered or disposed of, adding both material and processing costs. The new method utilizes ammonia and acetone, which are commodity chemicals with stable pricing and abundant global supply. Furthermore, the simplified one-step reaction reduces energy consumption by shortening reaction times and eliminating the need for multiple concentration and redissolution cycles, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: Dependence on specialized ion-exchange resins or high-purity silver salts can create bottlenecks in the supply chain, particularly during periods of raw material scarcity. By utilizing palladium acetate and ammonia as primary feedstocks, manufacturers can leverage existing, robust supply networks for these common industrial chemicals. The robustness of the synthesis also implies a lower sensitivity to minor fluctuations in reaction conditions, reducing the rate of batch failures and ensuring a more consistent output of finished goods. This reliability is critical for maintaining just-in-time delivery schedules required by major automotive and pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful transition from laboratory to pilot-scale examples in the patent data. The absence of halogenated byproducts simplifies wastewater treatment protocols, as there is no need for complex dechlorination stages before discharge. This aligns with increasingly stringent environmental regulations regarding industrial effluent quality. Moreover, the use of acetone as a precipitating agent allows for easy solvent recovery and recycling through distillation, further minimizing the environmental footprint and supporting sustainability goals within the manufacturing facility.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this precursor into their existing workflows, we have compiled answers to common inquiries regarding the chemical properties and handling requirements of tetraamminepalladium(II) acetate. These responses are derived directly from the experimental data and characterization results presented in the patent literature, ensuring accuracy and relevance to industrial applications. Understanding these nuances is essential for optimizing catalyst formulation and ensuring safe handling practices during the impregnation and calcination stages.
Q: Why is tetraamminepalladium(II) acetate preferred over palladium chloride for automotive catalysts?
A: Conventional palladium chloride precursors introduce chloride ions which can corrode exhaust system components and poison catalytic activity. The acetate variant described in patent CN103450280A is halogen-free, ensuring longer catalyst life and compliance with strict automotive emission standards.
Q: What represents the primary cost advantage of this new synthesis method?
A: Traditional methods often require expensive silver acetate for anion exchange or complex ion-exchange resin columns. This one-step method utilizes readily available palladium acetate and ammonia, significantly lowering raw material costs and simplifying the purification workflow.
Q: How does the thermal stability of this complex benefit catalyst manufacturing?
A: Thermogravimetric analysis indicates the complex decomposes cleanly into metallic palladium at relatively low temperatures (around 275°C), facilitating efficient activation on support materials without requiring extreme thermal treatment that could damage sensitive substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraamminepalladium(II) Acetate Supplier
The transition to advanced, halogen-free palladium precursors represents a strategic opportunity for chemical manufacturers to enhance the performance and longevity of their catalytic products. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent technology can be fully realized in your supply chain. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of tetraamminepalladium(II) acetate meets the exacting standards required for high-performance automotive and fine chemical catalysts. We understand the critical nature of precursor purity in determining final catalyst activity and are committed to delivering materials that consistently perform.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this chloride-free precursor for your operations. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and quality in your catalyst manufacturing processes.
