Revolutionizing Platinum Catalyst Production with Chlorine-Free Water-Soluble Pt(IV) Complexes
The landscape of heterogeneous catalysis is undergoing a significant transformation driven by the demand for cleaner, more efficient, and durable catalyst systems, particularly in the automotive and新能源 sectors. A pivotal advancement in this domain is detailed in patent CN113278034B, which discloses a novel class of water-soluble Pt(IV) complexes, specifically trans-[Pt(NH3)4(OH)2](CH3COO)2 and [Pt(NH3)5(OH)](CH3COO)3. These compounds represent a paradigm shift away from traditional chloroplatinic acid and platinum nitrate precursors, offering a chlorine-free and nitrate-free alternative that addresses critical pain points in catalyst manufacturing. For R&D directors and procurement specialists seeking a reliable catalyst precursor supplier, understanding the structural and chemical advantages of these hexacoordinate octahedral complexes is essential. The invention leverages ammonia and hydroxide ions as internal ligands to form stable cationic species balanced by acetate anions, resulting in materials that combine high water solubility with exceptional thermal stability profiles suitable for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of supported platinum catalysts has relied heavily on chloroplatinic acid (H2PtCl6) and platinum nitrate solutions, both of which present substantial technical and environmental liabilities. Chloroplatinic acid, while effective for certain reforming catalysts, introduces chloride ions that are notoriously difficult to remove completely; these residual chlorides can migrate during high-temperature operation, leading to the corrosion of reactor components and the poisoning of active sites, thereby drastically shortening the catalyst's service life. Similarly, platinum nitrate, typically sold in 10% nitric acid solutions, poses severe handling risks due to the strong acidity which can damage sensitive support materials like activated alumina or modified activated carbon. Furthermore, the thermal decomposition of these traditional precursors often releases corrosive acid mists such as HCl and HNO3, creating hazardous working conditions and necessitating expensive scrubbing systems to meet environmental compliance standards. The presence of impurities such as sulfur, phosphorus, or alkali metals in lower-grade precursors can further exacerbate catalyst deactivation through sintering or chemical poisoning.
The Novel Approach
In stark contrast, the novel synthetic route described in the patent utilizes a mild, aqueous-based methodology starting from commercially available Pt(II) tetrammine nitrate. This approach avoids the use of harsh halogenated reagents entirely, instead employing axial oxidation with hydrogen peroxide followed by anion exchange and neutralization with acetic acid. The resulting Pt(IV) acetate complexes are ionic compounds that dissociate readily in water to release stable complex cations, ensuring uniform distribution on negatively charged carrier surfaces during the impregnation process. This method not only eliminates the risk of chloride-induced corrosion but also simplifies the waste treatment process by avoiding the generation of nitrogen oxides or chlorine gas during calcination. The ability to tune the physicochemical properties of the precursor through ligand modification allows for precise control over the final metal dispersion, a critical factor for maximizing catalytic activity in applications ranging from automotive exhaust purification to fuel cell electrodes.
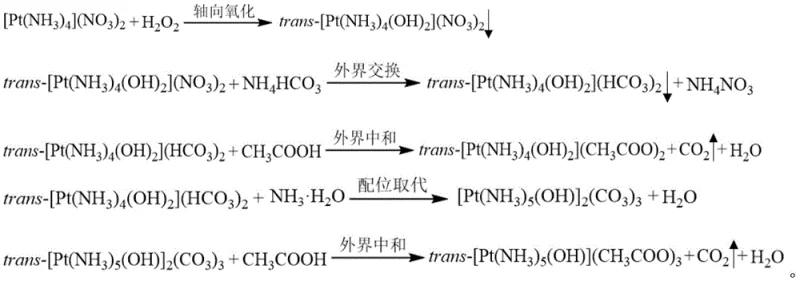
Mechanistic Insights into Pt(IV) Coordination and Thermal Decomposition
The chemical efficacy of these new precursors lies in their unique coordination geometry and decomposition kinetics. Unlike the square planar geometry typical of Pt(II) species, the Pt(IV) complexes adopted in this invention feature a six-coordinate octahedral structure. This spatial configuration provides a larger steric volume and greater kinetic stability, which facilitates better dispersion of the active platinum component upon reduction. The internal ligands, consisting of ammonia and hydroxide groups, are arranged such that the hydroxide groups occupy trans positions, creating a symmetric and robust cationic core. During the synthesis, the axial oxidation step converts the Pt(II) center to Pt(IV), enabling the subsequent substitution of external anions. The replacement of nitrate or bicarbonate ions with acetate groups is crucial, as the acetate anion acts as a charge balancer while simultaneously enhancing the water solubility of the entire complex to levels as high as 100 g/L at room temperature.
Furthermore, the thermal behavior of these complexes offers distinct advantages for the calcination stage of catalyst production. Upon heating in air, the complexes undergo a clean thermal decomposition reaction at relatively low temperatures, typically below 300°C. This process yields metallic platinum along with volatile byproducts such as ammonia, carbon dioxide, and water vapor, leaving no solid residue or corrosive ash behind. The narrow decomposition temperature range ensures a controlled reduction process that minimizes the risk of platinum particle agglomeration or sintering, which are common causes of activity loss in heterogeneous catalysts. This clean decomposition profile is particularly beneficial for preparing high-loading catalysts where uniform metal distribution is paramount for achieving optimal conversion rates of pollutants like CO, NOx, and hydrocarbons.
