Advanced Rhodium-Catalyzed C-C Bond Activation for High-Purity Chiral Intermediates
Introduction to Next-Generation Chiral Boronate Synthesis
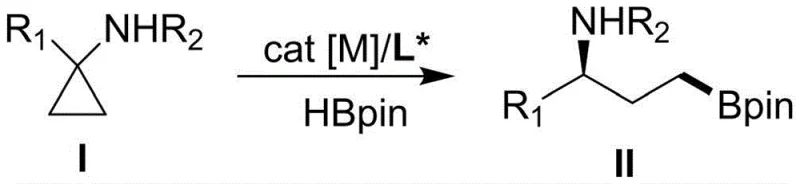
The pharmaceutical industry is currently witnessing a paradigm shift towards single-enantiomer medicines, driven by stringent regulatory requirements and the need for higher therapeutic efficacy with reduced side effects. In this context, Patent CN114605458A introduces a groundbreaking methodology for preparing chiral gamma-amino boric acid esters, which serve as critical building blocks in modern drug discovery. This technology leverages the unique reactivity of transition metals to activate carbon-carbon bonds within cyclopropane rings, a strategy that fundamentally differs from traditional functionalization approaches. By utilizing pinacol borane as a boron source under the synergistic action of a rhodium catalyst and a specialized chiral ligand, this process enables the precise cleavage of C-C bonds to generate a series of enantiomerically enriched gamma-amino borates. The significance of this invention lies not only in its chemical novelty but also in its ability to address long-standing challenges in asymmetric synthesis, such as controlling regioselectivity and suppressing competitive side reactions.
For R&D directors and process chemists, the implications of this patent are profound. The described method effectively circumvents the common pitfall of competitive C-H bond borylation, which often plagues traditional routes and leads to difficult-to-separate impurity profiles. Furthermore, the catalytic system demonstrates an exceptional ability to distinguish between distal and proximal C-C bonds within the cyclopropane ring, promoting near-end selective hydroboration. This level of control is paramount when synthesizing complex molecular architectures where the position of the boron moiety dictates the success of downstream cross-coupling reactions. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances allows us to offer clients not just a commodity, but a strategically optimized pathway for their API development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral alkyl borates has relied heavily on asymmetric C-H bond borylation mediated by transition metals. While mature catalytic systems exist for this transformation, they often suffer from inherent limitations regarding regioselectivity and enantioselectivity control, particularly when dealing with substrates containing multiple reactive sites. A major challenge in the prior art is the difficulty in distinguishing between different C-C bonds in cyclopropane derivatives, which possess significant ring tension. Without a highly specific directing group or catalyst system, the ring-opening reaction can proceed non-selectively, leading to a mixture of regioisomers that are costly and time-consuming to separate. Additionally, the competitive borylation of C-H bonds adjacent to the reaction site frequently occurs, generating byproducts that compromise the purity of the final intermediate. These issues create substantial bottlenecks in the commercial scale-up of complex pharmaceutical intermediates, where impurity thresholds are increasingly strict.
The Novel Approach
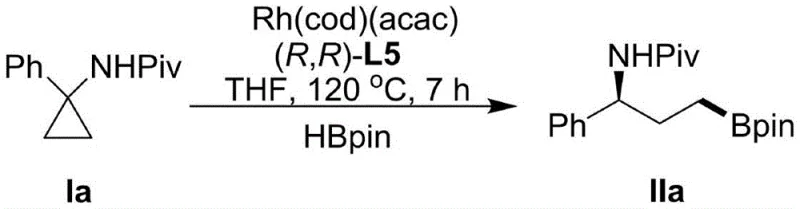
The novel approach detailed in the patent overcomes these hurdles by employing a rhodium-catalyzed system specifically designed for C-C bond activation of cyclopropanes bearing an amide directing group. This strategy transforms the inherent ring strain of the cyclopropane into a driving force for selective bond cleavage. Unlike conventional methods that struggle with site selectivity, this system promotes proximal selective hydroboration, ensuring that the boron atom is installed at the desired position relative to the amino group. The use of pinacol borane as the boron source further enhances the stability and utility of the resulting product. By shifting the focus from C-H activation to C-C activation, the method opens up new chemical space for synthesizing chiral gamma-amino borates that were previously inaccessible or economically unviable. This represents a significant technological leap, providing a robust platform for the cost reduction in API manufacturing by streamlining the synthesis of high-value chiral building blocks.
Mechanistic Insights into Rhodium-Catalyzed C-C Bond Activation
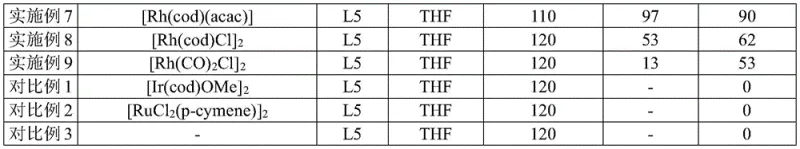
The core of this innovation lies in the sophisticated interplay between the rhodium catalyst and the chiral ligand environment. The reaction initiates with the coordination of the rhodium center to the amide directing group on the cyclopropane substrate, which pre-organizes the molecule for selective C-C bond insertion. The choice of ligand is critical; the patent highlights the superiority of chiral monophosphites, specifically ligand L5, over other candidates like bis-oxazolines (L1, L2, L3) or other phosphorus ligands (L4). The bulky binaphthyl backbone of L5 creates a highly defined chiral pocket around the metal center, which sterically biases the approach of the boron source and the subsequent migratory insertion step. This steric differentiation is what allows the catalyst to distinguish between the proximal and distal C-C bonds of the cyclopropane ring, a feat that simpler ligand systems fail to achieve with high fidelity.
