Revolutionizing Platinum Catalyst Precursor Production via Novel Iodide Intermediate Route
Revolutionizing Platinum Catalyst Precursor Production via Novel Iodide Intermediate Route
The synthesis of high-purity platinum complexes remains a critical bottleneck in the manufacturing of advanced heterogeneous catalysts and conductive materials. Patent CN101367557A introduces a transformative methodology for producing Tetraammine Platinum(II) Nitrate, a vital precursor for Pt/C and Pt/SiO2 catalysts, by fundamentally re-engineering the reaction pathway. Unlike traditional routes that struggle with low-yielding chloro-intermediates, this innovation leverages the unique coordination chemistry of iodide ligands to achieve overall yields exceeding 90% with product purity reaching 99.0% based on platinum content. For R&D directors and procurement strategists in the fine chemical sector, this patent represents a significant opportunity to decouple production from the oligopolistic supply of expensive intermediates, offering a robust, scalable, and cost-effective alternative for high-purity platinum complex manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tetraammine Platinum(II) Nitrate has been plagued by its reliance on the intermediate dichlorodiammine platinum, [Pt(NH3)2Cl2]. As detailed in the background art of the patent, preparing this specific intermediate from the common starting material K2PtCl4 is an arduous task requiring stringent control over temperature and pH levels. The thermodynamic and kinetic barriers associated with substituting chloride ligands in the presence of ammonia often result in mixed isomer populations and poor conversion rates, with yields typically stagnating below 70%. This technical difficulty has created a market monopoly where only a handful of multinational corporations possess the proprietary know-how to produce this intermediate efficiently. Consequently, downstream manufacturers face exorbitant raw material costs and supply chain vulnerabilities, making the conventional chloride-based route economically unviable for large-scale cost reduction in noble metal chemical manufacturing.
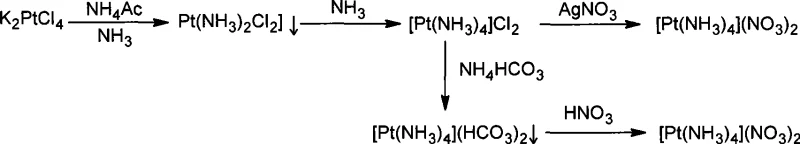
The Novel Approach
The inventive process described in CN101367557A circumvents these historical bottlenecks by introducing an iodide-mediated substitution strategy. Instead of struggling with chloride ligands, the process utilizes potassium iodide (KI) to convert K2PtCl4 into a cis-diiododiammine platinum intermediate, cis-[Pt(NH3)2I2]. This transformation is kinetically favorable and results in a distinct yellow precipitate that can be easily isolated with exceptional efficiency. By shifting the halogen identity from chlorine to iodine, the reaction avoids the formation of intractable polymeric species and ensures a clean, high-yielding pathway. This strategic modification not only simplifies the operational parameters but also drastically improves the atom economy of the platinum usage, establishing a new standard for commercial scale-up of complex platinum precursors.
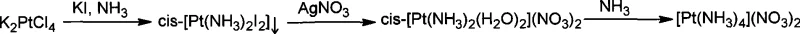
Mechanistic Insights into Iodide-Mediated Ligand Substitution
The success of this novel route lies in the superior leaving group ability of the iodide ion compared to the chloride ion within the platinum coordination sphere. In the first step, the reaction of K2PtCl4 with excess KI likely proceeds through the formation of a transient iodo-species, such as K2PtI4, which then undergoes ammonolysis. The strong trans-effect of the iodide ligand facilitates the subsequent substitution reactions, directing the formation of the desired cis-isomer with high stereoselectivity. The precipitation of cis-[Pt(NH3)2I2] acts as a thermodynamic sink, driving the equilibrium forward and preventing the reverse reaction or the formation of unwanted byproducts. This mechanistic clarity allows process chemists to operate with wider safety margins regarding temperature and pH, reducing the risk of batch failures that are common in the sensitive chloride-based protocols.
Furthermore, the subsequent conversion of the iodo-intermediate to the final nitrate product exploits the insolubility of silver iodide (AgI). When the cis-[Pt(NH3)2I2] is treated with silver nitrate, the iodide ligands are quantitatively removed as AgI precipitate, generating a highly reactive diaqua complex, cis-[Pt(NH3)2(H2O)2](NO3)2, in situ. This aqua complex is extremely labile, allowing for the rapid and complete coordination of additional ammonia molecules in the final step to form the stable tetraammine cation. This sequence ensures that impurities are effectively scrubbed from the system via precipitation (first the iodo-complex, then the silver iodide), resulting in the reported 99.0%+ purity essential for reliable platinum catalyst precursor supplier standards.