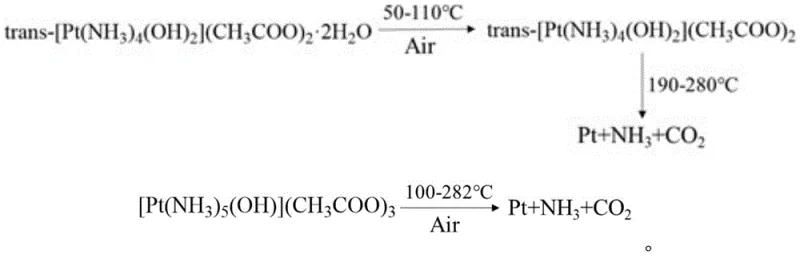
How to Synthesize Water-Soluble Pt(IV) Acetate Efficiently
The synthesis of these high-performance precursors is designed to be scalable and operationally simple, relying on standard unit operations familiar to fine chemical manufacturers. The process begins with the dissolution of the Pt(II) starting material followed by controlled oxidation, precipitation, and final neutralization. Each step is optimized to maximize yield and purity, ensuring that the final product meets the stringent specifications required for catalytic applications. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and purification protocols, are outlined in the guide below to assist technical teams in replicating this advanced chemistry.
- Oxidize commercially available [Pt(NH3)4](NO3)2 with excess hydrogen peroxide to form trans-[Pt(NH3)4(OH)2](NO3)2.
- Perform anion exchange using ammonium bicarbonate to precipitate trans-[Pt(NH3)4(OH)2](HCO3)2.
- Neutralize the bicarbonate intermediate with acetic acid to obtain the final water-soluble acetate complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of these chlorine-free Pt(IV) complexes translates into tangible operational improvements and risk mitigation. The elimination of corrosive halogens and strong mineral acids from the precursor supply chain significantly reduces the maintenance costs associated with storage tanks, piping, and dosing equipment, as the materials are compatible with standard stainless steel infrastructure. Moreover, the high water solubility of the acetate complexes allows for the preparation of concentrated stock solutions, reducing shipping volumes and logistics costs compared to dilute nitric acid solutions. The stability of these complexes in aqueous media ensures a longer shelf life and consistent quality upon delivery, minimizing the risk of batch-to-batch variability that can disrupt production schedules. By switching to this cleaner technology, manufacturers can achieve substantial cost savings through simplified waste treatment processes and extended catalyst lifespan, without compromising on performance metrics.
- Cost Reduction in Manufacturing: The transition to acetate-based precursors eliminates the need for expensive corrosion-resistant equipment and complex off-gas scrubbing systems required for handling chloroplatinic acid or nitric acid. This qualitative shift in process chemistry reduces capital expenditure on specialized infrastructure and lowers ongoing operational expenses related to safety compliance and hazardous waste disposal. Additionally, the high yield and purity of the synthesis route minimize raw material wastage, contributing to a more economical overall production cost for the catalyst manufacturer.
- Enhanced Supply Chain Reliability: Sourcing precursors that are stable and non-hazardous simplifies logistics and regulatory compliance, ensuring smoother cross-border transportation and storage. The robustness of the synthesis method means that production can be scaled up rapidly to meet surging demand in the automotive and energy sectors without the bottlenecks often associated with handling highly reactive or unstable chemical intermediates. This reliability is crucial for maintaining continuous production lines and meeting just-in-time delivery commitments to downstream catalyst formulators.
- Scalability and Environmental Compliance: The benign nature of the decomposition byproducts (NH3, CO2, H2O) aligns perfectly with increasingly strict environmental regulations regarding industrial emissions. Scaling this process does not introduce new toxicological hazards, making it easier to obtain permits for facility expansion. The ability to produce high-purity catalysts with minimal environmental footprint enhances the corporate sustainability profile, a key differentiator in today's green-conscious market.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, application, and handling of these novel platinum complexes. The answers are derived directly from the experimental data and characterization results presented in the patent documentation, providing a factual basis for evaluating their suitability for your specific catalytic systems.
Q: Why are chlorine-free platinum precursors critical for fuel cell catalysts?
A: Chlorine residues from traditional chloroplatinic acid precursors can poison the catalyst active sites and corrode support materials, significantly reducing durability and performance in fuel cell applications.
Q: What is the thermal decomposition behavior of these Pt(IV) complexes?
A: These complexes decompose at relatively low temperatures (below 300°C) in air to generate metallic platinum, ammonia, and carbon dioxide, avoiding the release of corrosive acid mists.
Q: How does the water solubility compare to traditional Pt(II) carboxylates?
A: Unlike traditional Pt(II) carboxylates which have low solubility (less than 20 mg/ml), these novel Pt(IV) acetate complexes exhibit high water solubility up to 100 g/L, facilitating easier impregnation processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Water-Soluble Pt(IV) Complex Supplier
As the global demand for high-efficiency emission control and energy conversion technologies accelerates, the need for advanced catalytic precursors has never been more critical. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging deep expertise in coordination chemistry to deliver premium Pt(IV) complexes that redefine performance standards. Our state-of-the-art facilities are equipped to handle complex synthesis pathways with precision, ensuring that every batch meets rigorous purity specifications and consistency requirements. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the capacity to support your growth from pilot-scale trials to full industrial deployment. Our rigorous QC labs employ advanced analytical techniques to verify the absence of harmful impurities, guaranteeing that our precursors provide the clean foundation necessary for high-performance catalyst manufacturing.
We invite you to collaborate with our technical team to explore how these chlorine-free precursors can optimize your specific formulation processes. By requesting a Customized Cost-Saving Analysis, you can quantify the potential operational efficiencies and total cost of ownership benefits tailored to your plant's infrastructure. We encourage you to contact our technical procurement team today to索取 specific COA data and route feasibility assessments, ensuring that your next generation of catalysts is built on the most advanced and reliable chemistry available in the market.