Furthermore, the mechanism effectively suppresses the thermodynamic preference for C-H borylation, which is often a competing pathway in rhodium-catalyzed reactions. The electronic properties of the [Rh(cod)(acac)] precursor, combined with the electron-rich nature of the monophosphite ligand, tune the reactivity of the metal-hydride species formed in situ. This tuning ensures that the oxidative addition into the strained C-C bond is kinetically favored over C-H activation. The result is a catalytic cycle that delivers products with excellent chemical, regional, and enantioselectivity, with ee values reaching as high as 99% in optimized examples. For technical teams, this mechanistic clarity offers confidence in the reproducibility of the process, as the selectivity is encoded in the catalyst structure rather than relying on fragile kinetic control.
How to Synthesize Chiral Gamma-Amino Boric Acid Esters Efficiently
The practical execution of this synthesis is designed to be operationally simple while maintaining rigorous control over reaction parameters to ensure high stereochemical purity. The process begins with the preparation of the cyclopropane substrate containing the necessary amide guiding group, followed by the key catalytic step involving the rhodium complex and pinacol borane. The reaction is typically conducted in dry tetrahydrofuran (THF) under an inert atmosphere to prevent catalyst deactivation by oxygen or moisture. Heating the mixture to approximately 120°C facilitates the overcoming of the activation energy barrier for C-C bond cleavage. Detailed standard operating procedures for scaling this reaction from milligram to kilogram quantities are essential for maintaining consistency. The standardized synthesis steps outlined below provide a clear roadmap for implementing this technology in a GMP-compliant environment.
- Prepare the cyclopropane substrate with an amide directing group by reacting the corresponding amine with an acid chloride in the presence of a base.
- Combine the substrate, rhodium catalyst (e.g., [Rh(cod)(acac)]), chiral monophosphite ligand (L5), and pinacol borane in an inert solvent like THF.
- Heat the mixture to 120°C under inert gas for 7 hours, then purify the resulting chiral gamma-amino boric acid ester via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers distinct advantages that translate directly into operational efficiency and risk mitigation. Traditional routes to chiral amino borates often involve multi-step sequences with resolution steps that inherently cap the maximum theoretical yield at 50%, creating significant material waste and cost inefficiencies. By contrast, this asymmetric catalytic method delivers the desired enantiomer directly with high selectivity, theoretically doubling the material throughput from the same amount of starting原料. This fundamental improvement in atom economy is a key driver for cost reduction in pharmaceutical intermediate manufacturing, as it reduces the burden on raw material sourcing and waste disposal infrastructure. Moreover, the high selectivity minimizes the formation of diastereomeric impurities, simplifying the purification process and reducing the consumption of chromatography media and solvents.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the high catalytic efficiency significantly lower the overall cost of goods sold (COGS). By avoiding the need for expensive chiral separating agents or recrystallization cycles that discard half the product, manufacturers can achieve substantial cost savings. The use of commercially available rhodium precursors and ligands further ensures that the catalyst system remains economically viable for large-scale production without relying on exotic or proprietary reagents that might pose supply risks.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system, which tolerates a wide range of functional groups including halogens, trifluoromethyl, and heterocycles, ensures a stable supply of diverse intermediates. This functional group tolerance means that a single platform technology can be applied to synthesize a broad library of analogs, reducing the need for developing entirely new processes for each new drug candidate. Consequently, this flexibility enhances supply chain continuity, allowing for rapid response to changing demand patterns in the pharmaceutical market without the lead time associated with process redevelopment.
- Scalability and Environmental Compliance: The method has been demonstrated to work effectively on a gram scale with maintained performance, indicating strong potential for commercial scale-up. The simplified workflow, which avoids harsh reagents and extreme conditions, aligns well with green chemistry principles. Reduced solvent usage due to higher yields and fewer purification steps contributes to a lower environmental footprint, helping companies meet increasingly stringent environmental, social, and governance (ESG) targets while maintaining compliance with global regulatory standards for chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-C bond activation technology. These answers are derived directly from the experimental data and advantageous effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows. The high enantioselectivity and yield data support the viability of this route for producing high-purity intermediates required for clinical and commercial applications.
Q: What is the primary advantage of this C-C bond activation method over traditional C-H borylation?
A: This method effectively avoids competitive C-H bond borylation and utilizes the ring strain of cyclopropane to achieve proximal selective hydroboration, resulting in superior regioselectivity and enantioselectivity (up to 99% ee).
Q: Which catalyst system provides the highest enantioselectivity for this transformation?
A: The combination of [Rh(cod)(acac)] as the catalyst and the chiral monophosphite ligand L5 in THF solvent at 120°C has been demonstrated to provide the highest enantiomeric excess, reaching up to 97-99% ee.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the patent demonstrates successful gram-scale preparation with maintained high yields and stereoselectivity, indicating strong potential for commercial scale-up in the manufacturing of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Gamma-Amino Boric Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed C-C bond activation technology for the next generation of chiral therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise measurement of enantiomeric excess required for chiral intermediates. We understand that the consistency of chiral building blocks is non-negotiable for API registration, and our quality management systems are designed to guarantee batch-to-batch reproducibility that meets the highest international standards.
We invite you to collaborate with our technical team to explore how this advanced synthesis method can optimize your specific project requirements. Whether you are looking to reduce lead time for high-purity pharmaceutical intermediates or seeking a strategic partner for long-term supply, we are ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your molecule. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your goals for cost-effective and sustainable chemical manufacturing.