How to Synthesize Tetraammine Platinum(II) Nitrate Efficiently
The implementation of this synthesis protocol requires careful attention to stoichiometry and light protection during the initial iodide substitution, as silver salts and certain platinum-iodo species can be photosensitive. The process is designed to be operationally simple, relying primarily on precipitation and filtration rather than complex chromatographic separations or distillations. Below is the structured guide for executing this high-efficiency pathway, optimized for laboratory validation and pilot plant trials.
- React K2PtCl4 with excess KI and ammonia water at 45°C to precipitate cis-[Pt(NH3)2I2].
- Suspend the cis-[Pt(NH3)2I2] intermediate in water and react with stoichiometric silver nitrate to form the aqua complex.
- Treat the resulting filtrate with concentrated ammonia and concentrate under reduced pressure to crystallize the final nitrate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic advantages beyond mere technical feasibility. The primary value driver is the elimination of the dependency on the scarce and expensive [Pt(NH3)2Cl2] intermediate. By sourcing commodity chemicals like potassium iodide and silver nitrate instead of proprietary platinum intermediates, manufacturers can significantly stabilize their raw material costs and mitigate supply risks. Moreover, the dramatic improvement in yield—from a typical 70% in old methods to over 90% in this new process—translates directly into substantial savings on the most expensive component of the bill of materials: the platinum metal itself. In the economics of precious metal chemistry, recovering an additional 20% of platinum input is not just an efficiency gain; it is a massive capital preservation strategy.
- Cost Reduction in Manufacturing: The economic model of this process is bolstered by the high recovery rate of platinum. Since platinum is a high-value asset, minimizing losses during synthesis is paramount. The qualitative shift from a low-yielding, multi-step chloride route to a high-yielding iodide route eliminates the need for extensive recycling loops or reprocessing of mother liquors. Additionally, the reagents used (KI, AgNO3, NH3) are widely available industrial commodities, ensuring that the variable costs of production remain predictable and competitive compared to the volatile pricing of specialized organometallic intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by decoupling production from the limited number of vendors who control the [Pt(NH3)2Cl2] market. By adopting a synthesis route that starts from the universally available K2PtCl4, manufacturers gain autonomy over their production schedules. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high vacuum systems, further ensures that production can be maintained consistently across different geographic locations without reliance on specialized infrastructure, thereby reducing lead time for high-purity platinum complexes.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on unit operations that are standard in the fine chemical industry, such as stirred tank precipitation and vacuum filtration. The generation of solid waste is manageable, primarily consisting of silver iodide, which itself contains recoverable silver and iodine values, adding a secondary revenue stream or cost offset. The aqueous nature of the reactions minimizes the use of volatile organic solvents, aligning the process with modern green chemistry principles and simplifying regulatory compliance for wastewater treatment in large-scale facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the operational benefits for potential licensees and manufacturing partners.
Q: Why is the traditional synthesis of [Pt(NH3)4](NO3)2 considered economically unfeasible?
A: Conventional methods rely on the intermediate [Pt(NH3)2Cl2], which is notoriously difficult to synthesize with yields rarely exceeding 70%. This bottleneck creates a supply chain dependency on a few major global manufacturers, driving up costs significantly.
Q: How does the new iodide-based route improve platinum recovery rates?
A: By utilizing potassium iodide to form the cis-diiododiammine platinum intermediate, the process achieves yields greater than 90%. This minimizes the loss of valuable platinum metal during purification steps, directly impacting the bottom line for precious metal processing.
Q: Is this synthesis method scalable for industrial catalyst production?
A: Yes, the patent explicitly states the method is suitable for mass production. The operations involve standard precipitation, filtration, and concentration steps that do not require exotic equipment or extreme conditions, facilitating easy scale-up from lab to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetraammine Platinum(II) Nitrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance precursors play in the development of next-generation catalysts and electronic materials. Our technical team has extensively analyzed the pathway described in CN101367557A and possesses the expertise to translate this laboratory-scale innovation into robust commercial reality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity specifications (>99%) and yield targets (>90%) are met consistently. Our rigorous QC labs are equipped to handle the precise elemental analysis required for platinum complexes, guaranteeing that every batch meets the stringent demands of global R&D centers.
We invite you to collaborate with us to optimize your supply chain for platinum-based materials. Whether you require custom synthesis or technology transfer for this specific nitrate complex, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the noble metal chemical market.
